
Laboratory Sciences
|
Dominic Clarke, PhD
Getting Cell Therapy Off to the Right Start
From starting material to personalized treatments
There is a great deal of discussion globally about the promise of cell therapies. A lot of this discussion centers on when, or even if, these therapies can go mainstream. There are some formidable challenges – cell therapies are difficult to make cost-efficiently, especially autologous therapies where treatment is personalized to the patient. Every patient and every donor are different, making manufacturing parameters hard to define. Scale-up is difficult. Cells can be expanded, but can consistent therapeutic efficacy be achieved from batch to batch and donor to donor? The primary obstacle to successful commercialization lies within the nature of the therapies themselves; cells are complex living materials. They are delicate, sensitive to their environment, and notoriously variable.
Beginning with the End in Mind
Probably the single greatest factor in determining both the efficacy and consistency of a cellular therapeutic is the cellular starting material from which it is made. From the point of collection to the point of administration, the number of healthy therapeutic cells in a cell therapy will determine the potency of the final product. Some therapeutic cell types, for example monocytes, cannot replicate, and consequently it becomes vitally important to retain cell health and viability during manufacturing.
Pharmaceutical developers are used to dealing with raw materials from several different suppliers, that can quickly be replicated with identical physical characteristics, and that have straightforward shipping and storage logistics.
Cell therapy manufacturing requires a whole new mindset. Starting materials are living biological products, sometimes with only a single viable source - the patient. For the first time, donors -often patients themselves - are part of the supply chain, which of necessity brings a closer connection to the patient.
Cell therapies are intended to treat serious diseases for which there are few, if any treatment options. Manufacturing batch failures have consequences beyond even serious financial concerns. What will the impact on the patient be? Is there enough starting material to repeat the process, and when can manufacturing and patient treatment resume? Has the exact cause of the failure been identified, and how does one reduce the risk that it will re-occur? The answers to these challenges exist, but the conversation regarding solutions needs to become more widespread if the industry is to succeed.
Donors are Key
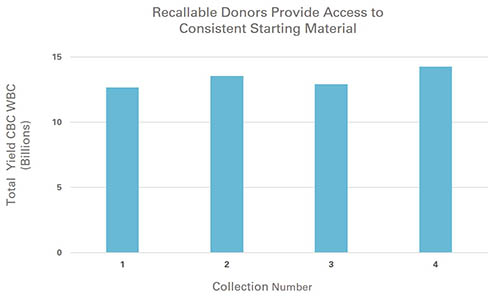
Donors are the key to successful cell therapies. There is no way to overstate this. Scaling up cell therapies means scaling up donor access. Scale-up of any therapy to commercialization requires establishing a reliable source of raw materials. For most cell therapy developers, this translates to finding a starting material supplier with access to a large donor network. But the reality is that it will take more than that. Finding the right donor, especially for a personalized therapy, is essential. However, when you need consistent access to specific physiological traits, it is just as important to have reliable, recallable donors.
This is where the “dedicated donor” model comes in. Dedicated donors are recallable donors who are reserved for the specific project for which they are the best match in terms of “desirable” donor traits. These traits could include high target cell yield, a specific donor demographic, or perhaps a rare genetic tissue marker. Cell therapy developers with dedicated donors have the benefit of being able to source essentially the same starting material repeatedly. Dedicated donor material is generally more consistent than material from multiple donors. It provides developers with the ability to pre-plan cell collection dates, or to cryopreserve multiple collections from the same donor. Greater scheduling flexibility and consistency in the manufacturing process will lower supply chain costs, and access to dedicated donors will help alleviate cell therapy starting material sourcing concerns.
Do Your Diligence
Working out supply needs and arranging adequate donor access early on are great ways to make sure your therapeutic gets off to a strong start. Another big hurdle the cell therapy industry needs to address is starting material variability.
Donor variability is inevitable; it is an intrinsic part of cell therapy and the reason why access to recallable donors is so critical. On the other hand, process variability can largely be controlled through the establishment of optimized cell handling processes and quality oversight systems.
A recent Australian publication examined the variability of cryopreserved CD34 cell products across 9 different laboratories. Each of these cell collections were intended for patient infusion, yet despite significant transit delays due to the COVID-19 pandemic, none had undergone any additional quality testing.
Rather alarmingly, the study found significant variability in post-thaw CD34 cell recovery and viability rates. In the publication’s study of 305 mobilized blood collections, both post-thaw viability and post-thaw recovery correlated with transit time and more complex sample handling. One of the publication’s main findings was that delays between cell collection and off-site cryopreservation resulted in significantly poorer cell viability and recovery. This is something which the industry can solve by shifting to a strategy where most starting materials are isolated and processed at the collection site. This mitigates risk from the start by truncating the cold chain and giving cell collection facilities better oversight of starting material quality.
It goes without saying that supply companies also need to be diligent about the collection process itself. Apheresis collection and bone marrow aspiration are highly technical skills requiring years of specialized training and experience.
The mother of one of the very first recipients of CAR T-cell therapy, Kimberly Schuetz, was herself an apheresis nurse. She often talked about how she realized that the most critical element to her young son’s chance of a cure was the apheresis material on which his treatment would be based. Kimberly Shuetz hand-picked a colleague of hers that she knew would be the most experienced person possible to perform the apheresis collection for her son. Skill, experience, and training all matter, so cell therapy developers need to be aware that choosing their starting material supplier is critical.
Ultimately, the story of cell therapy is the story of patients…cancer patients whose lives were given back to them; MS patients with their disease progression stopped, arthritis patients able to face the day without pain. Scientists and physicians have an obligation to patients to do what they can to make these living therapies successful. Because where there is life, there is hope.
Laboratory Sciences
Quality, scientific integrity, cost-effectiveness and regulatory compliance are essential considerations when selecting an outsourcing partner. Charles River is adept at balancing these key values within the most stringent of time frames to deliver comprehensive laboratory services from early screening through preclinical and clinical support.
Dominic Clarke is the Global Head of Cell Therapeutics for Charles River HemaCare.
