
Discovery
|
Christoph Eberle, PhD
Making Mice a Bit Human
How Charles River sites in Massachusetts are collaborating to develop a better cancer research model
Eighteen years before Max Planck theorized his radiation law, the Leiden University appointed a new professor of experimental physics. In 1882 while delivering his inaugural lecture Heike Kamerlingh Onnes, a cryogenics expert and later Nobel Laureate, suggested that every physics laboratory should bear the motto, “Door meten tot weten – through measurement to knowledge.”1
Onnes emphasized how significant the scientific method is for his own field of physics research. This goes for any research that relies on quantifying observations of natural phenomena. However, it is not enough to probe the unexplored without reasoning; and there needs to be assurance that measuring is done in the same manner irrespective of when and by whom. Its validity is at stake without a standard, a reference, a procedure commonly practiced and upheld. As theory needs experimental backup, the sense of experimental findings can be explained by a theoretical concept. This reciprocity is what modern natural sciences have been based on; its disciplines advanced through exploring measurable relationships. “Door meten tot weten – through measurement to knowledge.”
Immunological Pieces to the Disease Puzzle
But can the same motto also hold true for drug discovery, where translation science is far from exact? It can. Quantitative measurements accompany every step of the lengthy process of researching and developing medicines. The scientific method is applied to gain knowledge for the benefit of patients but also to collect proof before any drug, any therapy can be administered, or any device can be utilized. An important part of this process is understanding human pathophysiology and responses to experimental drugs in animal models. Insights gained from these models are only as good as they can translate to the human body. Mimicking the latter one as exactly as possible is therefore essential to find the best possible therapy. As we collect more immunological pieces to the puzzle of diseases yet to be cured such as cancer, the relevance of human-specific models is reemphasized in drug development. In cancer research humanized mice have been established and now may become part of the future of immuno-oncology.
Genetically, mice bred for these projects are a chimera of human immune cells and all other murine cell types. Within its legal boundaries this kind of engineering aims at getting better models to market, i.e. models more relevant to studying human immune responses when exposed to drug candidates from product pipelines. Likewise, the demand for translational oncology models is increasing. Can mice bearing a stable human immune phenotype offer the predictive value needed for evaluating new cancer drugs prior to clinical trials?
It turns out that to truly understand cancer the immune system matters, which begs the question: Why can’t we all clear cancer from our bodies? Ever since this recognition gained ground, researchers have grappled over which state the immune system needs to be in to mobilize an anti-tumor attack, and why we lose this fighting ability when we need it the most?
Despite all the treatment successes reported from checkpoint inhibitors, a universal immunotherapy for all cancer types has not been found. In fact, only a relatively small percentage of patients treated with immunotherapy (about 20%) mount a sustained response. One likely reason for the low number of responders is immunological diversity. Not every person’s immune system is identical to the next. Nor is every patient’s cancer identical in its genetic makeup, another reason driving the immunological response.
Humanized Models on the Rise
For decades biomedical research has been utilizing mice as disease models. An approximation to human diseases and a response to therapies each comes with its own limitations. On the drug development journey they may offer no shortcut. Yet, some can help us to make a step in the right direction. For a while now humanized immune system mice (HIS) have been one of the tools oncology research has been resorting to because we can better measure how well experimental immunotherapies induce responses to tumors. Improved models that fully mimic human immunity with implanted human tumors eventually could expedite the next generation of cancer immunotherapies.
However, major setbacks for humanized mouse model development continue to be the high costs and the risk of mice suffering from graft-versus-host disease (GVHD), which shortens their life cycle. Ultimately, refining humanized models challenges developers twofold: to copy human immunity as exactly as possible and to delay its loss because of GVHD as long as needed. What we know from transplantation immunology points to general biomarkers of tolerance in humans. In past clinical trials such markers could be detected with statistical significance in patient samples to test study hypotheses. Therefore, applying the same markers in screening humanized mice pre- and post-engraftment is worth exploring. In an internal collaboration across Charles River facilities in Massachusetts2 we continue monitoring the following markers among others in NCG mice engrafted with human hematopoietic cells:
- CD30: During acute GVHD T cells express more CD30. Elevated levels of its soluble form are detectable as well. Usually, the higher these levels are, the more severe the GVHD turns out.
- iNKT/T-cell ratio: It increases, as GVHD develops.
From a quality control perspective, we first need to understand whether these biomarkers prove useful for separating injected animals into “good” and “bad” batches prior to release for research studies. Besides that, exploring engraftment efficiency and xenotransplant rejection in more quantitative terms help us to tighten standards for humanization of a model and its baseline performance.
Flow Cytometry as Information Seeker
When it comes to determining the identity of cells, their numbers, state and fate, the obvious choice falls on flow cytometry. With the speed of fluorescence, it can collect a lot of information from any single cell. Thanks to its adaptability the technique is increasingly utilized. Flow cytometry allows verifying the humanization of a mouse’s immune system. Following engraftment questions pertain to how human the reconstituted immune system in the mouse is, more precisely the composition of its cell compartments and their capacity to self-renew. Prior immune profiling proves important for deciding which animals to enroll in pre-clinical studies. The optimal mouse for “humanization” needs to maintain peripheral hCD45+ frequencies above a predefined cut-off (typically ≥25% of live cells), while residual murine immune cells remain low. (See Figure 1.)
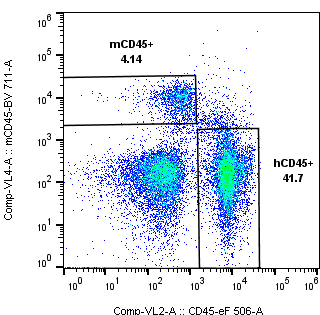
Simply put, human hematopoiesis must replace that of the mouse. Once injected human stem cells mature to functional immune cells to occupy the murine compartments over time. The more complete this takeover is, the better, i.e. the more workable the humanized mouse model can be for drug developers. Alternately, humanization can be defined as a quotient of detectable hCD45+ and mCD45+ cell frequencies. Yet even if a mouse is humanized, individual animals can still lack lymphoid and myeloid subset differentiation.
It is crucial in our iterative refining of the performance of these models on human-specific studies to examine which human stem cell material can be transplanted the best and when the graft starts getting rejected. Furthermore, it is important to know which human immune cell frequencies can be maintained in the periphery and in mouse tissues. In the clinic immune check inhibitors (ICI) and combinations thereof induce autoimmunity, presenting another factor to keep in mind for developing HIS. To serve as an ideal model, these mice must lose their own immune system and fully reconstitute a human one. Providing all its effector functions is equally needed. The closer these parts fit the human original, the more conclusive efficacy data can be.
In the end, biomedical research is about understanding human diseases as much as finding cures. These endeavors consume time and resources, require testing and revisiting of what has been learned. Despite all efforts in drug development cancer has dodged many “magic bullets”. Up to now it remains on our target list. Yet along the way we have practiced aiming at this target more and more precisely. What could accelerate our learning curve? Optimized animal models of disease and the columns of data we can gain from these will remain in our arsenal for the foreseeable future.
Our knowledge is just as less absolute as our measurements. By improving the latter one the first can be validated. Both are permanently evolving; both are permanently in danger to get lost or perverted. Over time the pieces that make up our knowledge fit together and though we keep sharpening our toolset and our scientific view, while holding Onnes’ maxim dear, we must not forget that whatever we can measure, it is never the whole.
References:
1 Laesecke, Arno. “Through Measurement to Knowledge: The Inaugural Lecture of Heike Kamerlingh Onnes (1882).” J Res Natl Inst Stand Technol. 107(3), 2002, 261–277. doi: 10.6028/jres.107.021.
2 Eberle, Christoph, and Steve Festin. “Flow Cytometry in Mouse Model Development: An Interview with Christoph Eberle and Steve Festin.” Bioanalysis Zone, Newlands Press, 20 May 2019, www.bioanalysis-zone.com/2019/05/20/flow-cytometry-mouse-model-development-interveiw-christoph-eberle-steve-festin_criver_spotl_flowcytom/.
