Biologics
|
Ulrike Herbrand, PhD
A Vaccine Potency Test that Doesn’t Rely on Animals
The novel bioassay is reliable, reproducible, and precise
Whether you are talking about the annual flu shot or a childhood vaccination, mass-produced immunization products need to be tested rigorously during manufacturing to make sure each vaccine lot induces a potent response—that is a response that models how well the vaccine will work in the real world.
Potency is a critical quality attribute or (CQA) for controlling quality, consistency, and relevant biological properties of vaccines. Until now, potency tests were mainly done in animals. But due to the high demand for animals, the lengthy operations and the high variability of in vivo methods, along with increasing focus on meeting our 3Rs (replacement, reduction, and refinement of research animals) objectives, laboratories have been trying to find in vitro alternatives to the animal. Finding in vitro alternatives that accurately reflect the immunogenicity of vaccines hasn't been easy, though.
Here's why.
Many preventive vaccines are based on protein antigens and injected into the muscle. The specific protein antigen is taken up by dendritic cells and transported to draining lymph nodes. The dendritic cells present the peptides of the vaccine protein antigen via MHC molecules, leading to the activation of T cells through their T cell receptors(TCRs). CD8+ memory T cells are formed, which can rapidly multiply if they encounter the pathogen in the future. CD8+ effector T cells play a crucial role in clearing infected cells (Figure 1).
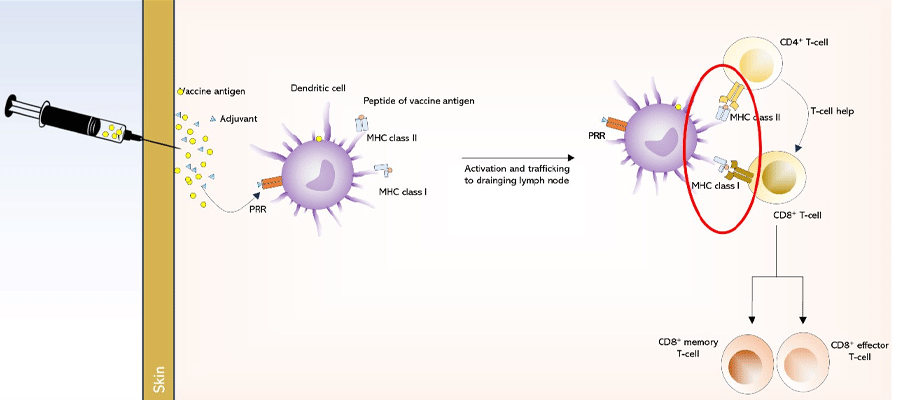
Figure 1: The generation of an immune response following immunization with a conventional protein antigen.
This complex cascade of events that occurs during vaccination must be captured by an in vitro assay to be effective.
While primary cells from healthy donors could be an option to recapitulate the crucial steps of vaccination and demonstrate functionality, their high donor variability and unfriendly quality control features make them unattractive for both lot release and stability testing. Established cell lines are easier to obtain and handle, but they often lose some functionality, rendering them unsuitable to reflect the mechanism of action (MoA) in the context of vaccine testing.
Fortunately, there are other alternatives than these options. Charles River and its collaborator Promega recently showed how a high-quality assay derived from a single cell line that differentiates into mature dendritic cells, and a modified T cell line, represents a solution to the problem of how to measure vaccine potency in vitro. The T cell line was used to generate a TCRαβ knockout platform. Based on this, Promega produces TCRα and TCRβ reporter cells specific for the respective vaccine. These reporter cells recognize the vaccine antigen presented by the antigen-presenting cells (APC). The assay’s readout is a luminescence signal based on the specific activation of the luciferase response element, which is the second modification of the T cell line. The time to result is 3 days in a standard cell culture lab. The assay can generate full dose-response curves of reference standard and test substance, suitable for statistical evaluation of a typical relative potency assay according to guidelines EP 5.3 and USP <1032> to <1034> (c.f. Figure 2).
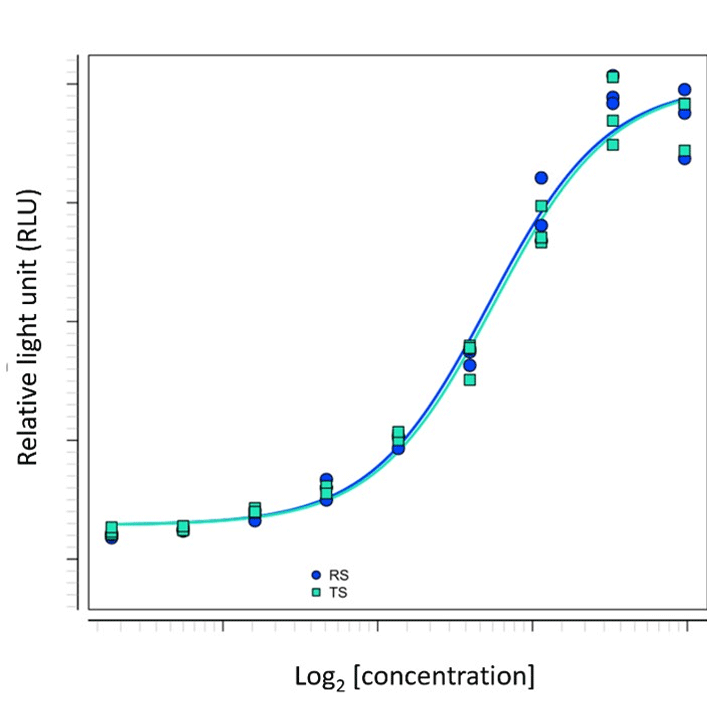
Figure 2: Typical dose response curves of reference and test item of a protein vaccine within the in vitro potency assay
This MoA reflective bioassay is reliable, reproducible, and precise. Importantly, it avoids the use of primary cells, which are tedious to handle, challenging to bridge from lot to lot and often highly variable. The MoA assay is also stability indicating and aligns well with the principles of the 3Rs. And most important, it is helping researchers to reduce if not replace the use of animals for vaccine potency tests, a worthy goal for any vaccine developer.
Sascha Karassek also contributed to this article. Sascha joined Charles River Laboratories in 2018 and is currently a subject matter expert in the Bioassay Research & Development team at Charles River Laboratories’ site in Erkrath, Germany. He has a PhD in Biochemistry from the Ruhr-Universität Bochum (Germany). Sascha is an expert in mechanism of action-reflecting bioassays for protein therapeutics as well as for advanced therapy medicinal products.
Check out the Alternative Methods Advancement Project (AMAP) and learn more about the scientific and technological innovations Charles River is using to lead the industry into the next frontier of drug discovery and development.




