Discovery
|
Julia Schueler, DVM
Better In Vitro Cancer Models for Better Cancer Drugs
Using innovation and state-of-the-art technology to find the right patient for the right therapy
Increased understanding of cancer biology, particularly how cancer cells communicate with their stromal and immune microenvironments is resulting in promising new therapeutic strategies for treatment of the disease. Paradoxically, this increased understanding has also brought us to the realisation that the most common preclinical models used for target validation and drug development fail to capture the complexity of human tumours. Despite major advances and new strategies in combating cancer, we still have high rates of attrition during cancer drug development because targets have not been validated in the context of the tumour microenvironment and lead compounds have not been optimised in systems that recapitulate the role of the tumour microenvironment on drug disposition and mechanisms of action.
Significance of the tumour microenvironment
Significant advances in understanding that tumour cells are not autonomous came from studies investigating the role of the extracellular matrix (ECM; the protein components of the tumour microenvironment) on cancer cell behaviour. Publications from pioneers in the field such as Mina Bissell at the Berkeley Lab and the late Zena Werb, who was part of the University of California-San Francisco, showed that proteins that make up the extracellular matrix (ECM) can signal to cancer cells to control tissue and context-specific gene expression. This is a reciprocal process as cancer cells can shape the ECM to their benefit.
We also need to consider the fact that tumours are complex communities comprising many different cell types including fibroblasts, endothelial cells and cells from the immune system. The number and type of different cell types found within and around tumours influences responses to a whole range of different anti-cancer therapeutics. The dense, fibrotic ECM (or desmoplasia) accompanying tumour infiltration of high numbers of fibroblasts is associated with poor penetration of immune cells and also of anti-cancer drugs. This is one of the reasons why pancreatic adenocarcinoma is so poorly responsive to anything that has been thrown at it including chemotherapy, targeted agents and novel immunotherapies.
How to replicate the complex tumour microenvironment in vitro
So how do we go about reconstructing the complex tumour environment and architecture for target validation and lead optimisation? The use of soft agar, a prototypic 3D matrix, allows estimation of anchorage-independent colony formation, considered to be a key attribute of cancer cells. Soft agar assays allow determination of the effects of anti-cancer drugs on colony forming efficiency (CFE) but they are not suitable for the higher throughput formats used in drug discovery. As soft agar is derived from seaweed, it’s not surprising that it does not replicate the human 3D ECM. Agar is not suitable for looking at mixtures of different cell types and it is difficult to retrieve the cancer cells grown in agar to allow correlation of molecular changes with effects of drugs on colony forming efficacy.
More recently, “ultra-low attachment” or ULA plastic tissue culture plates and flasks have become available. As the name suggests, the culture surfaces have been treated such that cancer cells can’t attach and grow in 2D. Instead, the cancer cells form 3D spheroids representative of anchorage-independent growth. ULA tissue culture plates are more amenable to higher throughput formats but, because the cell colonies are not attached, liquid handling can be difficult. The other drawback is that ECM proteins can’t be included and so a major human tumour component is absent. However, it is easy to harvest the spheroids for further analysis and the format allows live cell imaging of drug effects with, for example, an Incucyte Live Cell Imaging system.
Many of the key findings concerning the role of the ECM on cancer cell behaviour came from experiments using the matrix produced by the Engelbreth-Holm-Swarm (EHS) mouse tumour which was commercialised as Matrigel. Matrigel is a complex mixture of collagen, fibronectin, laminin and varied growth factors and is representative of the basement membrane surrounding both normal and malignant cell structures. However, Matrigel also has significant drawbacks when it comes to its use in target validation and drug discovery. It’s very gloopy and prone to solidifying at room temperature so is not suited to automated liquid dispensing systems. There can be significant batch- to-batch variation in the number and type of growth factors bound to the matrix proteins which may confound experimental results. It is also not easy to retrieve tumour cells for molecular analysis after they have been grown in Matrigel.
As alternatives to Matrigel, several groups have been experimenting with the use of neutral hydrogels such as sodium alginate for culture of complex mixtures of cell types found in cancers. In one study, encapsulation of human breast cancer micro-fragments in sodium alginate maintains the complex tissue architecture and viability for up to 30 days. Like soft agar, sodium alginate is derived from seaweed and doesn’t resemble the matrix found in human tumours. However, and unlike soft agar, it has been found that the cells within the micro-fragments continue to produce their own matrix proteins including fibrillar collagen. Moreover, expression of cancer cell-intrinsic proteins such as the oestrogen receptor (ER) is also maintained. In many of the microstructures, viable infiltrating immune cells could be detected at the end of the culture period (Cartaxo et al, J Exp Clin Cancer Res 2020; 39:161).
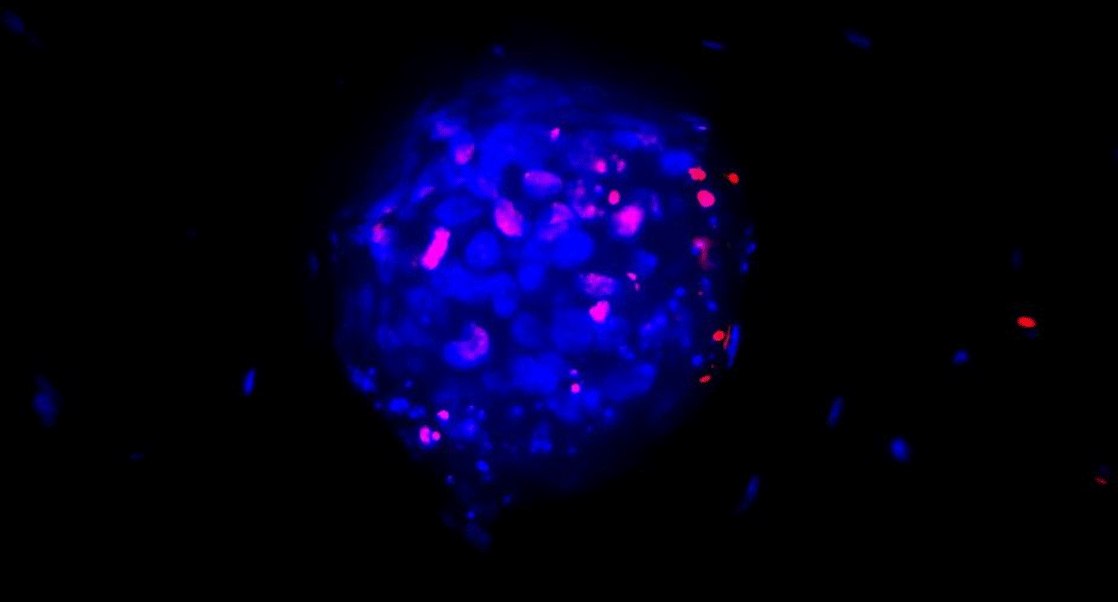
A novel 3D tumour model platform
The most recent advances in matrix “engineering” combine hydrogels with 3D printing technology to reconstitute the tumour microenvironment. Toward that end, Charles River Labs are now collaborating with Cypre , a company which has developed a novel 3D tumour model platform. In this system tumour cells are suspended in a chemically defined collagen-based ECM hydrogel (VersagelTM) and layered into microwell plates. A brief exposure to blue light induces cross-linking and the formation of a 3-dimensional, interconnected mesh network that more closely resembles the tumour matrix environment. The addition of fibroblasts to the cultures replicates the desmoplastic reaction seen in many tumour types e.g. pancreatic adenocarcinoma. Overlaying immune cells on top of the cultures or adding them directly to the mix coupled with the use of high content imaging techniques allows examination of whether immune cells can infiltrate and kill the tumour cells within the matrix. This is a significant advance in methodology that allows simulation of immunogenic and immune-excluded tumour types for testing of novel immuno-oncology drugs or combinations that could overcome immune-resistance.
In summary, there have been recent and highly significant advances in understanding how the microenvironment of tumours influences their behaviour and response to treatment. These advances have lead to the development of innovative methods of recapitulating the tumour microenvironment in vitro. The most sophisticated methods such as the Cypre platform combine novel matrices with a 3D “patterning” technique to generate highly defined model systems. These systems can be readily modified to represent the range of conditions found in clinical tumours. This should allow more secure validation of potential therapeutic targets and the testing of novel drugs in the context of the appropriate tumour microenvironment.
It is to be hoped that these innovations will lead to the development of more effective targeted- and immuno-therapies alongside an understanding of which patients will benefit the most from them. Additionally, these more relevant in vitro systems should reduce the high attrition rates associated with the drug discovery process and may reduce the need for in vivo models for target validation.
If you enjoyed this blog post, listen to this podcast on 3D platforms.
