Discovery
|
Blandine Mille-Baker; Aditya Ambade; Elisa Pasqua
The GLP-1RA Pipeline
Beyond obesity, drug developers are exploring new uses for these therapies from kidney disease to neurological conditions
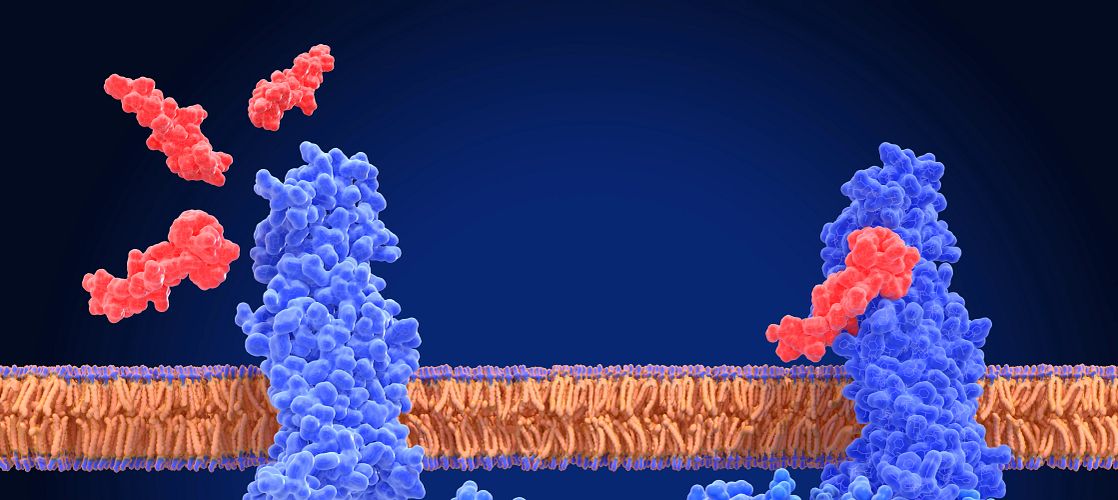
Following the phenomenal success of glucagon-like peptide 1 (GLP-1) receptor agonist medications (GLP-1RAs), such as semaglutide (active ingredient of Ozempic and Wegovy), multiple companies have focused on developing additional drugs to either complement or enhance current treatments for obesity. A multitude of peptides, small molecules, monoclonal antibodies and ADCs with different mechanisms of action to treat obesity and other indications is progressing through CRL’s pre-clinical drug development services.
In this scenario, combination therapies targeting multiple pathways are emerging as particularly promising to tackle obesity, due to the complex nature of this chronic metabolic disease.
Tirzepatide (active principle of Mounjaro and Zepbound), a dual co-agonist for gastric inhibitory polypeptide (GIP) receptor and glucagon-like peptide-1 (GLP-1) receptor, in clinical trials performed well for body weight loss (greater than 20% over 72 weeks) and cardiometabolic parameters, and also demonstrated additional benefits, such as reduction in heart weight and fat surrounding the heart.
Interestingly, despite opposite in mechanism to drugs like tirzepatide, antibody-peptide conjugates acting as dual GLP-1 receptor agonist / GIP receptor antagonists, such as MariTide, are delivering promising results in clinical trials (average 20% weight loss over 52 weeks) , and appear to provide improved weight-loss maintenance upon treatment discontinuation.
Retatrutide, a GGG-tri agonist (triple GLP-1/GIP/glucagon receptor agonist), displayed even better results in clinical trials, with an average 25% body weight loss in two-thirds of the time, potentially surpassing the efficacy of gastric bypass. Additionally, a reduction in blood-sugar levels in diabetic participants was observed.
CagriSema, a combination of semaglutide and cagrilintide (long-acting analogue of amylin) outperformed both drugs as single therapies, with a 23% body weight loss in a 68-week study in a clinical trial in adults with obesity or overweight with one or more comorbidities and without type 2 diabetes.
Bimagrumab is a monoclonal antibody antagonist of activin Type II receptor that stimulates skeletal muscle growth, whilst safely decreasing fat mass and cardiometabolic parameters without affecting muscle mass. It is currently under clinical evaluation in combination therapies with other obesity medications, such as semaglutide and tirzepatide.
Conversely to GLP-1RAs focusing on energy input, alternative approaches are pursuing the energy expenditure (i.e. mitochondrial uncoupling) as a more sustainable strategy to preserve lean muscle.
Alongside combination therapies, pharmaceutical companies are also pushing forward alternative forms of GLP-1RAs aiming to addressing some of their initial challenges, such as cost and weekly injections. Orforglipron is an example of an oral non-peptide GLP-1 receptor agonist currently in clinical trials that, if successful, could potentially offer the benefit of a daily pill.
Finally, emerging therapies based on alternative mechanisms of action, such as synthetic analogues of amylin hormone (i.e. petrelintide) or cannabinoid receptor antagonists, are also showing some promise in providing a higher-quality weight loss coupled to muscle and bone density preservation, which could be beneficial to treat frail or older patient populations. Some of these therapies, such as a dual amylin/calcitonin receptor agonist, may also be associated with less GI side effects.
Future Directions for GLP-1RAs
Beyond their well-established metabolic benefits including glycemia and body weight control, GLP-1RAs may also be beneficial to liver health and represent a promising therapeutic option for metabolic dysfunction-associated fatty liver disease (MAFLD) and steatohepatitis (MASH). Through their direct activity on circulating insulin and glucose, they decrease liver fat accumulation that can lead to liver fibrosis and progression to cirrhosis and hepatocellular carcinoma.
GLP-1RAs have also demonstrated cardiovascular (CV) protective effects. They can significantly reduce the risk of major adverse cardiovascular events (MACE) through potential mechanisms including blood pressure and lipid profile improvement, reduction in inflammation, amelioration of endothelial function, cardioprotective effects and delay in kidney disease progression. In January 2025, the FDA approved the GLP-1RA, semaglutide to reduce kidney and CV risk in patients with type 2 diabetes (T2D) and chronic kidney disease (CKD). GLP-1-RAs are shown to reduce albuminuria and protect estimated glomerular filtration rate (eGFR), attributed to their anti-inflammatory, antioxidant, and vasodilatory properties.
GLP1 receptor agonists are also known for slowing down neurodegeneration in pathologies such as dementia, Parkinson’s (PD) and Alzheimer’s (AD) diseases. Both in vitro and in vivo studies have demonstrated GLP1-RAs modulation of pathogenic mechanisms such as autophagy, neuroinflammation, mitochondrial dysfunction and abnormal phosphorylation of pathogenic proteins. So far, clinical trials testing GLP-1RA for diseases such as PD, AD, cognitive and psychiatric disorders have generated mixed outcomes for cognitive and motor functions and additional studies will be necessary to elucidate disease-modifying effects.
In addition, GLP-1RAs use was recently associated with a reduced risk of coagulation disorders, infectious illnesses and several respiratory conditions and may offer a potential new therapeutic option in addiction medicine.
Besides their enhancing cardiometabolic activity, growing evidence suggest the potential of GLP-1RA class of medications to be re-purposed for other therapeutic indications, including neuropsychiatric conditions. Additional efforts are however needed to elucidate the mechanistic basis of such benefits and test them in a clinical context. High costs, route of administration and tolerability still represent major limitations to wider prescription of these drugs which opens an exciting perspective on future development.
Blandine Mille-Baker is a Research Leader for Discovery Sciences. Aditya Ambade is an Associate Director for Pharmacology. Elisa Pasqua is a Senior Group Leader for Chemistry.