Exquisite Science
|
Sandy Kimber
Vaccine Platforms 101
What are the different types of vaccine platforms, and how are they used?
Over the course of human history vaccines have transformed public health. The WHO estimates that 2-3 million lives are saved each year by current immunization programs, notably for children under 5 years old.
Protection through vaccination is based on the ability of the immune system to remember the pathogens that are encountered. Through this mechanism it’s possible to provide a small amount of the pathogen as a vaccine in a format that will not cause infection, but will be able to stimulate the immune system. It’s these immunological preparations that leads to the generation of antibodies that provides protection for “if” or “when” the pathogen is met throughout life.
There are many different types of vaccine platforms, and this short article will take a walk through them and highlight which platforms protect against which pathogens in the event of an infection.
Types of Vaccine Platforms
Live Attenuated Vaccines
The first vaccines were live attenuated vaccines (e.g. smallpox introduced in 1798 by Edward Jenner), where a small amount of the live pathogen is the vaccine material. In an immunocompetent individual the low-level replication simulates infection and in turn induces a robust immune response. Booster vaccinations will be often administered to provide greater protection. However, these vaccinations would not be suitable for those that are immunocompromised where there would be a possibility for uncontrolled replication. Figure 1 highlights several examples of live vaccines those developed for measles, mumps, rubella, yellow fever, influenza and more.
Inactivated Vaccines
Another traditional approach includes the inactivated non-live vaccines. Within this group there are several categories as listed in Figure 1. For instance, vaccines against polio and whooping cough will contain whole inactivated polio virus and whole cell Bordetella pertussis bacteria respectively. This technology is well established and safe for those with weakened immune systems and proven to generate a broad immune response due to the whole cell repertoire of antigens being available to the immune system. With the vaccine material being inactivated these vaccines are also incredibly stable providing an advantage logistically.
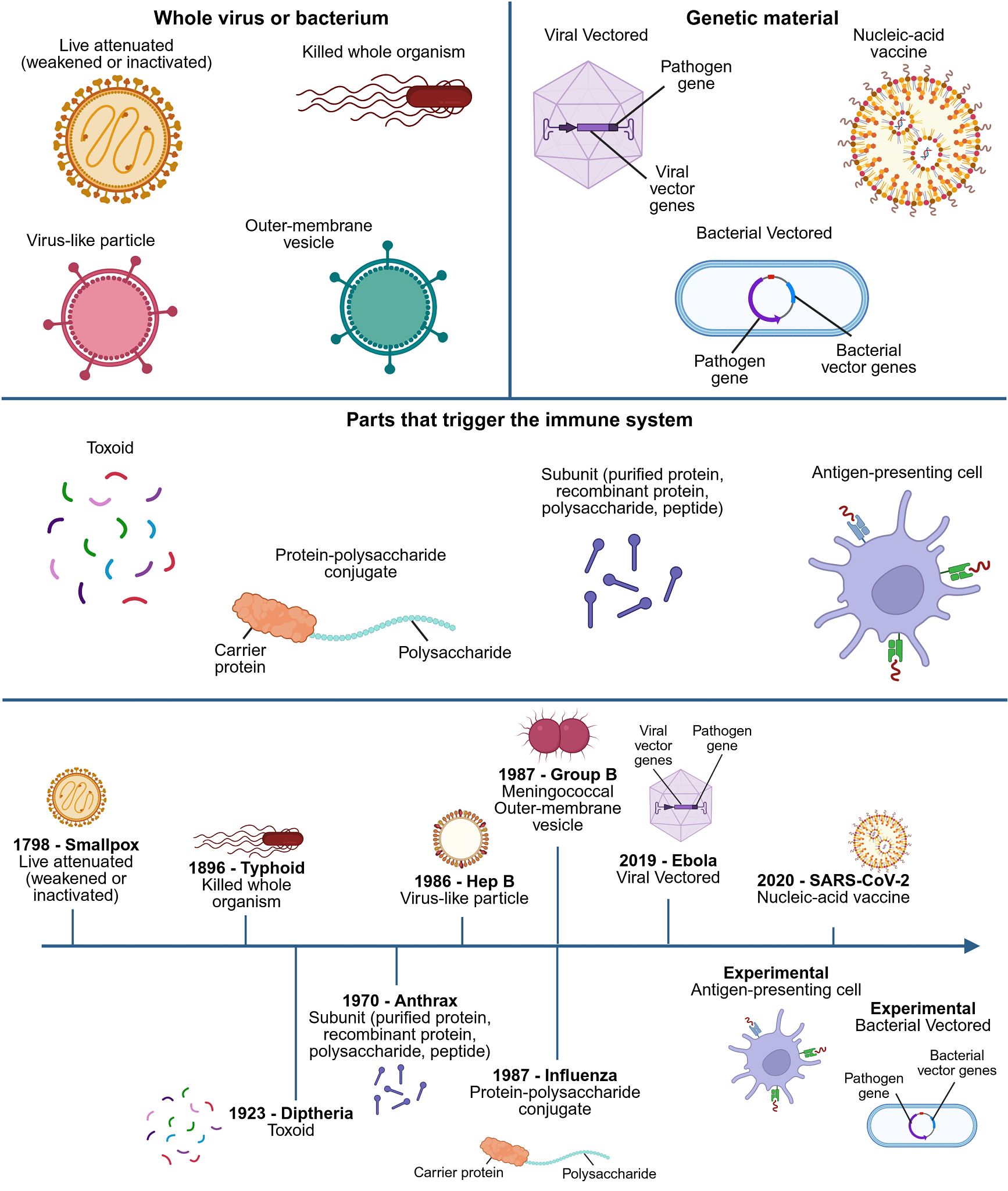
Figure 1: Overview of the different types of vaccines and a timeline for their introduction.
Pathogen v Toxin
When designing a vaccine, scientists will consider the development of the disease and not just the presence of the pathogen. This is certainly the case for the vaccines that are constituted with toxins. For instance, the vaccine for diphtheria includes the toxin that is released by Corynebacterium diphtheriae because it’s the toxoid that leads to breathing difficulties, heart damage, nerve damage and even death, not just the presence of the bacteria. This is also true of the vaccine for tetanus containing tetanus toxoid, an inactivated form of the toxin produced by Clostridium tetani.
Like with the use of toxins as vaccines, again other components of pathogens can provide ample stimulation of the immune system. This would be the case for vaccines designed for whooping cough, influenza, hepatitis B and others where the sugars or proteins that exist on the outer membrane of the pathogen are manufactured in bulk to produce the vaccine.
Across all vaccine platforms it’s not uncommon to see the addition of an adjuvant in the formulation. The word adjuvant is derived from the latin word adjuvare which means “to help” and in this context it’s to help boost the immunogenicity of the vaccine, (because the individual components need help to stimulate a protective immune response). E.g Aluminum salts are a common adjuvant and are an essential part of the vaccine for Diphtheria, Tetanus and Pertussis.
Other Types of Vaccine Platforms
Virus like particles (VLPs) are particles that mimic the structure of real viruses using surface proteins. They don’t contain any genetic material and therefore cannot replicate (e.g. vaccine for human papillomavirus vaccine). These vaccines are safe and highly effective but the manufacturing costs can be high.
Outer membrane vesicles are small (20–250 nm) components derived from the membrane of Gram-negative bacteria such as those from Neisseria meningitidis. The main causal agent of meningitis. They are considered easy to manufacture using cost-effective bacterial fermentation techniques.
Protein-polysaccharide conjugate vaccines combine bacterial polysaccharides with carrier proteins. These can turn weak antigens into potent ones by inducing both a B cell response and a strong, T-cell-dependent immune response, particularly in infants. Thus stimulating the innate and adaptive immune system (e.g vaccine for Haemophilus influenzae Hib).
A powerful tool in modern vaccinology is the viral vector vaccine. These are a combination of a harmless virus (the vector e.g. adenovirus that causes a mild cold) which carries genetic instructions for a pathogenic antigen into cells. This prompts the body to express the antigen and recognize it and become prepared for future encounters e.g. vaccines for COVID-19.
Nucleic acid vaccine are similar in that they deliver a message to be encoded to the cells of the recipient but do not require a viral vector. They deliver either DNA or mRNA and utilise a synthetic carrier like a lipid nanoparticle instead.
Newer vaccine platforms where candidates are still being tested include bacterial vector vaccines to encode antigens. These utilise non-pathogenic strains of Salmonella, Listeria or lactic acid bacteria to carry antigens and stimulate immune responses in the mucosa, with the aim to protect against pathogens entering through the gut or respiratory tract.
While broadly protection against infection has been described so far, there are also specialist vaccine platforms for cancer. These Antigen-Presenting Cell (APC) vaccines target antigens with immune cells that would typically interact with immune cells (such as dendritic cells) that fight infection or tackle a cancerous cell. These vaccines work by displaying the target antigens on the cell surface of the immune cells to activate the cellular arm of the immune system for clearance. These vaccines represent a personalised approach by modifying the patients’ own cells but are labour intensive and costly. Research is on-going to make this platform more accessible and cheaper to deliver.
Summary
Vaccine platforms have evolved over time and much research continues to further develop vaccines for a variety of pathogens. This evolution has moved from the early trial and error approach of the early pioneers such as Edward Jenner with the live attenuated vaccines to the killed whole pathogens with adjuvants and then onto modern sophisticated genetic-based platforms.
Future trends in vaccine development include the ability to respond quickly and flexibly with the modern platforms (such as the viral vector vaccines) to support pandemic preparedness, stability without refrigeration and novel solutions to provide immune protection against whole families of pathogens as opposed to specific strains.
References
Pollard AJ, Bijker EM. A guide to vaccinology: from basic principles to new developments. Nat Rev Immunol. 2021 Feb;21(2):83-100. doi: 10.1038/s41577-020-00479-7. Epub 2020 Dec 22. Erratum in: Nat Rev Immunol. 2021 Feb;21(2):129. doi: 10.1038/s41577-020-00497-5. PMID: 33353987; PMCID: PMC7754704.

Webinar: Development, Validation, and Implementation of GMP Release Potency Assays for Vaccines
This presentation will provide some guidance and information on the development, validation, and ultimately, the implementation of GMP batch release potency assays for vaccines.
Watch Now





