Safety Assessment
|
Daniel Klein
AI Drives Sustainable Histology at Charles River Laboratories
Being able to streamline pathology and control staining variability also turns out to be a big win for the environment
As near as historians can tell, the first systematic, scientific approach to medicine — the earliest application of the scientific method — was over 900 years ago during the reign of the Abbasid Caliphate. While much progress has happened since then, the basic idea remains the same: through observation, one can determine the cause of disease. The essential tenet of medical science is pathology — the process of identifying the causes and effects of disease.
The conceptual basis of modern scientific medicine dates back several centuries to the work of 19th century pathologist Rudolf Virchow . His legacy is cellular pathology, the study of disease in organs, tissues and cells. By observing cells under a microscope — ‘microscopy’ — he could pinpoint cellular dysfunction, which he believed caused most of the diseases plaguing mankind. And, for all the many advances in medical science since then, the basic theory has not changed. Even the physical processes of observing said dysfunction have remained quite similar for many decades.
For example, in a cancer biopsy, a slice is cut from the tumor. The living cells are soaked in fixative chemicals, to ensure they keep their shape throughout the histology process. The cells are coated in paraffin, and the resulting ‘block’ is sliced into tiny microscope-slide sections. Each slide is painstakingly dyed — “stained” — using a standardized variety of chemicals, in order to ensure that the details of each are clearly differentiated by the pathologist tasked with identifying exactly what they are seeing. With few variations, this is how it’s been done. And, insofar as it’s possible to ascribe monolithic opinions of large groups of people, this is the way they like it — and they are very particular about it.
There is a deep pervasive irony in this, since despite the large-scale attempt to bring standardization and consistency to pathology, it remains inherently difficult.
Dr. Dan Rudmann, Scientific Director of Digital Toxicologic Pathology at Charles River Laboratories, doesn’t mince words: “These stains — that are theoretically very simple and standardized — can look slightly different in every lab… we have an imperfect system right now.” And while the days of pathologists individually hand-staining their slides are mostly past us, variability is still a potential problem for pathologists, as are the health and environmental impacts of widespread creation and use of these chemicals. And even when all variables are controlled, pathologists are still at the mercy of the block. Each tissue slice can vary as much from each other as from similar samples at different labs. There are even significant variations from technician to technician and from day to day. As the field stands today, true standardization is a pipe dream.
The solution to many of these problems may well be on the horizon, though, thanks to whole slide image (WSI) digitalization technology and ‘deep learning’ AI algorithms. In 2018, bioengineers from UCLA created an Artificial Intelligence-based method that transformed digitally imaged unstained tissue slides to virtually stained tissue sections that match the standardized histology stains without the use of chemicals — in short, digital staining. The results were promising, and the bioengineers started a company — Pictor Labs — to develop the technology further. “ In a blind study, the pathologists were unable to tell the difference,” says Christopher Wilson, Chief Business Officer at Pictor Labs. The digital stains don’t just look right, CEO Yair Rivenson explains, “the diagnoses they were rendering were the same.” Not only that, several of the pathologists in the study were unaware that any of the stains they were using for their diagnostics were digitally generated. “Our long-term outlook is to replace the staining workflow in histology… we think we’ll be able to do it virtually for everything,” says Rivenson.
While this represents an enormous potential for streamlining pathology, controlling staining variability, and generating an unprecedented variety of virtual stains on the same tissue section, the biggest impact may end up being environmental — eliminating or reducing the use of chemicals, thereby reducing waste, and further limiting human exposure. It was this potential that led Charles River Laboratories to select it as a Sustainability Fund project. (Charles River established the Capital Fund last year to identify and support sustainable projects including new construction, facility retrofits, and the procurement of more sustainable equipment.) Wilson has no illusions about what drove their partnership, “We get to a business discussion, then all of a sudden, ‘By the way, doesn't this save a lot of water?’ It’s one of those things that, frankly, we kind of overlooked at the beginning, and now it's one of our main focuses.”
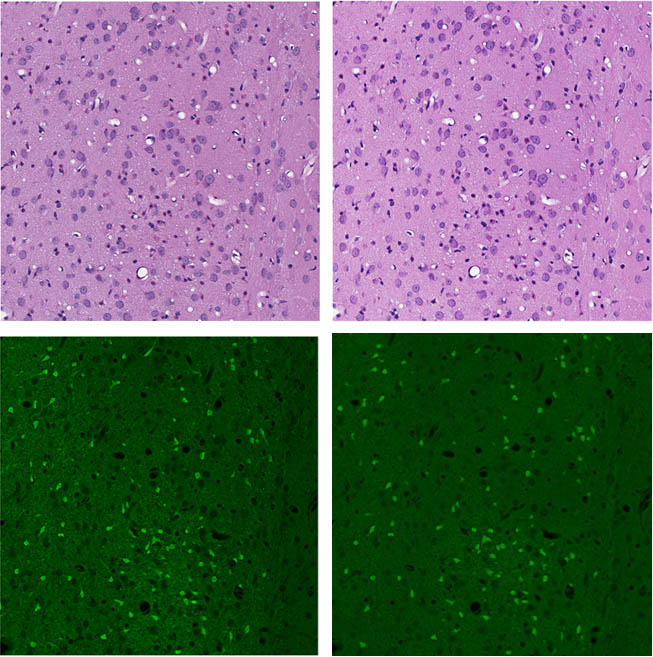
diagnostics and ID of neuron injury (left) and equivalent chemical free AI-based virtual stains (right).
The project was piloted at Charles River’s Mattawan, Michigan site, prior to receiving the sustainability grant. “They were able to show really nice staining of rat brain sections as well as one special stain,” says Dr. Esther Crouch, a veterinary pathologist on Rudmann’s team. “And they showed that it accurately depicted all of the features we needed to see.” The project was successful, and they’re now moving forward with more extensive studies and with more Charles River sites involved. “It’s important to test the technology rigorously, so that we can make that decision whether or not it's scalable and really ready for prime time,” says Rudmann.
Despite plenty of caveats, the answer seems like it will eventually be yes. The potential upsides are enormous: virtual staining is potentially faster, more flexible and reproducible, cost effective, deployable on limited tissue specimens, better for health and the environment, and it’s digital. The primary concern, according to Rudmann is “the ability for us to get enough data to really be confident this won't result in virtually stained sections that cause us to miss findings or misinterpret findings in comparison to standard chemical-based stains.” But, Crouch says, that issue is “at the forefront of every pathologist's mind. So, we’ve got our work cut out for us to try and convince the whole cohort of [Charles River] pathologists that this technology is going to be trustworthy and rigorous.” Crouch and Rudmann are both optimistic and Crouch is especially effusive: “What I've seen so far has really blown my mind.”
Pathologists are a notoriously tough crowd. “We tend to be pretty detail-oriented, exacting, and critical. And that makes change difficult for us. This is not going to be something that we're going to just roll off the shelf… it's going to be a journey,” says Rudmann. But, he notes, “even with our normal stain, we have problems. So, we want this to be equal or better. I'd like it to be better, to be honest with you. That's my goal.”
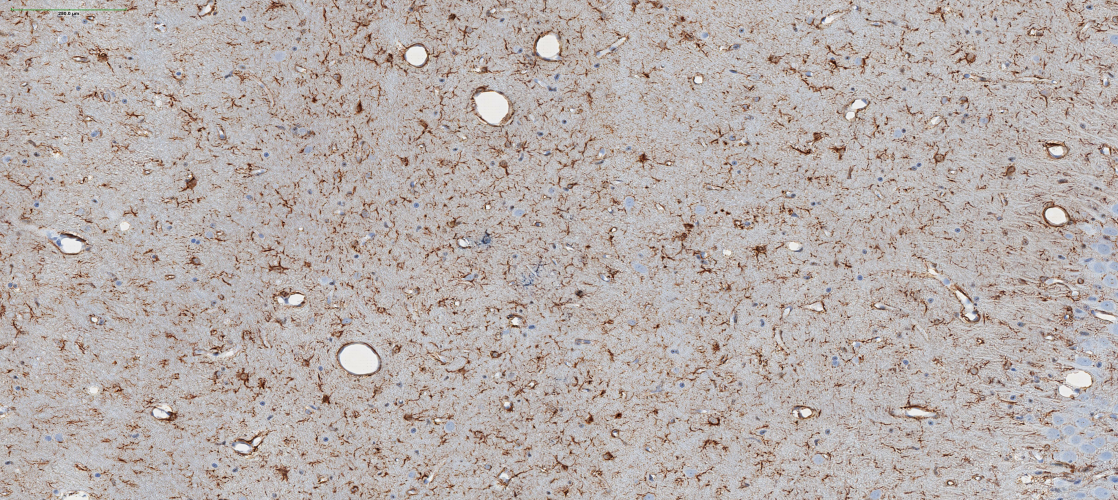
Image at top identifies astrocytes (brown) using a virtual glial fibrillary acidic protein or (GFAP).