Explore AAV and Lentivirus Plasmids: RG/HQ/GMP
Whatever stage of development you’re in, our off-the-shelf adeno-associated virus (AAV) and lentivirus plasmid products offer reduced development costs, risks, and production timelines for your cell or gene therapy program. Research Grade, High Quality, and GMP quality standards are available to support all stages of pre-clinical, clinical and commercial supply with associated CMC support, batch documentation, Certificate of Analysis (COA) and stability data.
Standard pack sizes are available from 2 mg – 60 mg. Custom quantities up to multi-gram scales can be provided on request.
- Immediate, reliable supply
- Fit-for-purpose, consistent quality
- Kanamycin antibiotic resistance, compliant with current regulatory guidelines
- Animal component-free production, reduces contamination risk while supporting 3Rs initiatives
- License and royalty-free, from research to commercial
- Proven record of success in viral vector production
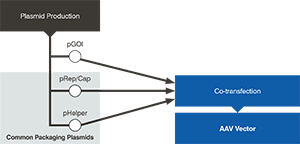
AAV Rep/Cap and Helper
To save time and streamline your path to clinic, we pre-manufacture common AAV packaging plasmids (Rep/Cap and Helper) to accelerate AAV production without compromising on quality.
Learn more
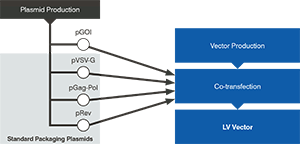
Lentivirus Gag-Pol, Rev, and VSV-G
Standardized off-the-shelf packaging plasmids (Gag-Pol, Rev, and VSV-G) for third-generation lentiviral vector production offer significant time and cost savings.
Learn more
“We are committed to supporting advanced therapy developers with a rapid supply of critical plasmid DNA starting materials to expedite their programs. The addition of in-stock packaging plasmids for RG, HQ, and GMP grade plasmids, combined with Charles River’s established plasmid and viral vector CDMO capabilities, will help to both accelerate timelines and ensure the highest quality product with one partner.”
Andrew Frazer, PhD, Associate Director, Scientific Solutions, Gene Therapy CDMO Services, Charles River
Need Custom Plasmid Production for Your Viral Vector Manufacturing?
With plasmid DNA also manufactured on request, if you do not see the catalog AAV/LVV plasmid product you want, our experienced CDMO team offers a full range of solutions for applications ranging from preclinical research studies to clinical trials and commercial scale.
Explore Plasmid Services
Need Custom CGMP AAV Packaging?
We are the world leader in AAV manufacturing, with a 17-year CGMP production track record. We have the capability and capacity to manufacture AAV viral vectors for applications ranging from preclinical to clinical, and commercial settings.
Learn more
Need Custom CGMP Lentivirus Packaging?
We have expertise in CGMP production of lentiviral vectors in HEK293 and 293T producer cell lines (adherent or suspension cells).
Learn more