Discovery
|
Christoph Eberle, PhD, Louise Brackenbury, PhD
What's Hot in 2024: Oncology
Advances in cancer diagnostics and CAR-T technology changing the game for patients
Liquid biopsy testing is becoming an essential tool for selecting cancer treatments
Precision medicine thinks outside the box of one-size-fits-all categories. It tailors medical interventions that minimize drug side effects while maximizing therapeutic outcomes. It looks at individual risks for developing a disease and takes into account that each patient's response to the same therapy may differ.
Though still limited in clinical practice, it is poised to expand in oncology thanks to liquid biopsy (LB), a non-invasive, low-cost testing method of evaluating and diagnosing tumors using blood and other biofluids. LBs are designed to primarily identify, in our circulatory systems, either tumor cells dissociated from the primary tumor, platelets modified by a tumor, or fragments of free nucleic acids (ctDNA or ctRNA) released from cancerous cells. Biomarkers gained from liquid biopsies offer diagnostic and prognostic information in real-time, often with greater clinical utility than traditional protein tumor markers.
As standardization of sample collection and analytical workflows continues, test results can also direct treatment selection. Since detecting cancer before it manifests as disease is optimal, look for greater utilization of LBs by oncologists in their day-to-day patient care: either as replacement of tissue biopsies or complimentary to those.
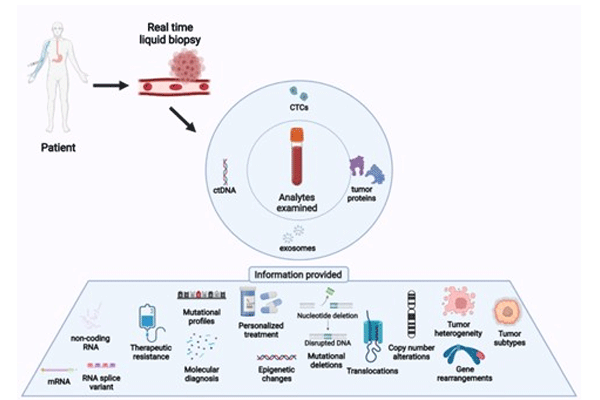
Information available from liquid biopsy testing. Figure reproduced from Lone, S.N., Nisar, S., Masoodi, T. et al. Liquid biopsy: a step closer to transform diagnosis, prognosis and future of cancer treatments. Mol Cancer 21, 79 (2022). https://doi.org/10.1186/s12943-022-01543-7
--Christoph Eberle, PhD, Principal Scientist III, In Vitro Pharmacology/Oncology, Charles River
In vivo CAR therapeutics: the next step?
Chimeric antigen receptor (CAR)-T cells have tremendous therapeutic benefit in patients with hematological cancers. Excitingly, the first cell therapy targeting solid tumors; Carisma’s CAR-Macrophage, was recently granted approval by the FDA to enter clinical trials. The fast-growing field is set to explode as the potential of CAR-X are fully explored, in conjunction with a range of novel tumor antigen targets, multi-specific target binding, ‘tunable’ CAR-T function, and ‘suicide switches.’
However, alarm bells will be ringing following a recent announcement by the FDA who are “investigating serious risk of T-cell malignancy” following administration of several CAR T cell immunotherapies. Many of the products currently in the clinic are generated using viral vector approaches which run this risk. Patients will need to be followed to assess whether cases of treatment-associated malignancy increase. One way to circumvent these concerns would be to harness non-integrative technologies to generate transient CAR T cells with limited genome modifications. Pre-clinical studies using nanocarriers or lipid nanoparticles to deliver an mRNA, circular RNA or DNA encoding the CAR construct provide a promising alternative and offer an approach which is likely to gather momentum over the coming year.
Indeed, looking towards the future, one major advantage is the ability to directly dose patients to generate CAR-T in their own bodies, significantly reducing the lengthy and costly manufacture of autologous cell therapies ex vivo.
-- Louise Brackenbury, PhD, Science Director, Charles River

Oncology & Immuno-Oncology Studies
To reach the clinic in record time, it’s vital to test your oncology therapies in systems that reflect the disease seen in humans. We are here to help with a range of translational oncology studies, including in vitro assays and in vivo models, that mirror human cancers.
