
Cell & Gene Therapy
|
Alex Sargent, PhD
Leveraging Lentiviral Vectors
How lentiviral vectors are bringing lifesaving therapies to patients to treat cancer
As the world emerges from a global pandemic and people venture out into their new, normal lives, we could all well be forgiven for not wanting to hear or read any more about “viruses” in the news. But consider this bit of uplifting information: this year, thousands of people with aggressive and life-threatening forms of cancer will be treated with a cutting-edge therapy that depends on viruses.1
This novel therapy is called Chimeric Antigen Receptor or “CAR” T-cell therapy. This approach involves collecting the patient’s blood, isolating a type of cell from there called a T-cell, and modifying these cells with a CAR that recognizes and destroys the cancer. A majority of patient receiving this therapy will be cancer free after treatment and will stay cancer free for years and even decades. This means CAR T-cells represent a new cure for devastating types of cancer.
For CAR T-cells to be made, a type of virus know as a lentivirus is used in a process known as genetic engineering. It truly is the stuff of science fiction, but it is real and happening in labs and hospitals across the world. It all started nearly 40 years ago, with the discovery of HIV, the virus that causes AIDS.2
HIV is an incurable and lethal type of lentivirus that is especially good at infecting T-cells. Once researchers discovered HIV was the cause of AIDS, we were able to produce antiviral therapies to help people infected with the virus live longer. But it also left an intriguing possibility. Could this HIV virus be modified and controlled so that it could safely infect T-cells? T-cells are the crux of the immune system; infecting them with certain genes could drive them and the immune system to attack and destroy things we do not want in our body, cancer being key among them.
Today modified lentiviral vectors are mass produced by pharmaceutical companies all over the world. In these instances, the lentivirus is changed so it cannot reproduce, but it will safely infect certain cells to introduce genes into them. In the T-cells, the lentiviral vector infects them and transforms them into CAR T-cells that will attack and destroy cancer.
Roundtable: Trends in Lentiviral Vector Manufacturing
Bringing together acknowledged experts in the field of LVV design, manufacture, and regulation, join us to examine key trends and point the way to future success.
Watch On Demand
There are other methods to introduce genes into cells. CRISPR for example is a different gene editing system that can do this and has garnered much press these days.3 But lentiviral vectors have been around for a lot longer and have proven themselves to be a safe and effective way to accomplish genetic engineering and make therapies like CAR T-cells. Currently, all of the approved CAR-T drugs are for blood cancers. However, many of the CAR-T drugs in development are for solid cancers, such as colon, breast, and stomach cancer, which if approved could dramatically widen the market for this type of cancer therapy.4
Challenges with using lentiviral vectors
Still, using lentiviral vectors is not without its drawbacks. One is cost. Lentiviral vectors can be expensive to make, especially at an industrial scale. With the rise of CAR T-cell therapies in the past few years and the growing need for lentiviral vectors it has been a challenge to produce enough lentiviral vector to keep up with demand.
So how are researchers trying to solve this problem? One answer lies in using better methods to infect the T-cells with the lentivirus. Consider Figure 1 below.
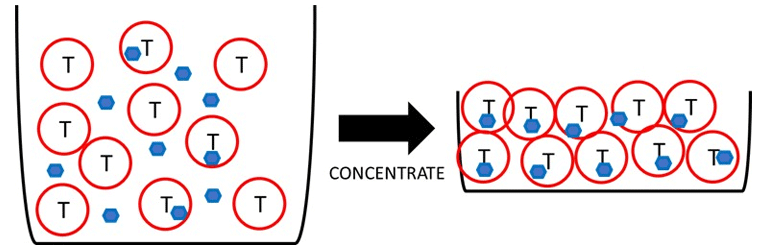
Figure 1. Concentrating the T-cells (red) and lentivirus particles (blue) together increases the likelihood that the virus will infect the cells to make CAR T-cells.
Concentrating the T-cells and the lentivirus together can increase the chance that a virus particle will successfully infect one of the cells and turn it into a CAR T-cell. New devices are emerging that use microfluidics to super concentrate the T-cells and lentivirus together to greatly increase the efficiency of making CAR T-cells.5 This means we can use a lot less lentivirus in our process of making CAR T-cells, helping to reduce costs in making this therapy and stretching out our supply of lentiviral vector so we can make more treatments for more patients.
As researchers learn how to better use lentivirus, we can make more effective and less expensive therapies with them. While recent technologies like CRISPR can also help us make these therapies, it is likely that lentiviral vectors are here to stay as a powerful tool in CAR T-cell therapies and other types of novel cell therapies.
So please take heart in knowing that while viruses can be very bad and cause serious illness, scientists across the world are learning to use them as a weapon in the fight against cancer and other dangerous diseases. The enemy of my enemy can be my friend, and when it comes to lentiviral vectors, they can be a formidable ally when used correctly.
References:
1. Charrot S, Hallam S. CAR-T Cells: Future Perspectives. Hemasphere. 2019 Mar 19;3(2):e188. doi: 10.1097/HS9.0000000000000188. PMID: 31723827; PMCID: PMC6746028.
2. Bourbon E, Ghesquières H, Bachy E. CAR-T cells, from principle to clinical applications. Bull Cancer. 2021 Oct;108(10S):S4-S17. doi: 10.1016/j.bulcan.2021.02.017. PMID: 34920806.
3. Dimitri A, Herbst F, Fraietta JA. Engineering the next-generation of CAR T-cells with CRISPR-Cas9 gene editing. Mol Cancer. 2022 Mar 18;21(1):78. doi: 10.1186/s12943-022-01559-z. PMID: 35303871; PMCID: PMC8932053.
4. Wang V, Gauthier M, Decot V, Reppel L, Bensoussan D. Systematic Review on CAR-T Cell Clinical Trials Up to 2022: Academic Center Input. Cancers (Basel). 2023 Feb 4;15(4):1003. doi: 10.3390/cancers15041003. PMID: 36831349; PMCID: PMC9954171.
5. Moore N, Chevillet JR, Healey LJ, McBrine C, Doty D, Santos J, Teece B, Truslow J, Mott V, Hsi P, Tandon V, Borenstein JT, Balestrini J, Kotz K. A Microfluidic Device to Enhance Viral Transduction Efficiency During Manufacture of Engineered Cellular Therapies. Sci Rep. 2019 Oct 22;9(1):15101. doi: 10.1038/s41598-019-50981-9. PMID: 31641163; PMCID: PMC6806008.
Alex Sargent – better known as “Sarge” – is currently the Director of Process Development at Charles River Laboratories. He obtained his PhD from Case Western Reserve University in Cleveland Ohio, where he studied the challenges and promises of stem cell biology, neuroimmunology, and Cleveland sports teams. He is passionate about the challenge of curing cancer, working on CAR-T and CAR-NK cell therapy process and analytical development from discovery, through regulatory submission, manufacturing, and clinical trials. He wakes up each day excited to help advance cell and gene therapy to treat and cure disease, with the steadfast goal of improving human lives.

Cell and Gene Therapy CDMO Solutions
We have supported the development of 10 FDA-approved cell and gene therapies and have conducted more than 900 studies in this field over the past year. Our unsurpassed end-to-end offering results in enhanced access to scientific and regulatory expertise via our multidisciplinary bench of experts to help you problem solve. Our focus is on accelerating and achieving your critical development goals from discovery to commercialization.

