Biologics
|
Matthew Pennington, PhD
The Viral Vector Quandary
How can we be sure that the viruses used to deliver the DNA don’t start replicating in cells to make you sick?
The fundamental goal of gene therapy is to introduce new genetic material into a target cell. It has a range of applications, including treating genetic diseases by supplementing abnormal genes, fighting cancer by redirecting a patient’s immune cells against the tumor, and in vaccines, especially for many COVID-19 vaccine candidates. Viruses are widely being used as vectors to deliver the therapeutic DNA because they have evolved mechanisms to be highly efficient at introducing their own DNA into host cells. Scientists have found ways to capitalize on the natural biology of viruses and turn them from infectious agents into powerful therapeutics.
In order to produce a viral vector, the first step is to pick a virus upon which to build it. Many families are being explored (Table 1), but adenoviruses, adeno-associated viruses, retroviruses, and lentiviruses are most commonly used in experimental and clinical settings due to an overall balance of how much DNA they can carry, how long they maintain gene expression for, the types of cells they target, and the types of immune responses targeting the viruses.
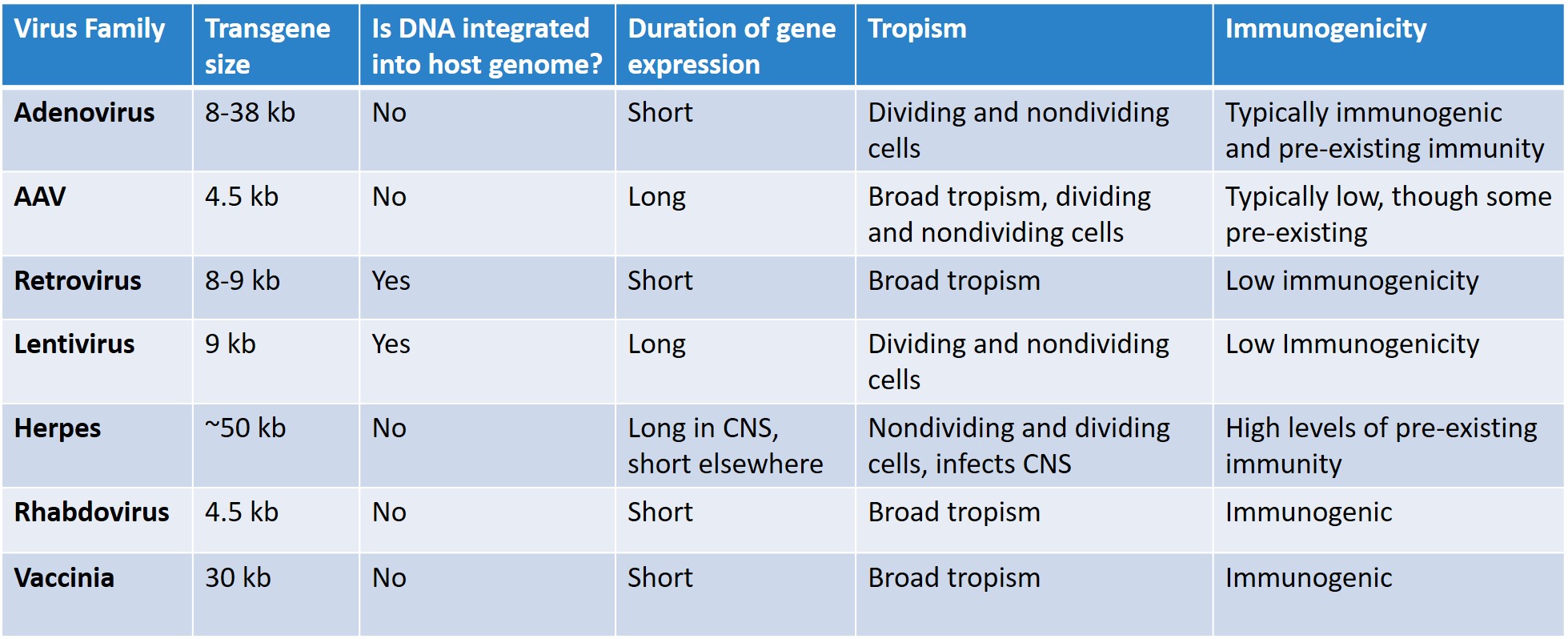 Table 1: Overview of common virus families used as gene therapy vectors. Adenoviruses, adeno-associated viruses (AAV’s), retroviruses, and lentiviruses are most commonly used today. Adapted from Principles of Virology, 4th edition by Jane Flint, et al.
Table 1: Overview of common virus families used as gene therapy vectors. Adenoviruses, adeno-associated viruses (AAV’s), retroviruses, and lentiviruses are most commonly used today. Adapted from Principles of Virology, 4th edition by Jane Flint, et al.
The next step is to modify the genome of the wild type virus. The goal here is two-fold: first the virus must be attenuated, or weakened, so that it cannot cause disease, and second the gene that is to be delivered to the patient, the transgene, must be added. This is done by targeting some region of the viral genome that is essential for replication and deleting it. The transgene is then inserted into this extra space to create the vector genome (Figure 1).
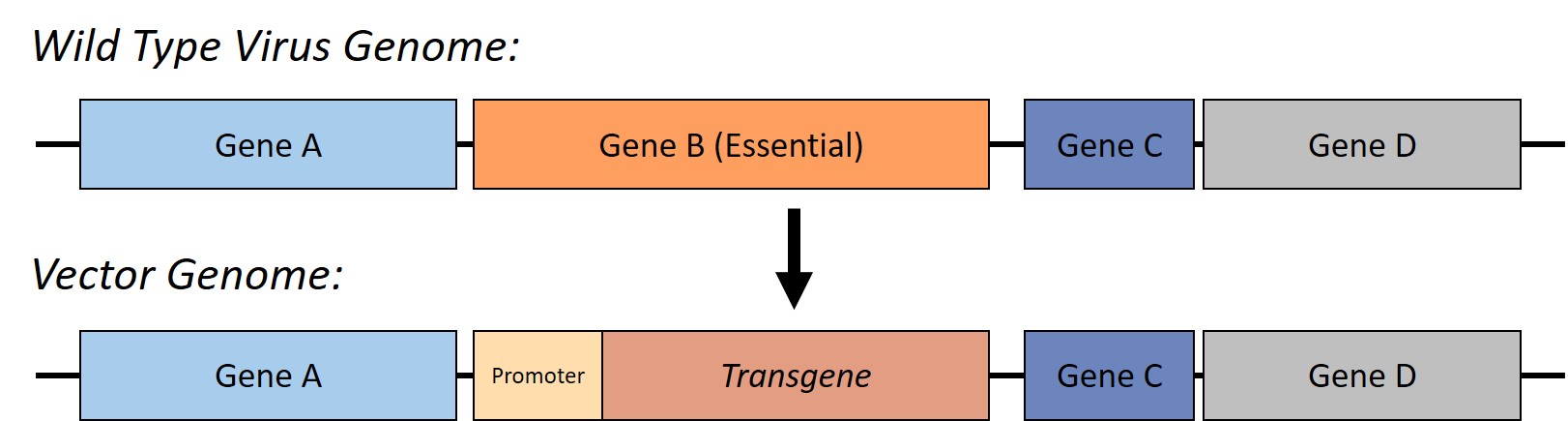
Figure 1: Creation of a viral vector. In order to attenuate the virus, an essential region of the genome is deleted. Without this region, the virus is not capable of replicating and causing disease. This space is used for the transgene, or the therapeutic gene that is being delivered to the patient.
However, this creates a problem. The vector has been designed to not replicate in a patient, but it still needs to be able to replicate in the lab to produce it. This is done by complementing the missing functions of the viral genes that were removed in trans using a packaging cell line. Multiple plasmids are typically used to generate a vector (Figure 2).
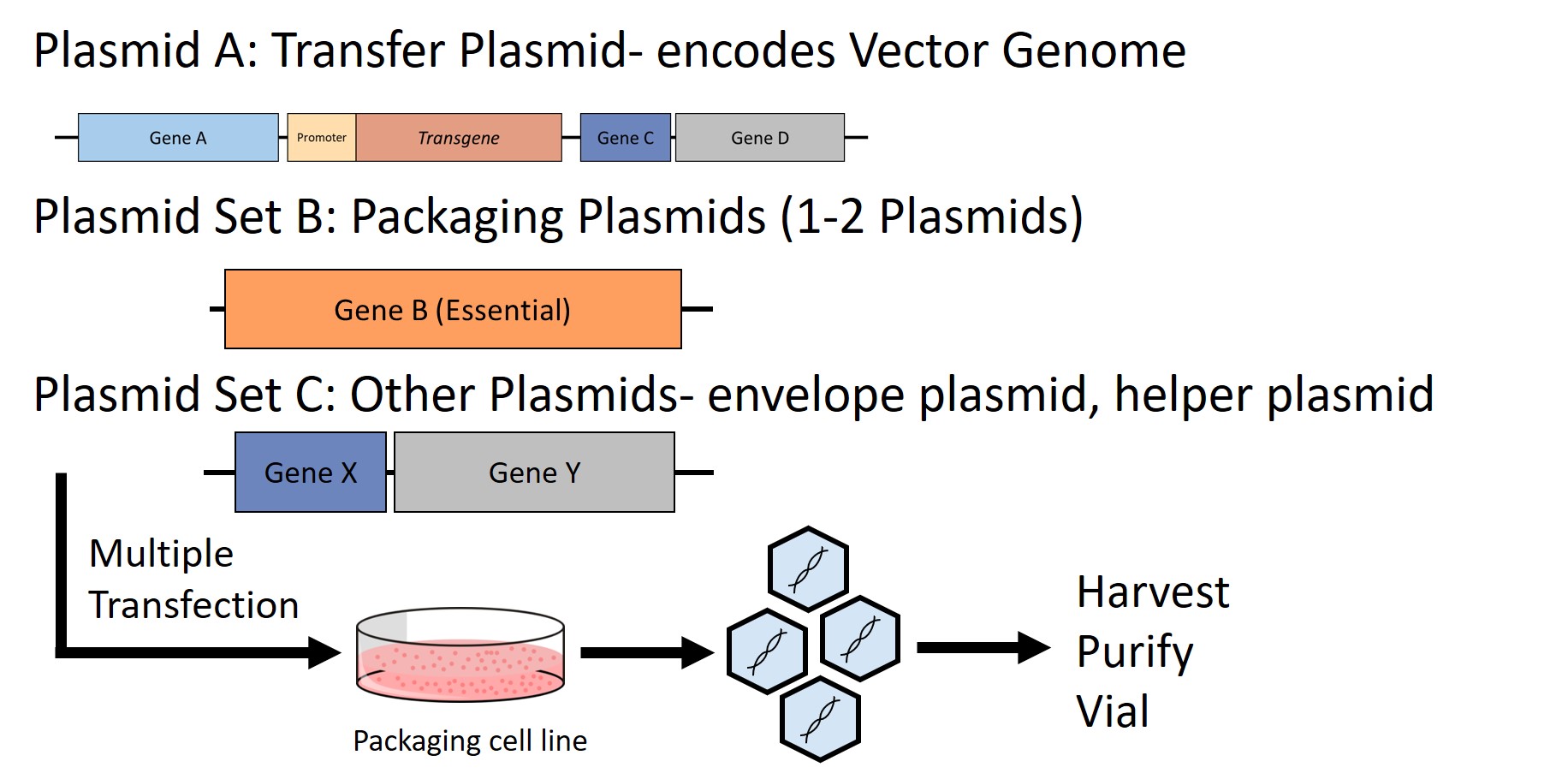
Figure 2: Production of a viral vector. Multiple plasmids, including a transfer plasmid encoding the vector genome, packaging plasmids, and other helper plasmids, are transfected into a packaging cell line. This line is able to produce the vector, but the vector itself will not be capable of replication due to the modifications made to the genome.
First is the transfer plasmid which encodes the vector genome (Figure 1). Second is one or two packaging plasmids that supply the missing functions that were deleted out of the genome. Finally, additional plasmids may be necessary depending on the virus family, such as envelope proteins to define the target of the vector or helper virus functions. When all necessary components are given to a packaging cell line, those cells are able to produce the vector. The vector will encapsidate the vector genome, and exclude the production plasmids, so that the vector itself will not be able to replicate.
One safety concern that arises from this production method is the generation of a replication competent virus (RCV’s). This can occur due to recombination events between the production plasmids or the genome of the packaging cell line with the transfer plasmid (Figure 3). This can result in the re-introduction of essential viral genes into the vector genome, generating a wild type-like, RCV. An RCV may also be inadvertently introduced from other reagents or due to contamination. If delivered to a patient, the RCV can, as the name implies, replicate and cause disease.
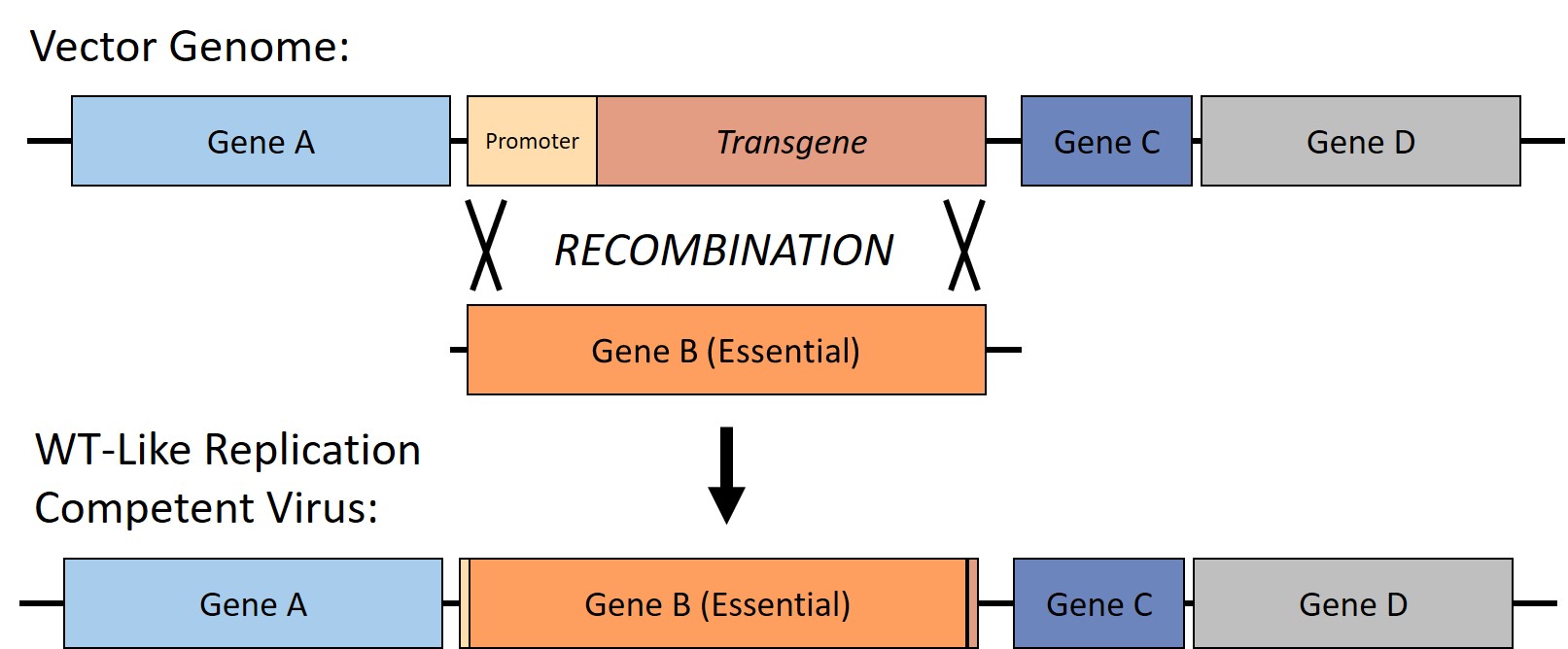
Figure 3: Generation of a replication competent virus (RCV). RCVs may be generated during the vector production process through recombination events between the plasmids and cell lines used to produce the vector and the vector genome, through contamination, or through other sources. This results in a replication competent, wild type-like virus that could cause severe disease if given to a patient.
For some families, the risk of an RCV is very low. Adeno-associated viruses are not associated with disease in humans, and so a replication competent adeno-associated virus (rcAAV) is unlikely to cause any harm. Replication competent lentiviruses (RCL’s), while theoretically possible, have never been reported. However, the risk is higher for other families. Replication competent adenoviruses (RCA’s) may cause severe or even lethal tissue damage and inflammation, especially in immuno-compromised patients. Replication competent retroviruses (RCR’s) have been shown to cause cancers in mice and non-human primate studies.
Additionally, some clinical trials using retroviral gene therapy have reported that some patients developed leukemia. While it was not conclusively established that an RCR was responsible for the leukemia, the method of replication and integration of an RCR would, on paper, be expected to cause cancer. Due to the serious nature of what might occur if an RCV was present, the US Food and Drug Administration has defined exact specifications for the testing of gene therapy products to ensure that they do not contain RCVs.
Detection methods for RCVs are similar for all vectors, though there are key differences for each virus family. A sample of the vector product is applied to a cell line susceptible for infection by that virus family, along with positive, negative, and interference controls. During the amplification phase, the infected cells are maintained and grown for 2-4 weeks, depending on the virus. This allows for potentially very low levels of RCV to expand and grow. Then, in the detection phase, an RCV is identified by a variety of different methods. RCA’s are detected visually based on the death of cells in the culture dish (cytopathic effect). rcAAV’s are detected using the polymerase chain reaction to identify their DNA. RCL’s are detected using an S+/L- assay in which the RCL is able to induce transformation of the PG-4 cell line, which can then be visually observed based on a change in the morphology of the cells. Finally, RCR’s are detected using PCR-based reverse transcriptase assay (PBRT), virus capsid specific enzyme-linked immunosorbent assays (ELISAs), and/or PCR. These assays are time consuming, often taking 4-6 weeks in the lab to perform, but new methods are in development that can speed up these timelines while ensuring they are sufficiently robust and sensitive.
Scientists are continuing to develop new vectors with reduced chances of developing RCV’s. Lentiviral vectors are in their third generation and are designed in such a way that generating an RCL is extremely unlikely. New second generation adenoviral vectors are being developed, with nearly all viral genes removed, rather than just one or two genes. However, even as new vectors are developed, robust and sensitive RCV detection assays will continue to be critical to ensure that these powerful and exciting therapeutics continue to be safe.