High-Quality Human Whole Blood
Robust, well-characterized healthy donor material is the foundation of any successful cellular therapy. All Charles River blood donors are tested for HBV, HCV, HIV, HTLV, WNV, Trypanosoma cruzi, and Syphilis prior to collection. All donor selection, screening, testing, and qualification processes are monitored by our rigorous quality system, ensuring orders meet your specifications and Charles River’s high-quality standards.
Donated human whole blood contains all liquid and cellular components, cytokines, and growth factors present in the healthy circulating blood system. Whole blood is available as a 10 mL vial, 100mL unit, or 450 mL unit. Freshly collected human whole blood should be used immediately upon receipt. Unless otherwise specified, peripheral human whole blood is collected in a CPD collection bag and specimen samples are collected in EDTA tubes.
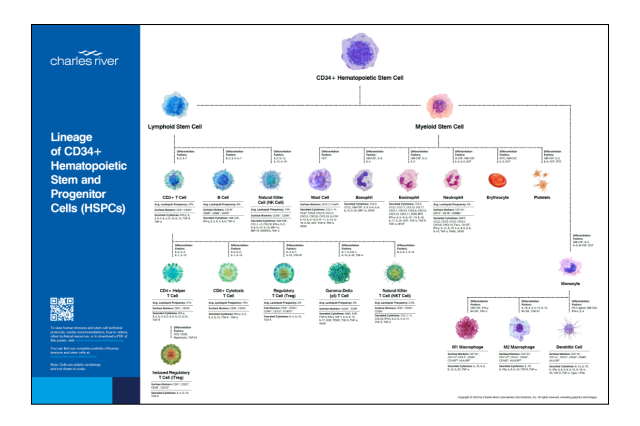
Lineage of CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs)
Receive a complimentary poster to access information on immune cell markers, differentiation factors, cytokines, technical protocols, media recommendations, how-to videos, and more. Download Poster
Charles River can recruit donors or provide samples from our biorepository that meet specific criteria:
- Repeat or unique donors
- Healthy and non-healthy donors
- HLA type, blood type, CMV status
- Demographics (gender, ethnicity, age, etc.)
- Lifestyle characteristics (BMI, smoker/non-smoker, etc.)
- Medical history
- Additional screening and testing
We also offer the following high-quality, well-characterized disease state peripheral blood-derived products:
- Leukopaks, Immune Cells, Whole Blood, Serum, and Plasma – collected in accordance with an IRB-approved protocol. Our disease state blood-derived products encompass a wide range of diseases.