Biologics
|
Caroline Overå
Art of the Science: cryo-EM
How this next-generation tool is elucidating complex protein structures that elude other methods
X-ray crystallography works by computationally reconstructing 3D structures from scattered patterns generated from X-raying crystallized proteins. For years, this method has been the workhorse of structural biology.
But in recent years, the rapidly emerging technique known as cryo-electron microscopy, or cryo-EM for short, is also being employed to solve the structure of proteins. Cryo-EM—which involves vitrifying samples—is becoming an incredibly useful alternative to X-ray crystallography, particularly for atomic-level structures that have resisted crystallization, are flexible, or are dependent on lipid environments.
The life sciences company Atem Bio has been working toward transferring cryo-EM science from academic to industrial applications with high availability. They tackle proteins that are difficult to crystallize, such as membrane proteins, proteins with conformational flexibility, and large complexes. They also offer 3D epitope mapping to determine binding regions of monoclonal antibodies.
Atem Bio, which formed a strategic partnership with Charles River in 2021 and opened a cryo-EM lab in Germany in March, has shared a video and image with Eureka’s Art of the Science that exquisitely captures some of the great science they are doing with the help of cryo-EM.
What are we looking at in this image, i.e. what does the image depict?
The pictures show a 3D model of the PRMT5:MEP50 protein complex together with a natural ligand solved at a 2.6 Å resolution by cryo-EM. This protein complex is an interesting target for cancer studies, and structural information can aid in therapeutical development.
How was the image generated? Describe the tools, techniques used to create the image.
Using cryo-EM, we vitrify purified protein samples, analyzed them in an electron microscope, selected thousands of protein particles in various orientations, and computationally combined them to create a 3D structure. Pretty cool, huh?
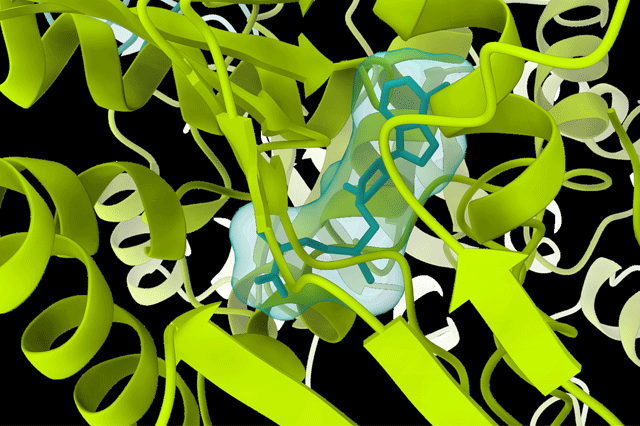
A natural ligand binding to PRMT5:MEP50.
What can scientists learn from these images that help to accelerate drug development?
They can explore complex interactions of drug molecules, investigate properties of disease-causing mutations, or use computer-aided drug design to screen for drug candidates.
How do you expect cryo-EM to develop?
We expect the method to solve higher resolutions on a routine basis and enhance AI (artificial intelligence) development for studying flexible proteins.
Are there particular types of proteins that this technique is ideally suited for?
Some targets that benefit especially from cryo-EM are larger complexes and membrane proteins, such as G protein-coupled receptors or GPCRs that constitute a substantial fraction (30-50%) of the drug targets of interest to the pharmaceutical industry.
Tell us a bit about Atem Bio?
We are passionate about structural biology and aim to make cryo-EM an accessible option for industrial and commercial applications.
Tell us something fun about the Atem Bio team / lab?
We have a group of chicken neighbors outside our new lab that act as our unofficial mascots. ?
Caroline Overå is a Scientific Growth Manager and cryo-EM scientist at ATEM Bio.
This story is part of our Art of the Science series, where we showcase the amazing scientific art (images, movies and cartoons) created by Charles River scientists and partners, in their labs. Do you have a good idea for Art of the Science? Contact [email protected] .