Safety Assessment
|
Regina Kelder
The Drug-Induced Liver Injury Puzzle
Do we have all the in vitro pieces needed to solve drug-induced liver toxicity, and how will they fit in with conventional animal tests
Buoyed by a flurry of regulatory guidance, pharmaceutical companies are eyeing—and, in some cases, adopting—human-relevant models to develop their drugs. From organ chips and organoids to AI, computational models, and cell-based protein arrays, the Discovery space is awash with examples of non-animal methods that are reducing and, in some cases, replacing animal models in the early vetting of new compounds. Further downstream, the decades-old rabbit pyrogen test, once used exclusively to detect fever-inducing, lethal pyrogens produced by bacteria, has been nearly replaced in Europe by an array of alternative methods. Meanwhile, virtual control groups, now under active review in the nonclinical space, could soon lead to major reductions in the number of animals used as controls in toxicology studies (Charles River is one of the laboratories pioneering this effort.) Meanwhile, cell-based protein arrays are a well-tested in vitro method for identifying off-target effects of therapeutic molecules. The test complements, and in some cases serves as an alternative to, tissue cross-reactivity studies conducted in animals.
Still, there are many tests for which there is no alternative to animals in the offing. Many scientists, and drug developers, too, believe that to correctly assess a drug’s toxic effects, you still need to test for them in whole living organisms. Reproductive toxicology studies are one example where animal studies remain the preferred tool, though the Dutch company Toxys recently developed an in vitro assay called ReproTracker that identifies developmental toxicity hazards in new chemicals, cosmetics, and drugs. It complements traditional toxicology methods.
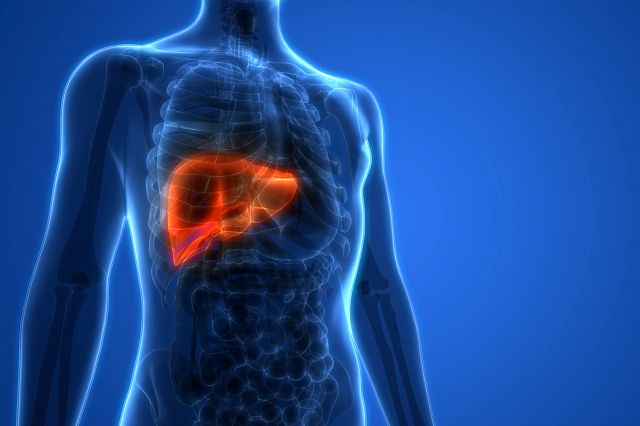
Another area where animals remain the only validated model is the assessment of liver toxicity in safety assessment studies. Currently, there are no methods accepted by regulators to measure drug-induced liver toxicity in a safety assessment setting using simple tissue culture methods, or more complex 3D models, like liver on a chip, organoids, or spheroids.
Part of the reason is the liver’s multiple roles. The liver is the ultimate multitasker. It filters toxins, processes glucose, regulates blood clotting and amino acid metabolism, and produces albumin, a protein that helps prevent fluids in the bloodstream from leaking into surrounding tissues. In fact, the liver performs over 500 vital functions.
 So, when it comes to capturing the biological complexity of the liver and assessing any acute or long-term damage from a compound, a simple in vitro or in silico test is really no match for a research mouse, let alone a human. While some New Approach Methods (NAMs) are being developed to assess diverse mechanisms of toxicity, animals are still the preferred route for predicting the risk of drug-induced liver injury (DILI) in humans.
So, when it comes to capturing the biological complexity of the liver and assessing any acute or long-term damage from a compound, a simple in vitro or in silico test is really no match for a research mouse, let alone a human. While some New Approach Methods (NAMs) are being developed to assess diverse mechanisms of toxicity, animals are still the preferred route for predicting the risk of drug-induced liver injury (DILI) in humans.
“The liver is the first point of contact for the blood flow that comes out of the intestine via the hepatic vein. It’s in the business of nullifying toxins, or whatever injurious agents come in,” said George Parker, Senior Scientific Director of Global Pathology for Charles River. “The heart, lung, and kidneys are certainly important in maintaining homeostasis, but toxicity amelioration is a major function of the liver. If a drug damages that capability, it can kill the animal in a preclinical drug study and cause long-term illness and potentially death in a person.”
Liver toxicity is commonly encountered in nonclinical safety assessment studies. Historical data indicate that liver toxicity is the leading cause of failure in drug development. While biologics, which constitute about 50% of the drug development pipeline, would not be expected to damage the liver, at least as far as direct toxicity is concerned, small-molecule drugs that constitute the other half of the pipeline are a different story. “Most of the biopharmaceutical products are basically proteins,” says Parker. “When the liver sees them, it’s just another protein because it receives protein out of the gut all the time. But when you get into small molecules, chemical-based drugs in particular, there are certain classes of them that are known to be hepatotoxic.”
Since the tragic chapter of thalidomide, a sedative developed in the 1950s for morning sickness that caused tragic birth defects in the babies of pregnant women who took the drug, regulators have followed rigorous testing procedures that stipulate drugs should be tested for potential side effects on the fetus, using mice, rats, or rabbits. Today, drugs and chemicals cannot move forward without a slew of animal safety tests and genotoxicity evaluations, which are designed to detect drugs and other substances that have the potential to alter or damage our genetic material and potentially lead to cancer.
Yet, with animal models being inconsistent predictors of human liver toxicity in the clinic, the drive to improve and validate alternative models that can provide data to help determine a drug compound’s translatability in the clinic is in high gear. Among the hottest areas of development are liver chips, but extensive research is also underway on two other 3D models—organoids and spheroids—as well as computational models (Charles River is doing experimental work in all of these areas).
Webinar: Strategies to Reduce Hepatotoxicity Risk
This webinar covers the complexities of DILI, key risk factors, and how to build a mechanistic de‑risking plan. It highlights the value of early hepatotoxicity assessment for better risk management and reviews in silico, in vitro, and in vivo tools and their predictive applications.
Watch Now
Liver-on-a-Chip Technology
Organ-on-a-chip technology, also referred to as tissue chips, is .jpg?crop=true&keep=c&q=80&color=ffffffff&u=8anjqk&w=400&h=200) about the size of an AA battery. They contain engineered or natural miniature tissues derived from various organs, grown within miniaturized fluid channels molded from glass, silicon, or polymer, or within 3D-engineered scaffolds embedded in well plates. These microphysiological systems (MPS) are generally combined with hair-fine microchannels that guide and manipulate minute volumes of solutions to create environments that recapitulate one or more tissue-specific functions by recreating human organs, tissues, and biological processes in vitro. Liver chips, like those designed by organ chip pioneer Emulate, correctly identified 9 out of 10 toxic drugs in the largest reported evaluation of organ-on-a-chip performance thus far. A number of large pharmaceutical companies, including AstraZeneca, Sanofi, and Merck, are experimenting with liver chips, as is Charles River, which, in partnership with CN Bio, is working on a liver-on-a-chip system to evaluate genotoxic potential.
about the size of an AA battery. They contain engineered or natural miniature tissues derived from various organs, grown within miniaturized fluid channels molded from glass, silicon, or polymer, or within 3D-engineered scaffolds embedded in well plates. These microphysiological systems (MPS) are generally combined with hair-fine microchannels that guide and manipulate minute volumes of solutions to create environments that recapitulate one or more tissue-specific functions by recreating human organs, tissues, and biological processes in vitro. Liver chips, like those designed by organ chip pioneer Emulate, correctly identified 9 out of 10 toxic drugs in the largest reported evaluation of organ-on-a-chip performance thus far. A number of large pharmaceutical companies, including AstraZeneca, Sanofi, and Merck, are experimenting with liver chips, as is Charles River, which, in partnership with CN Bio, is working on a liver-on-a-chip system to evaluate genotoxic potential.
Despite all this positive activity, significant challenges remain. One multi-group study led by Texas A&M University and published last year found that the efficacy of liver-chip system technology tends to wane over time and that the system is not capable of evaluating things at the scale of a 2D culture. It’s also resource-intensive, complicated to maintain, and ultimately expensive to use.
In the matter of genotoxicity studies, Annie Hamel, MSc, Scientific Director of Genetic Toxicology at Charles River’s Safety Assessment site in Senneville, QC (just outside Montreal), said they initially worked with a closed liver-on-a-chip system because it offered more versatility than other models. But closed systems are also difficult to work with, so two years ago, Hamel’s team decided to apply what they learned from the closed chip to a more user-friendly open plate well format developed by organ chip company CN Bio. Two years ago, they published findings showing that a liver chip model featuring a closed-loop design was a promising tool for identifying genotoxicity.
Hamel said the biggest hurdles in this arduous journey will be confirming the liver model’s sensitivity and efficacy across multiple compounds. She says they also need to determine if sequencing DNA from the tissue used in the liver model can detect gene mutations. “Right now, the mutation evaluation is performed using bacteria,” says Hamel. “We would like to replace that assay with the sequencing assay, which would be a good way of showing that we can get better data and be more in line with the responses seen in humans.”
Organoids and spheroids for measuring liver toxicity
Two other models with biologically relevant components that can be integrated in MPS are organoids and spheroids. Organoids are 3D cell cultures derived from  stem cells that self-organize and give rise to organ-specific cell types. Organoids aim to mimic the structure, function, and cellular complexity of human organs, making them, in some ways, a more translatable tool to humans than animals. Spheroids are also 3D single-type cell models that can mimic some organs, tumors, and other biological systems. They form clusters of cells, such as hepatocytes, by adhering to one another, recapitulating the 3D dimensionality of the tissue. Unlike organoids, spheroids can’t self-assemble or regenerate and, thus, aren’t as advanced as organoids.
stem cells that self-organize and give rise to organ-specific cell types. Organoids aim to mimic the structure, function, and cellular complexity of human organs, making them, in some ways, a more translatable tool to humans than animals. Spheroids are also 3D single-type cell models that can mimic some organs, tumors, and other biological systems. They form clusters of cells, such as hepatocytes, by adhering to one another, recapitulating the 3D dimensionality of the tissue. Unlike organoids, spheroids can’t self-assemble or regenerate and, thus, aren’t as advanced as organoids.
Like organ-on-a-chip systems, academic and commercial laboratories are embracing liver organoids in a big way. For instance, Cincinnati Children’s Hospital, in partnership with Roche, has developed a next-generation human liver organoid microarray platform that can help predict which drugs may cause harmful immune reactions in some people. Some drugs that pass early safety testing can still cause idiosyncratic drug-induced liver injury (iDILI)—a rare reaction, sometimes immune-related, that can lead to severe illness or even drug withdrawal. Standard lab tests and animal models cannot reproduce these complex, patient-specific immune mechanisms, but the Cincinnati/Roche system described last year in the journal Advanced Science captured hallmark features of adaptive immune-mediated hepatotoxicity.
Charles River, which has active programs for both intestinal and tonsil organoids, as well as PDX organoids for cancer, recently started generating liver organoids to derisk drugs for liver toxicity. They used induced pluripotent stem cells rather than primary cells to generate organoids, which, along with epithelial cells, also contain a tiny fraction of mesenchymal and immune cells. Ludovico Buti, PhD, Senior Research Leader at the Charles River Discovery site in Leiden, The Netherlands, said they chose to generate iPSC-derived multi-lineage liver organoids because they wanted the organoids to contain three different cell types from the same genetic background and source. “Obviously, the liver is a very complex organ,” says Buti. “While we are not recapitulating all of the physiology of the liver, it is still remarkable what we have been able to accomplish.”
Buti says they are still testing and learning from the liver organoid model, but he anticipates they will be ready to start testing it on client compounds and comparing the results with those from spheroids or primary-derived organoids soon.
So how do in-vitro models stack up against in vivo models? Buti says it is worth noting that retrospectively, the US Food and Drug Administration tested six different liver in vitro models and compared the results with three animal model studies. The general outcome of the analysis is that the in vitro models are more accurate than the in vivo models, but more often than not, the data are not fully extracted from those complex in vitro models. Parker added that the study was limited to known liver toxicants. “We need to know how well the in vitro methods would work in the detection of the de novo toxicants that are currently detected by animal models.”
Nonetheless, DILI is a crowded space at the moment, says Buti. “All the major MPS companies are heavily invested in the liver organoid.”
 Spheroids, another promising 3D model, maintain long-term cellular viability, structure, and functional stability compared to 2D cultures. They also improve in vivo relevance, enable detection of chronic toxicity, and allow for high-throughput screening of compounds.
Spheroids, another promising 3D model, maintain long-term cellular viability, structure, and functional stability compared to 2D cultures. They also improve in vivo relevance, enable detection of chronic toxicity, and allow for high-throughput screening of compounds.
However, like organ-on-a-chip and organoids, they still present challenges in measuring liver toxicity. They are time- and labor-consuming, methodologically complex, and require expensive tools.
Scientists from Charles River’s Discovery site in Budapest, Hungary, have been developing and refining a liver spheroid model that they originally created in collaboration with the University Szeged, within a framework of a research grant. Zsuzsanna Gáborik, PhD, Associate Director of Developmental Services for Charles River, Hungary, said along with the spheroid model, they have also developed a novel in vitro hepatic model that works like this: Hepatocytes are cultured in a sandwich configuration, and the liver cells are then treated with a five-compound chemical cocktail known as (5C). The 5C cocktail is designed to help to preserve hepatocyte-like characteristics of liver cells by reversing their loss of identity, dysfunction, senescence, pro-inflammatory gene expression and inhibiting epithelial-mesenchymal transition (EMT).
Last month, the Hungarian team published findings in Nature’s open-access journal Scientific Reports, showing that the model maintained hepatocytes in a fully mature state for up to 3 weeks, with full metabolic and transport capacity. The study also showed, for the first time, the utility of using multi-donor lots for long-term culturing rather than single-donor lots, thereby helping to avoid donor variability.
How this hepatic model ultimately compares to spheroid models or to animals remains to be seen, says Gáborik. “Our paper describes how the novel model works, and the validation is on the way to see how it performs in terms of predictive value compared to other models and animal data,” says Gáborik. “However, the new modalities beyond small molecules require more complex models. The ultimate focus of these models will be on derisking for drug-induced liver injury in the early stages of drug development.”
Don’t Discount the Value of Animals
Despite the steady uptick in research on alternative models for measuring DILI and iDILI, animals still offer features that their alternatives cannot. Amid the hunt for new tools and technologies to help reduce drug failure rates in the clinic, Parker says what is sometimes overlooked is how often animals prevent overly risky or ineffective drugs from ever reaching the clinic. Ironically, this results in the omission of important data, which undermines the true value of animals. “There is no way to retrieve data on studies where the drug failed in safety assessment. It doesn’t go forward. It doesn’t go into humans,” said Parker. “So, when the question becomes what’s the association between safety assessment in the human experience, absent from this discussion is a large cohort of studies that failed during the safety assessment procedure and thus are excluded from the final database used to compare animal vs non-animal detection methods.”
Still, Parker says there is a place for alternatives, particularly as a way to weed out questionable candidates in the early stages of drug discovery. “Some of the alternative models being developed are very useful and show a lot of promise. In my opinion, they should be performed prior to animal studies and should help refine the number of compounds that go into animals, thus reducing animal use,” says Parker.