Find Human Bone Marrow Products that Best Suit Your Project Needs
Human bone marrow aspirate contains a large concentration of primitive CD34+ hematopoietic stem cells as well as mesenchymal stem cells and is used in the development of cell-based therapies and regenerative medicine for the treatment of various disease types including blood disorders and other immune or genetic diseases. Due to the self-renewal and multipotent nature of bone marrow stem cells, there is great interest in their potential applications for next-generation drug discoveries, cell replacement studies, and self-repair.
Safeguard Your Stem Cells: Strategies to Preserve Bone Marrow Stability
This webinar will explain strategies to preserve fresh bone marrow stability by comparing the results of a recent study where bone marrow aspirate was stored at controlled room temperature (CRT) and cold temperature (2-8 oC) over a three-day period. Watch on Demand
Charles River provides high-quality human bone marrow-derived products, including fresh bone marrow aspirate and isolated stem cells, to help support your therapies from discovery to commercialization. We offer both research-use-only (RUO) as well as GMP-compliant fresh bone marrow aspirate to support your basic research needs, process development, and clinical studies.
Human Bone Marrow-Derived Products
-
Fresh Bone Marrow Aspirate (RUO)
Human bone marrow aspiration is performed at our FDA-registered collection center by a qualified clinician from healthy human donors who have consented under an IRB-approved protocol. The aspirate is drawn from the posterior iliac crest (top ridge of the back of a hip bone) using a syringe containing either citrate dextrose solution A (ACD-A) or heparin as an anticoagulant.
Our RUO (HemaPrime™) bone marrow aspirate is available in either 50ml or 100ml sizes and is shipped in a sterile gas-permeable transfer pack with coupler.
The concentration of white blood cells (WBC) and CD34+ hematopoietic stem cells that are present in the bone marrow aspirate is subject to variability and is donor-dependent. The WBC concentration may range between 10 million to 40 million WBC/mL with an average of 2.39 billion cells per collection with >90% viability (Figure 1), and the concentration of CD34+ cells in the bone marrow aspirate may range between 1% and 2%. (Figure 2).
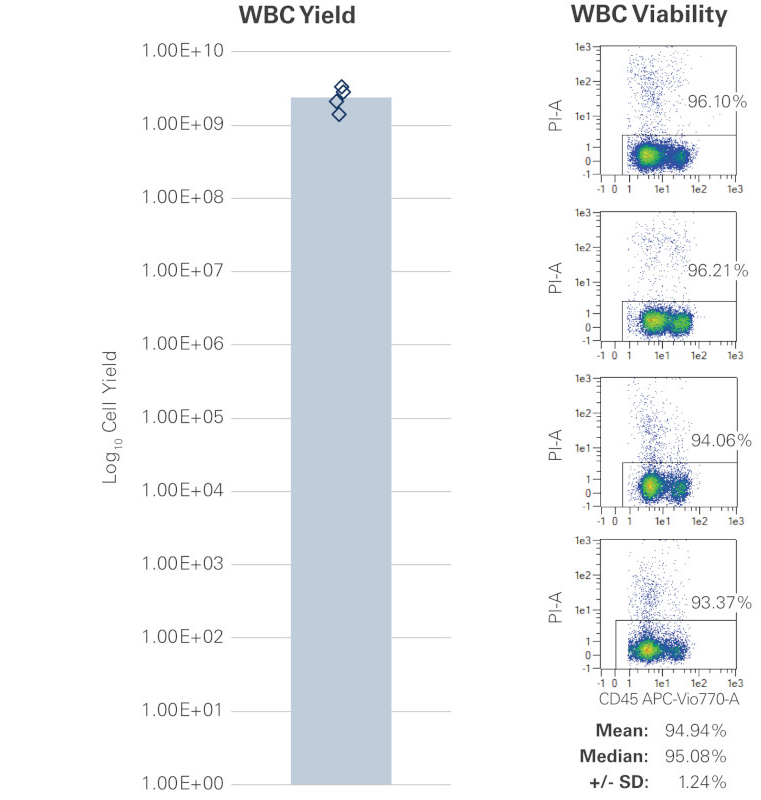
Figure 1 Representative bone marrow aspirate samples were assessed for white blood cell (WBC) yield and viability. As with other raw materials, the yield is variable and within this sampling ranged from a max of 3.29E+09 to a min of 1.38E+09 (blue diamonds) with an average WBC yield of 2.39E+09 (light blue bar). High viability was observed with all samples averaging 94.94%.
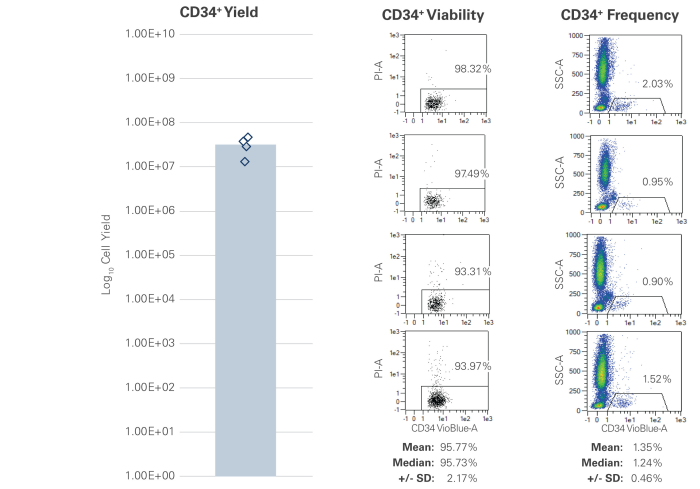
Figure 2 Representative bone marrow aspirate samples were assessed for CD34+ hematopoietic stem cells (HSC) yield and viability. The percentage of CD34+ cells was variable within this sampling and ranged from a max of 2.03% to a min of 0.90%. Average yield was 3.15E+07 (teal bar). As with WBCs, CD34+ HSC viability was high and averaged greater than 95.77%.
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Fresh Bone Marrow Aspirate (GMP)
GMP-compliant (GMPrime™) human bone marrow is collected in our donor center using stringent quality management systems and consistent, high-quality materials. The donor center is FDA-registered, AABB-accredited, CLIA-certified, and state-licensed.
Our GMPrime™ bone marrow aspirate, and its research-use-only (RUO) counterpart, HemaPrime™ bone marrow aspirate, are collected following consistent collection procedures and processes. Thus, partnering with Charles River for RUO and GMP-compliant raw materials during the early stages of development can simplify and streamline your journey to clinical work and commercialization.
GMP compliance adds an extensive level of quality review and documentation, such as the inclusion of a Master Batch Record (MBR), that serves to standardize raw materials and processes, eliminating most external sources of variability with the exception of those associated with the donor. The transition to GMP-compliant materials is a necessary step in the development of cell therapies to satisfy regulatory guidelines and ensure the safety of recipients of these therapies.
In addition to GMP fresh human bone marrow aspirate, our GMP-compliant portfolio also includes fresh and cryopreserved GMP leukopaks, and fresh and cryopreserved GMP PBMCs. Our GMP-compliant cellular starting material stands unparalleled in the market, exhibiting exceptional quality and reliability that meets or exceeds the GMP requirements of our clients.
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs)
In addition to fresh bone marrow aspirate, we offer cryopreserved, isolated CD34+ hematopoietic stem and progenitor cells (HSPCs). CD34+ hematopoietic stem cells are multipotent and can give rise to all cell types in blood including white blood cells, platelets and red blood cells. Our CD34+ HSPCs are isolated onsite from freshly collected bone marrow aspirate. The CD34+ stem cells are isolated via automated immunomagnetic cell separation techniques and are immediately cryopreserved to maintain cell viability. Generally, bone marrow-derived CD34+ stem cells are associated with low risk of graft-versus-host disease (GVHD), but are slow to engraft. Bone marrow-derived CD34+ stem cells are available in four different cryopreserved vialing formats including 100K, 250K, and 500K cells/vial, as well as 1M cells/vial.
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Mesenchymal Stem Cells (MSCs)
Mesenchymal stem cells (MSCs) are cultured from freshly collected human bone marrow aspirate and are then cryopreserved at 1M cells/vial. MSCs are multipotent stromal cells that can differentiate into a variety of cell types including osteoblasts (bone), chondrocytes (cartilage), adipocytes (fat), myocytes (muscle), and neurons (nerve). Because MSCs are undifferentiated cells, they can self-renew and have a high proliferative capacity.
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
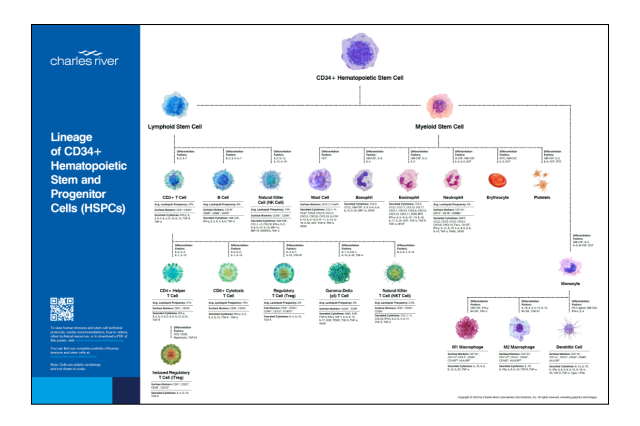
Lineage of CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs)
Receive a complimentary poster to access information on immune cell markers, differentiation factors, cytokines, technical protocols, media recommendations, how-to videos, and more. Download Poster