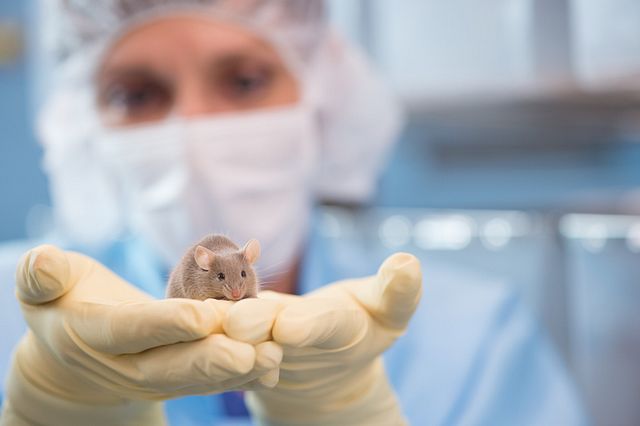
Research Models
|
Ken Henderson, PhD
In Search of Appropriate Animal Models
No vaccine candidate, including ones for COVID-19, can get to the finish line without animal safety data. Identifying it can be a challenge, though.
Vaccine development traditionally takes years, even decades. How then could companies deliver COVID-19 vaccines in a matter of months? Can we trust the process? These two questions have been dominating the airwaves even before the first three candidates in the pipeline were authorized by regulators, a mere 11 months after SARS-CoV-2 emerged in China. We hope Eureka’s five-part series, The Vaccine Journey, on how vaccines are developed—from discovery through manufacturing—helps answer some of these questions and hopefully reassures readers that vaccines are generally very safe products. Our third installment, from Ken Henderson, PhD, deals with animal models.
 Without a doubt, animal models are vital tools in the development and testing of new therapies. Most drugs and vaccines, including COVID-19 drugs and vaccines, cannot reach human trials without animal data because regulators require it to assess both functionality, but more importantly, safety
Without a doubt, animal models are vital tools in the development and testing of new therapies. Most drugs and vaccines, including COVID-19 drugs and vaccines, cannot reach human trials without animal data because regulators require it to assess both functionality, but more importantly, safety
Selecting the right models varies and the most obvious ones are not always the best ones. Consider COVID-19. Mice—easy to handle and breed—have long been the mainstay of biomedicine, and a good mouse model would be a boon for COVID-19 research. But mice repel SARS-CoV-2—the virus that causes COVID-19—because the key enzyme that the virus targets, a protein receptor called angiotensin-converting enzyme 2 (ACE2)— is structurally differently in mice than in humans. When humans are exposed to SARS-CoV-2 the effects can be devastating, while mice remain unscathed. Additionally, laboratory mice are exceptionally clean in respect to the absence of pathogens, and as a result have a poorly trained immune system comparable to that of a human neonatal child. Therefore, even if they were susceptible to SARS-CoV, they would not be a good model for measuring the host immune response to a vaccine.
In contrast, LVG Golden Syrian hamsters are highly susceptible to SARS-CoV-2, manifesting many of the same upper and lower respiratory tract symptoms seen in people. Moreover, there is extensive hamster data dating back to SARS-CoV-1 outbreak in 2002-4, which was caused by a coronavirus in the same subgenus as SARS-CoV-2. It is not surprising that the Golden Syrian hamster quickly emerged as a dominant choice in the study of multiple aspects of COVID-19, as were ferrets, which are common in the study of other respiratory disease, such as influenza. In the past 12 months, hamsters have informed dozens of studies related to the efficacy of vaccines and drugs, viral genetics, mask effectiveness, aging, convalescent plasma, side effects of COVID-19 and how neutralizing antibodies—Y-shaped antibodies that latch onto the surface of a virus and neutralize it—might work against SARS-CoV-2. Rabbits have also been useful. Humanized models, rodents re-engineered to contain components of the human immune system, are being developed to study respiratory aspects of COVID-19. Future models will be developed with a focus on establishing the virus target receptor in a model that most closely resembles the human immune and disease response.
coronavirus in the same subgenus as SARS-CoV-2. It is not surprising that the Golden Syrian hamster quickly emerged as a dominant choice in the study of multiple aspects of COVID-19, as were ferrets, which are common in the study of other respiratory disease, such as influenza. In the past 12 months, hamsters have informed dozens of studies related to the efficacy of vaccines and drugs, viral genetics, mask effectiveness, aging, convalescent plasma, side effects of COVID-19 and how neutralizing antibodies—Y-shaped antibodies that latch onto the surface of a virus and neutralize it—might work against SARS-CoV-2. Rabbits have also been useful. Humanized models, rodents re-engineered to contain components of the human immune system, are being developed to study respiratory aspects of COVID-19. Future models will be developed with a focus on establishing the virus target receptor in a model that most closely resembles the human immune and disease response.
But for assessing the efficacy of vaccines and drugs, certain large animal species are historically the preferred model because they are genetically closest to humans, and often the gatekeeper for human trials. Animal models can also flag dangers from COVID-19 vaccines and drugs. Testing experimental vaccines in animals requires immunizing animals and then “challenging” them with the virus to see how the animal’s immune system responds. These experiments must be conducted in a biosafety Level 3 lab due the ability of the virus to cause disease in humans.
Due to the urgent need for a vaccine, manufacturers were given regulatory approval in the US and in Europe to simultaneously test their vaccines on large and small animals while they were conducting Phase 1 clinical trials. This unusual step was taken given the tremendous public health need for a vaccine to control COVID-19. In order to conduct these studies, the facilities are not the only part of the equation that need to be clean. This begins with animals to be used for this work must live in “clean” environments that are necessary to prevent infection with bacteria and viruses that may interfere with laboratory results.
Caring for Precious Animal Models
Animal husbandry and care also ultimately matter in drug development in order to protect their health and well-being. Variation in animal models caused by the presence of pathogens, environmental stresses or genetic variance could confound research by undermining consistent and meaningful data. To address part of this, animal research facilities must obtain and maintain animals in a health-privileged environment. Although some studies that research behavior may use “wild caught” rodents, virtually all rodents are purpose-bred and raised as pathogen-free in an environment that caters to specific model needs and enrichment. This includes nutritious formulated feed and bedding that has been decontaminated so no pathogens are introduced. For good reasons the veterinary care and routine observation of health given to research models far exceeds what most can provide to our own pets at home. We rely on these precious animals for our own health and safety.
Few of us would ever get a vaccination or unseal a bottle of contact solution if we thought they were contaminated, so responsible laboratories devote considerable resources making sure bacteria and fungi don’t end up in the supply chain. Furthermore, a negative impact on human  health by a vaccine goes beyond the presence of opportunistic or primary pathogens. Historically, unchecked compounds used as human therapies have resulted in toxic impacts that can alter the recipient or even the next generations. Therefore, the inclusion of safety assessment performed in animal models prior to releasing for human used is a requirement before human clinical trials can begin. Routine sampling and testing in our research animal reference testing laboratories is standard.
health by a vaccine goes beyond the presence of opportunistic or primary pathogens. Historically, unchecked compounds used as human therapies have resulted in toxic impacts that can alter the recipient or even the next generations. Therefore, the inclusion of safety assessment performed in animal models prior to releasing for human used is a requirement before human clinical trials can begin. Routine sampling and testing in our research animal reference testing laboratories is standard.
The research model environment used to investigate the efficacy and safety of therapeutics are designed to protect the models, the veterinary and scientific staff, and the integrity of the study. To minimize contamination and exposure to non-test item antigens, experienced labs with high quality control standards house and handle animals in facilities that have adequate biosecurity measures in place, e.g. IVC cage-changing stations, biological safety cabinets/workstations. Staff wear appropriate personal protective equipment to keep them safe from compounds and animals they are manipulating. In the case of vaccine development, all of this is done to assess precisely how and how well the immune system responds to a pathogen.
In conclusion, research animals have played a critical role in developing a COVID-19 vaccine and one of the reasons the bar is so high in minimizing safety risk. This begins with the biosecurity of the production of animals for vaccine studies and is the result of the hard work of our laboratory animal technicians and staff. Without clear signals in animal studies, vaccines cannot move forward.
Research Models & Services
In the rapidly-evolving world of biomedical science, Charles River’s comprehensive portfolio of research models and services allows us to help our clients research, discover, and develop new therapies.
Ken Henderson received his PhD in molecular virology at the Ohio State University and currently is the Senior Director of Laboratory Services at Charles River Laboratories where he investigates emerging infectious agents and supports global diagnostics for monitoring laboratory models.
We hope you have found this information helpful. Our next installment in the series discusses toxicology testing of vaccines. And check out this important resource about the different ways animals are contributing to the COVID-19 fight.