Direct Inoculation and Membrane Filtration Methods
Sterility testing is required to ensure viable contaminating microorganisms are not evident in a product. This testing is conducted by direct inoculation or membrane filtration methods and can be performed in an isolator or cleanroom environment.
Sterility Testing Techniques
Direct Inoculation
- The test article is directly inoculated into two types of media to allow for the detection of both aerobic and anaerobic microorganisms.
- After inoculation, both media types are incubated for 14 days. Intermittent observations as well as a final observation at the end of the testing period are conducted to detect evidence of microbial contamination.
Membrane Filtration
- Sterile, enclosed units allow for the simultaneous filtration of equal volumes of test samples through two membrane filters.
- Samples are incubated in two types of media for 14 days, facilitating the detection of both aerobic and anaerobic microorganisms.
Need help kicking off your sterility testing project? Our specialists are here to support you.
Request A Consult
Bacteriostasis/Fungistasis Testing – B/F Testing
In conjunction with the sterility test, bacteriostasis fungistasis testing is performed to assess whether or not the test article is inhibitory to the growth of microorganisms. The performance of the B/F test is necessary to validate the sterility result and confirm that there are no antimicrobial properties within the test article that would inhibit the detection of microbial organisms during the sterility assay.
Vaporized Hydrogen Peroxide (VHP) Ingress Testing
Vaporized hydrogen peroxide (VHP) ingress testing is performed within an isolator that undergoes VHP decontamination. This assay evaluates if VHP ingress into a test article container is evident, which may affect the validity of the results.
Outsourced Rapid Sterility Testing
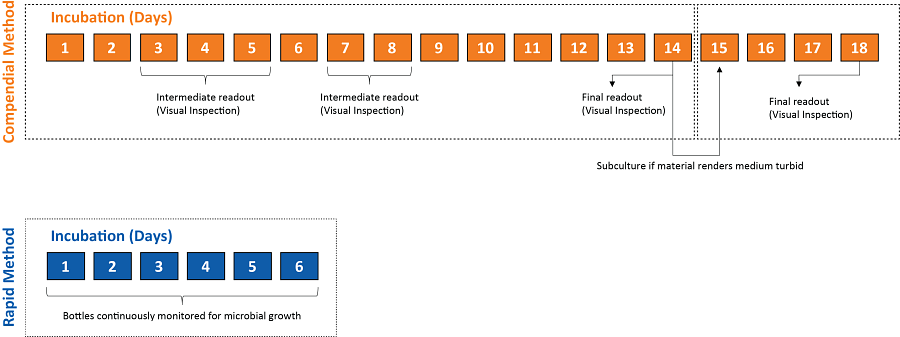
Traditional culture-based sterility testing has been the “gold standard” over the past century for ensuring the production of microbiologically safe products. However, evaluating pharmaceuticals and biopharmaceuticals using the compendial sterility test method requires a lengthy incubation period and visual inspections of macroscopic growth at defined intervals throughout the incubation period.
To overcome the limitations of the traditional sterility testing methods, we offer rapid sterility testing using fully automated microbial detection technology. Using this rapid test method provides you with a means to quickly evaluate microbial growth independently of culture turbidity, making the testing platform suitable for short shelf-life drug products (e.g., cell and gene therapies, vaccines, etc.).
Benefits of Rapid Sterility Testing
- Time to results cut by > 50% from compendial method
- Intermediate results provided upon request
- Fully Validated method per USP <1223>
- Results in as little as 6 days
Compendial vs. Rapid Sterility Assessment
Regulatory Changes: USP 73 & 1071 Updates at a Glance
The latest chapters of USP <73> and <1071> introduce key updates impacting sterility testing and rapid microbiological methods (RMMs). Understanding these changes is crucial for compliance and optimizing your microbial testing strategy. Our expert guide breaks down what’s new and how Celsis® can help you stay compliant and efficient.
Explore the Guide
Frequently Asked Questions (FAQs) for Sterility Testing
-
What is sterility testing?
Sterility testing is designed to demonstrate the presence or absence of extraneous viable contaminating microorganisms in biological parenterals designed for human use.
-
Why is sterility testing performed for 14 days?
The sterility assay is 14 days allowing for an appropriate incubation period to detect slower-growing microorganisms. While many bacterial and fungal organisms can be detected in a shorter incubation period, some microorganisms require a longer incubation period to proliferate. For shorter incubation times, try our Celsis® rapid microbial detection instruments and get results in as little as six days.
-
What is USP <71> sterility testing?
USP <71> sterility testing reflects the chapter within the United States Pharmacopeia (USP) that outlines the manner in which a sterility assay should be performed, including details of the methodology and how a product should be tested based upon sample size and fill volume.
-
How long does sterility test method validation take per USP?
The new chapter of USP <73> means that for short-life products sterility test method validation per USP <1223> is no longer needed. Following USP <73>, it is anticipated that method suitability and incubation time could be completed in under two months. If you choose Charles River to execute your USP <73> study, we will provide an approved report within 60 days.
-
What are the USP guidelines for sterility testing?
USP below <1000> are general chapters. General chapters may contain descriptions of tests and procedures, general information on the interpretation of compendial requirements, or general guidance on official substances or official products. USP <71> provides sterility test descriptions. USP <72> and <73> provide sterility test descriptions for short-life products via respiration-based microbiology method and ATP-bioluminescence technology respectively. The USP publishes General Information chapters which have reference numbers greater than a <1000>, including <1071> and <1223>. The chapters in this section are information, and aside from excerpts given herein from Federal Acts and regulations that may be applicable, they contain no standards, tests, assays, nor other mandatory specifications, with respect to any Pharmacopeial articles. The excerpts from pertinent Federal Acts and regulations included in this section are placed here in as much as they are not of Pharmacopeial authorship. Revisions of the federal requirements that affect these excerpts will be included in USP Supplements as promptly as practical. The official requirements for Pharmacopeial articles are set forth in the General Notices, the individual monographs, and the General Tests and Assays chapters of this Pharmacopeia.
