Safety Assessment
|
Mary Parker
Skin Sensitization Tests: Doing More With Less
A tiered approach to skin sensitization test of chemicals helps minimize the need for in vivo studies. The latest in our ongoing series, Chemical Attractions
In order for regulators to ensure human and environmental safety of new chemicals, especially for agricultural or industrial purposes, tests using animals are required to determine any possible harmful effects. The pain, distress and death experienced by the animals during these tests have been under public debate for a long time.
 “There is an ongoing shift from animal testing to non-animal testing,” said Walter Westerink, section head Flexible Innovation Team of the Department Discovery & Environmental Sciences at Charles River’s Den Bosch, Netherlands site. “More non-animal tests that are accepted by the regulators have become available, and are frequently requested by our clients. Where we can, we will use these alternatives.”
“There is an ongoing shift from animal testing to non-animal testing,” said Walter Westerink, section head Flexible Innovation Team of the Department Discovery & Environmental Sciences at Charles River’s Den Bosch, Netherlands site. “More non-animal tests that are accepted by the regulators have become available, and are frequently requested by our clients. Where we can, we will use these alternatives.”
Pressure to replace animal testing whenever possible comes also from the Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) regulation from the EU. Part of the legislation states that for various endpoints testing must be started with non-animal alternatives.
Tests to predict skin sensitization are one example of required chemical testing that has traditionally been performed on animals only. Luckily, the past few years have seen great advances in the field of non-animal substitutes for these tests.
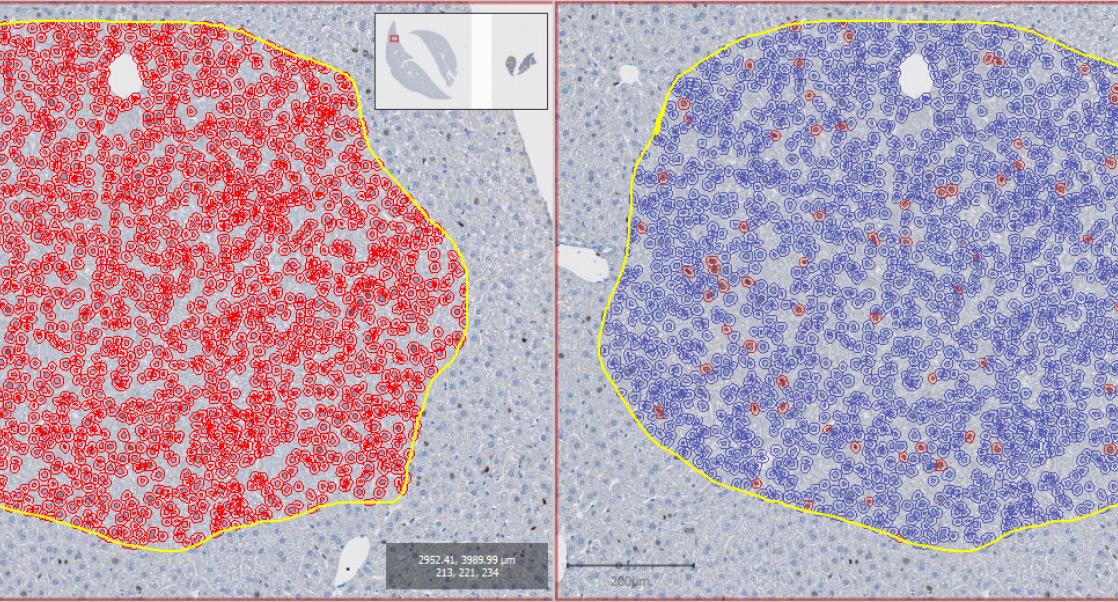
To this end, Charles River developed a tiered approach to skin sensitization tests that help minimize the need for in vivo studies. This approach uses in vitro, in chemico, and in silico tests combined to provide as much data on the chemical as possible without using animals. Although in vivo studies are still sometimes necessary, this approach can limit and sometimes eliminate the need for animal models. Currently this test strategy reduces the number of animals required for skin sensitization testing with 50%.
Commitment to 3Rs
The efforts to find more in vitro alternatives arises from Charles River’s commitment to the 3 R’s of animal research: Replacement, Reduction, and Refinement. The Three R’s were defined to make animal testing as humane as possible by using every means available to reduce the number of animals used. Charles River is helping to validate a new skin sensitization tests that could prove exciting for researchers dedicated to the 3R’s.
One new alternative is the Kinetic Direct Peptide Reactivity Assay (DPRA), developed by researchers at BASF and Givaudan. Traditionally DPRA has been used in the testing strategy to help predicting whether or not a substance could cause an allergic skin reaction. However, this non-animal test strategy could only offer yes or no results. With the kinetic DPRA, researchers may be able to determine the degree of the reaction, thereby this test will help to further decrease the number of animals required.
For other endpoints like eye and skin irritation, or corrosion, there are already non-animal test models. “The advantage of the models is that less animals are needed,” Westerink said. “These models have a very predictive for irritation and corrosion in humans. [Because of these models] 80-90% less animal testing is required.”