Cell & Gene Therapy
|
Regina Kelder
Cell Donors: Rolling Up Their Sleeves for Research
Recallable donors play a critical role in the chase for exciting new cell therapies

The first time Molly Sinkewicz thought about cell donation was when she was studying biology at Worcester Polytechnical Institute. The idea that you could give your own cells to heal someone else intrigued her. But it was something much bigger and deeply personal that convinced Molly to roll up her sleeve and donate three hours of her time – and her white blood cells.
When she was in her 20s, she found out she had thyroid cancer. The golf-ball sized tumor was treated successfully at Massachusetts General Hospital, and Molly realized how lucky she was to be surrounded by so many excellent hospitals and research centers. “I’ve benefited a lot from good medical care, so I like to give back wherever I can,” says Molly, who is now 30 and owner of a small chain of gyms.
That desire to give back led her to the HemaCare Donor Center in Lowell, MA, about a 30-minute drive from her home in Harvard, MA, where she began donating her blood for basic research and the development of new therapies. She has since become what is known as a “recallable donor” – the term for individuals who have demonstrated longstanding commitment and are reliable.
The need for recallable cell donors is escalating
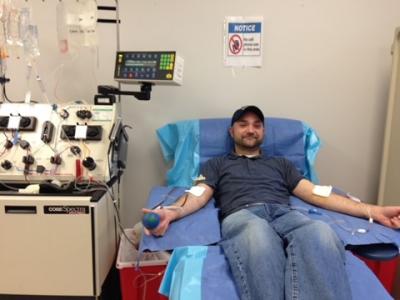
The cell donation process Molly and hundreds of other individuals undergo every day is known_0.jpg) as apheresis. Unlike a typical bloodmobile appointment, which takes about 45 minutes of a donor’s time, apheresis is a more complex and longer process that separates out specific components of the blood of unique importance for scientific exploration.
as apheresis. Unlike a typical bloodmobile appointment, which takes about 45 minutes of a donor’s time, apheresis is a more complex and longer process that separates out specific components of the blood of unique importance for scientific exploration.
The cells provide important raw material needed for the development of a cell therapy, which involves the transfer of intact, live cells into a patient to help lessen or cure a disease. While there are only a handful of cell therapies approved for use in the US and Europe, mostly for rare cancers, there are many more cell therapy products in the pipeline. The site clinicaltrials.gov, which tracks clinical trials worldwide, estimates there are more than 2,500 cell and gene therapy trials enrolling or in progress, mostly for rare cancers but increasingly for autoimmune diseases and more common conditions. As proof clinicians are becoming more confident in the power and availability of cell therapies, there is now evidence that some approved cell therapies are becoming earlier treatment options.
The impetus then is on blood donation centers like HemaCare Donor Center—which has branches in Massachusetts, Tennessee, California, and Washington State—to provide stable, sufficient, and comprehensive supplies of donated cells, fluids, and tissues to research and pharmaceutical concerns. Having large donor pools means clients can access cells with a high degree of specificity. They can filter by blood type, body mass index, age, gender, or any number of other factors.
Eileen Karr, Senior Manager of Apheresis Operations at HemaCare Donor Center’s Lowell site, said demand for starting materials has grown exponentially over the past 5 to 7 years, driven by the incredible success of chimeric antigen receptor or CAR-T cell therapies, which rely on engineered white blood cells—T cells—that seek out and destroy malignant cells in the body. “Scientists discovered that these [cell therapies] were effective and in some cases could cure illnesses, such as lymphomas, once considered incurable,” says Karr. “As an apheresis professional, this is extremely exciting because suddenly we are part of this world that is going to make a huge difference.”
There is no way to overstate how much donors like Molly are key to the continued success of cell therapies, including the development of off-the-shelf cell therapy products that rely on donor cells or umbilical cord blood rather than the patient’s cells. Scaling up cell therapies means scaling up donor access.
Darrin Mellinger, a Bids and Contract Manager in Memphis, has been donating for two decades. It began in college, and he admits at the time the main draw was the money. Because these cells are highly sought by researchers, most centers will pay donors upwards of several hundred dollars each time they give. Darrin’s other motivation was his mother – she worked at the donor center and encouraged him to become involved.
Once he finished his studies and landed a full-time job, it became less and less about the money and more about the connections he had formed with the donor center. “I ask them ‘How come you guys call me so much’ and they said ‘because you answer, because you always come in and you are never late. You are a dependable donor,’ “ says Darrin. “So, I guess it’s a personal integrity thing for me. On top of that, it’s the thought that I could be helping one person somewhere dealing with some type of medical issue. It makes me feel good to be able to help.”
What caused cell therapies to take off?
Cell therapies can be autologous (also known as self-to-self, using cells from the patient receiving the treatment) or allogeneic (using cells from a donor for the treatment). While this field of treatment has recently begun to expand, some forms of cell therapy like the cancer-treating hematopoietic stem cell transplantation (HSCT) have been in practice for decades.
But the cell therapy field really took off in 2006, when researchers figured out how to allow some cells to be “reprogrammed” genetically leading to a new type of stem cell known as induced pluripotent stem cells. This discovery led to the use of stem cell therapies for Type 1 diabetes, Parkinson’s disease, and spinal cord injuries. Around the same time, there was also a growing focus on using immune cells to treat cancer, leading to the development of CAR T cell therapies, and more recently engineered T cell therapies, natural killer cell therapies and tumor infiltrating lymphocyte cell therapy, the latter three of which are still in the experimental phases.
Casey Hilton, the Site Operations Manager at Charles River’s HemaCare Donor Center in Memphis, said without donors their business “would not exist, plain and simple.” Hilton has been working in the cell solutions business for about four years, but he also has spent a lot of time with patients. He spent a decade in the Army, and part of that time was as a Medical Laboratory Technician. Casey worked at Landstuhl Regional Medical Center in Germany that served as a stopover for wounded veterans from all NATO nations. Most of the time he worked in a high-level CDC surveillance lab screening dangerous pathogens but every few months he would do ward rounds, drawing blood from patients — many of them amputees and paraplegics—and it was this interaction that sealed in his mind a desire to continue this work after his tour of duty ended. One of the reasons he is drawn to the cell solutions business is because he knew that the “products were going into that pipeline of research.”
Donor apheresis and a donor app
The donor apheresis process works something like this. A healthy person donates blood using the apheresis machine, which is programmed to collect only the desired blood component—either red blood cells, white blood cells, platelets, or plasma. For instance, if a donor’s white blood cells are retrieved during apheresis, the non-targeted plasma or platelets are returned to the volunteer donor.
If collecting white blood cells, blood is drawn from one arm and sent through sterile tubing into a centrifuge located in a cell-separator machine. The machine spins the blood to separate the white blood cells from the other components. The white blood cells are collected, and the remaining components are returned to the donor via the other arm. Only a small portion of blood is in the machine at any time (less than a cup). The entire procedure may take approximately four hours and is not uncomfortable, says Hilton.
Cultivating recallable cell donors has typically been a manual, labor-intensive process that

takes anywhere from two to three months between the time a volunteer cell donor fills out the first questionnaire to the moment when they are scheduled to give blood. Recently, Charles River Laboratories’ HemaCare Donor Center in Memphis launched a new digital recruitment experience that allowed the site to recruit cell solutions donors completely digitally, without the need for manual intervention by recruiters. After an initial pilot period, the success of the donor app was quickly proven. Consequently, this digital donor experience is now available at all of the HemaCare Donor Center locations.
April Hall, who lives in Southern California’s San Fernando Valley, first heard about the apheresis process 14 years ago when she was working as a cashier at Trader Joe’s. A customer kept coming through her line with company gift cards that she later found out he received for donating his platelets. April decided to donate, too, and when she learned the incentives became better if you donated consistently, April became a recallable donor.
About a year after she signed up, April’s brother passed away. She remembers learning he had low platelet count, and suddenly the value of what she did every few weeks became clearer. Today, April is a manager at Trader Joe’s, and now donates white blood cells consistently every five weeks at 7 am. “The people there are really cool, thankful and appreciative,” she said.

