Biologics
|
Ana Catarina Carvalho
Dangerous Diseases in Safe Spaces
How a Ballina laboratory upgraded to take on risky research safely
Biological Agents and their classification:
Biological agents are defined by the World Health Organization (WHO) as a microorganism, virus, biological toxin, particle or otherwise infectious material, either naturally occurring or genetically modified, which may have the potential to cause infection, allergy, toxicity or otherwise create a hazard to humans, animals, or plants.
While many of these agents are harmless or even essential to human or animal health, like commensal bacteria in the gut microbiota, some have the potential to cause harm or disease.
As a result, Biological Agents can be classified into risk groups / classifications using different systems. The World Health Organization recommends each country employs a classification system, but the risk classification and the definitions of risk groups depend on the local context (local policy or legislation, environmental or climatic factors, whether an agent is present or eradicated from the community or region, among other factors). The US Center for Disease Control and Prevention (CDC) and European Centre for Disease Prevention and Control (ECDC) set risk groups for biological agents ranging from 1-4, least to most hazardous. And similarly, Biological Safety Levels (BSL) or containment levels for laboratories, vivaria, isolation hospital facilities, or industrial facilities, ranging from BSL-1 to BSL-4, least to most secure:
Risk Groups in the EU and US:
Risk group 1 consist of biological agents unlikely to cause disease to humans and/or animals – most microorganisms fall into this category.
Risk group 2 biological agents can cause harm/disease to humans and/or animals, but they are unlikely to spread to the community and there is usually effective prophylaxis or treatment available – examples include the Streptococcus spp. bacteria responsible for strep throat or the zoster virus responsible for chicken pox and shingles.
Risk group 3 biological agents have the potential to cause severe human and/or animal disease and present a serious hazard which may present a risk of spreading to the community, examples include bacteria such as Mycobacterium tuberculosis responsible for TB and viruses such as SARS-CoV-2, Hepatitis B, and HIV-1. While these pose serious hazard to health, effective prophylactic vaccines or therapies/treatments are available.
Risk group 4 biological agents like Ebola, Marburg or the virus behind Crimean-Congo haemorrhagic fever (CCHF) are in the highest level of risk categorization – these viral agents can cause life threatening disease, and no therapy, prophylactic vaccine or cure exists.
Biological safety levels in the EU and US:
Informed by a risk-based approach or a risk classification, the minimum biological safety level or so-called containment level where a biological agent can be handled in a laboratory or industry is determined. Each Biological Safety level builds on the previous level and where, elimination and substitution of the hazard, the top two layers of the safety hierarchy of controls, are impossible in the context of high containment levels, there is an increased importance placed on implementing additional engineering controls and safe systems of work.
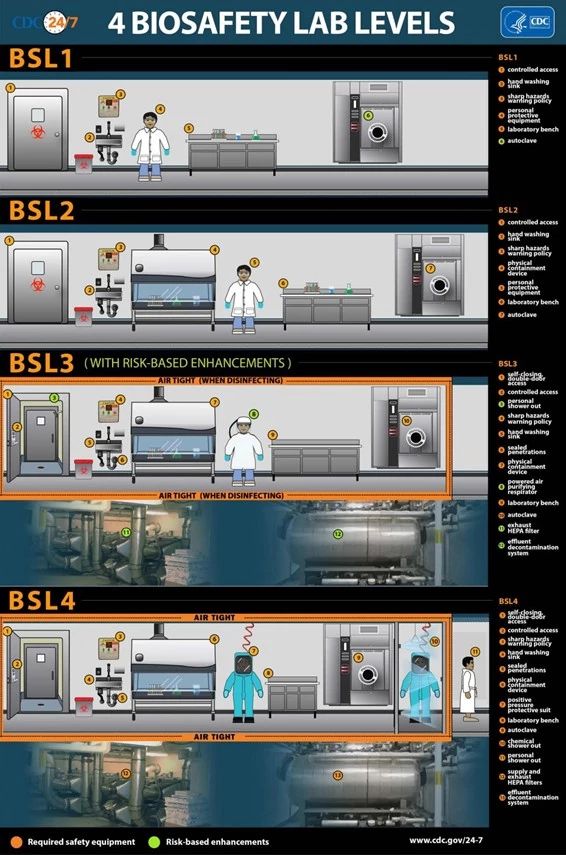
Image source: US, CDC
Upgrading facility from BSL-2 to BSL-3:
At our Ballina site in Ireland, we underwent the lengthy process of upgrading a pre-existing BSL-2 facility with enhanced controls for influenza testing in vivo to a multipurpose BSL-3 facility.
The upgrade process entailed a comprehensive review of systems, processes, facility layout and controls which took several months and entailed extensive collaborative work between our specialised technical staff and our EHS&S, Facilities, Calibrations, Validations, Quality Assurance, Procurement teams and other supporting services. Where required, we engaged with specialised contractors or expert consultants and engineers to assess and implement adequate safety and engineering controls.
One side of the upgrade was the demolition and construction work, the extensive overhaul of the air handling units and the implementation of passive monitoring systems designed to alert personnel of any malfunction.
A second stage involved the commissioning and testing of the suitability of the facility and the controls installed, as well as very extensive efforts in the commissioning and validation of a large amount of analytical equipment.
In parallel to this process, a review of regulatory framework was performed.
Across different territories, regulations and governing bodies may differ. In Ireland we had to engage and seek approval from the relevant regulatory bodies: the Health and Safety Authority with oversight of Biological Agent, Health and Safety in the workplace regulations, and with the Environmental Protection Agency with oversight of Genetically Modified (Micro-)organisms regulations.
Additionally, internally we underwent a review of systems of work and their compliance not only with the relevant legislations but also with current good manufacturing practices (cGMP) and guidelines, as we thrived to open the first commercial and cGMP-compliant BSL-3 laboratory in Ireland.
The BSL-3 laboratory in Ireland:
The facility is approximately 200sqm and the core BSL-3 area has been designed with viral agents testing in mind: it includes virus culture rooms fitted with engineering controls specific to bioaerosol-related hazards, molecular biology areas (for sample handling, a post-extraction/ pre-amplification area and an amplification area) and an area dedicated to ELISA-based methods – with these capacities we are currently supporting testing of HIV-1-based gene and cell therapy products.
For in vivo and ex vivo testing, multipurpose areas for animal housing and in vivo and ex vivo procedures have been created: the facility was designed so it can be converted to house different rodent species and animal models in individually ventilated and HEPA-exhausted cages. We currently support drug efficacy studies and infection models including SARS-CoV-2 infection in the Syrian hamster model.
There are also additional supporting BSL-3 and BSL-2 areas: in full containment level 3 we have an ensuite decontamination area required because waste contaminated with risk group 3 microorganisms must be inactivated ensuite, i.e., within full containment level 3. A dedicated temperature-controlled storage area was also essential to ensure biological agents are stored within the restricted area.
In associated BSL-2 areas, there are mammalian cell culture and Master Mix laboratories to ensure the non-hazardous steps of work performed is executed with appropriate level of controls minimizing undue foot traffic at high containment level and unnecessary risks to personnel.
The current focus on viral agents arose from the market needs in the wake of the Covid-19 pandemic, however, the modular nature of the setup facilitates the possibility to scale up and expand to include a segregated area where other microorganism such as bacteria, mycobacteria and fungi that are incompatible with the current work and controls can be safely stored and handled.