Discovery
|
Melvin Lye, Christoph Eberle, PhD
How Merging NAMs and Traditional Models Look in Discovery
Realistically, animal models will not be completely abandoned any time soon. An emerging hybrid approach fuses the best of established preclinical standards with the potential of humanized, automated, and scalable future tools.
The drug discovery landscape is undergoing a paradigm shift. As the pharmaceutical industry grapples with high attrition rates and increasing ethical concerns over animal testing, integration of New Approach Methodologies (NAMs) — such as organoids, organs-on-a-chip, high-throughput screening, and AI-driven in silico models — is gaining momentum.
Yet, despite their promise, NAMs are not a panacea. The near future of drug discovery will depend on blending the strengths of traditional animal models with cutting-edge alternatives. Animal models have long served as the backbone of preclinical testing, offering systemic insights that are difficult to replicate in vitro. They provide valuable data on pharmacokinetics, biodistribution, and immune responses — aspects where NAMs still face limitations1,2. However, the translational gap between animal studies and patient outcomes remains a persistent challenge, with 90% of drug candidates failing in clinical trials despite promising animal data3.
NAMs offer human-relevant data earlier in the discovery pipeline, enabling better candidate selection and reducing unnecessary animal use. For instance, organ-on-a-chip models have demonstrated improved prediction of human-specific drug toxicity and metabolism4. Furthermore, AI and machine learning are accelerating target identification and compound optimization with remarkable precision5.
But neither approach can fully replace the other yet, which is why the path forward lies in merging these two very different approaches. A hybrid framework enables a tiered strategy: early-stage NAMs for high-throughput screening and human-relevant insights, followed by targeted animal studies to validate systemic effects and support regulatory submission. Such integration not only reduces ethical burdens but also enhances scientific rigor. As government-funded research institutions, such as the US National Institutes of Health (NIH), and global regulatory authorities now endorse NAMs in preclinical research, Contract Research Organizations (CROs) are increasingly tasked with leading the strategic shift away from traditional animal models. This transition, while promising, presents challenges for researchers and CRO clients alike—particularly in selecting fit-for-purpose platforms that balance scientific rigor, cost-efficiency, and translational relevance.
Rethinking Translational Strategy in the Era of NAMs
The NIH's funding incentive for non-animal testing, like organ-on-chip, organoids and other microphysiological systems (MPS) reflects a growing need to humanize toxicology and pharmacology research. Platforms from companies like CN Bio and Emulate have brought remarkable innovation to areas such as liver toxicity, absorption, and metabolic profiling. Charles River has been partnering with both companies to validate their technologies. However, scalability, reproducibility, and assay adaptability remain practical hurdles. For biotech startups or academic laboratories operating without the support of a CRO or external innovation partner, these hurdles can mean cost and resource drains. Below is a readiness self-assessment table for drug developers considering transitioning to NAMs-only or hybrid approaches (NAMs and animal models). It outlines key capability areas, describes what to evaluate, and highlights potential gaps that may need to be addressed.
Capability Area | Assessment Criteria | Potential Gaps |
|---|---|---|
Scientific Expertise | Availability of internal expertise in NAM technologies (e.g., organ-on-a-chip, organoids, MPS, AI models) | Can current methods handle fragile 3D models? Limited training in novel methodologies; reliance on traditional pharmacology |
Data Infrastructure | Ability to store, analyze, and integrate complex NAM and in silico data | Inadequate bioinformatics tools or data interoperability |
Validation and Reproducibility | Availability of validated NAMs with reliable endpoints for specific drug classes; development of assay transfer protocols (portable, standardized) | NAMs not yet validated for certain therapeutic areas or toxicity types |
Regulatory Strategy | Familiarity with regulatory requirements and submission of NAM-generated data | Uncertainty in regulatory acceptance; lack of precedents for approval pathways |
Animal Ethics Policy | Internal ethics policy supportive of 3Rs (Replace, Reduce, Refine) principles | Misalignment between corporate policy and emerging animal research standards |
Preclinical Workflow Integration | Capability to integrate NAMs into existing discovery and preclinical pipelines | Fragmented workflows; manual transfer of data across NAM and in vivo platforms; are workflows audit-ready or semi-manual? |
Budget and Investment | Sufficient allocation of funding for NAM tools, equipment, and training | High initial cost of transitioning to or implementing parallel systems |
Partnerships and Outsourcing | Access to CROs or academic partners experienced in NAMs | Lack of external collaborations or trusted NAM service providers |
Change Management | Organizational readiness to adopt new scientific paradigms and shift resource allocation | Lack of incentives or leadership support |
Predictive Performance Tracking | Mechanisms to benchmark NAM outcomes against animal and clinical data | Absence of performance tracking frameworks or historical baselines |
This initial framework empowers biotech leaders to identify friction points early and assess readiness across technical, operational, and regulatory domains. A core benefit of the hybrid model is that it tailors assay platforms to research objectives:
- In Vitro Target Profiling (e.g., Oncology): Leveraging cancer cell subtypes across different concentrations and molecular targets enables highly multiplexed testing for therapeutic selectivity.
- In Vitro ADME: Employing hepatocytes and microsomal systems to study circulation, metabolism, and excretion aligns better with human-relevant endpoints than with rodent models.
By advising study sponsors when and how to use NAMs within these divisions, CROs can offer strategic assay roadmaps that preserve translational accuracy while reducing reliance on animal studies.
The Role of Curiox C-FREE™ Pluto in Enabling Scalable Automation
Transitioning from animal to NAM-based workflows necessitates a robust automation infrastructure6. This is where Curiox's C-FREE Pluto system delivers both operational efficiency and cost-saving value. Designed to eliminate centrifugation in sample preparation, Pluto automates key steps such as:
- Antibody cocktailing for high-dimensional immunophenotyping
- Gentle cell washing and fixation
- Reagent handling in standard microplate formats
These capabilities are vital for workflows using fragile organoid models, primary human cells, or high-throughput cytometry platforms. Unlike conventional liquid handlers, novel processing systems like the Pluto require minimal optimization and operate on a benchtop footprint — making them ideal for laboratories seeking scalable, personalized automation solutions. In a recent implementation, Charles River used the Pluto LT platform to enhance lot-to-lot antibody validation workflows aligned with CLSI H62 guidance. The reduction in manual variability helped streamline QC, while Pluto’s gentle wash mechanism preserved cell integrity for downstream flow cytometry.
De-Risking Innovation: The Case for a Hybridized Pipeline
The pharmaceutical ecosystem is risk-averse by necessity, particularly in preclinical stages. The hybrid model offers a low-risk entry point to NAM adoption by allowing clients to retain traditional assays for benchmarking while progressively integrating advanced platforms. This dual-track strategy enables:
- Comparative data sets between animal models and human-relevant MPS platforms
- Reduction of resource-intensive animal studies in early screening stages
- Faster decision-making based on consistent, reproducible automated workflows
Regulatory agencies are beginning to support this paradigm. The U.S. Food and Drug Administration and European Medicines Agency have signaled openness to data from NAMs, provided they are validated and fit-for-purpose (FDA, 2023; EMA, 2022). Already, NAMs have replaced over 40% of dermal and skin irritation assays according to OECD TG431/TG439 data7, with the U.S. EPA committed to phasing out animal use for skin sensitization tests, albeit without giving firm deadlines8. Meanwhile, blood-based NAMs—particularly for immunotoxicology, cytokine profiling, and T-cell engagement—are gaining traction due to their scalability, access to human biology, and lower regulatory barriers. Applications are rapidly expanding in receptor occupancy, cytokine release assays, and CAR-T characterization workflows, particularly those requiring high cell viability and multiplex marker expression. As acceptance grows, hybrid strategies will become the standard, not the exception.
In this context, the C-FREE Pluto platform enables precise automation of TBNK panels using whole blood. A recent internal performance comparison demonstrated that Pluto achieves:
- 95% retention of CD45+ leukocytes post-lysis
- Consistent staining index across CD3, CD19, CD16/56, and CD4/CD8 subsets
- Reduced background noise and improved resolution in rare T-cell populations
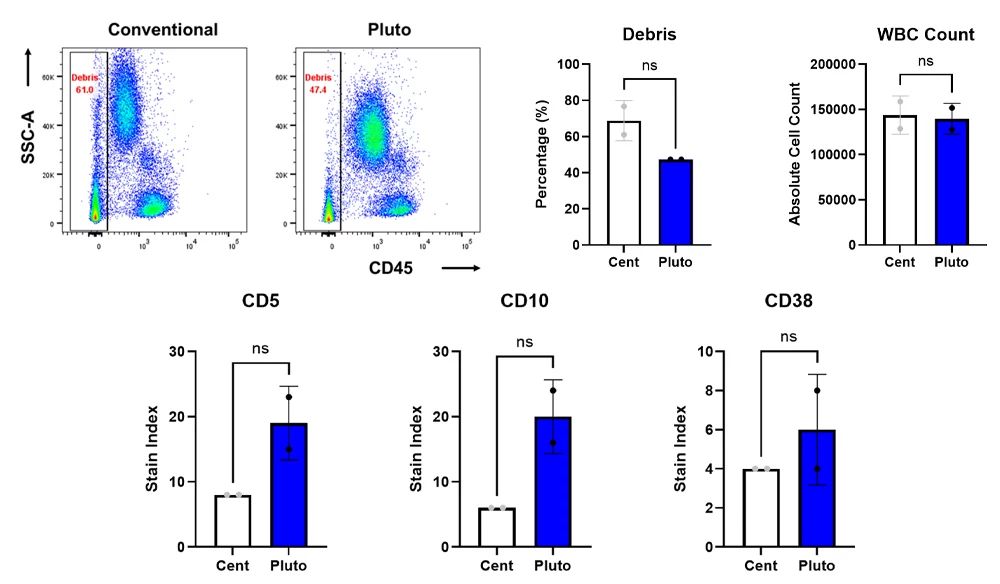
Figure 1. Comparison of sample quality between conventional and Pluto systems with representative scatterplots (a), percentage of debris (b), and white blood cell (WBC) counts. (c) Comparable performance based on major immune marker staining indices for stained samples processed by conventional centrifugation (Cent) and the Pluto platform.
These data validate the Pluto platform as an automation-ready tool for NAM workflows in immunophenotyping. The platform can further de-risk these transitions by providing validated, reproducible automation for sample prep—often the most variable step in flow cytometry-based assays.
Equipping Researchers with Strategic Options
Ultimately, this partnership between Charles River and Curiox Biosystems is about more than technology. It’s about giving researchers and biotech clients strategic optionality:
- When to use organ-on-chip models vs. traditional rodent in vivo platforms
- How to select assay workflows based on mechanism of action
- What automation tools ensure reproducibility, especially in flow cytometry and high-throughput toxicology
By combining Charles River's domain expertise in CRO services with Curiox's innovation in non-centrifugal sample handling, we are creating a new standard for translational strategy. As NIH and other agencies accelerate their commitment to non-animal methodologies, the research community must act with equal precision and foresight. A hybrid approach offers a pragmatic and scientifically sound pathway for the future of drug discovery. Leveraging the complementary strengths of biotech leaders from the traditional and novel methods sides, as well as translational researchers, and program managers, can drive innovation while improving both efficacy and ethics.
References:
- Mak IW, Evaniew I, Ghert M, Lost in translation: animal models and clinical trials in cancer treatment, Am J Transl Res, 2014, 6:114-118. PMCID: PMC3902221
- Swell F, Alexander-White A, Brescia S, et al. New Approach Methodologies (NAMs): identifying and overcoming hurdles to accelerated adoption, Toxicol Res (Camb)., 2024, 13:tfae044. https://doi.org/10.1093/toxres/tfae044
- Hay M, Thomas DW, Craighead JL, et al. Clinical development success rates for investigational drugs, Nat Biotechnol, 2014, 32:40-51. https://doi.org/10.1038/nbt.2786
- Bhatia SN, Ingber DE, Microfluidic organs-on-chips, Nat Biotechnol, 2014, 32:760-772. https://doi.org/10.1038/nbt.2989
- Mak KK, Pichika MR, Artificial intelligence in drug development: present status and prospects, Drug Discov. Today, 2019, 24:773-780. https://doi.org/10.1016/j.drudis.2018.11.014
- Holland I, Davies JA, Automation in the Life Science Research Laboratory, Front. Bioeng. Biotechnol., 2020, 8. https://doi.org/10.3389/fbioe.2020.571777
- OECD Test Guidelines for Skin Irritation/Corrosion (TG431/TG439), 2021.
- Grimm D, EPA scraps plan to end mammal testing by 2035, Science. https://doi.org/10.1126/science.zbmpq89
