Dendritic Cell Product Information
What are Dendritic Cells?
Dendritic cells are derived from purified monocytes. CD14 monocytes are positively selected and are cultured in the presence of GM-CSF and IL-4 to differentiate them into DC cells. Presence of DC cells is confirmed by morphology and surface marker expression.
CD11c is a glycoprotein that is highly expressed on the surface of monocyte-derived immature dendritic cells. This marker protein is used to identify the frequency of dendritic cells within the white blood cell population (WBC), and to validate post-selection purity. DC cells make up approximately 1% of circulating peripheral blood mononuclear cells. DC cells have been used to develop in vitro assay methods for immunological reactions including T-cell stimulation, antigen presentation, contact sensitization, and more.
Validated Quality and Purity
Dendritic cells from Charles River are guaranteed to have >90% purity and >80% viability as measured by flow cytometry. DC cells are cultured in vitro from monocytes and are then cryopreserved in a serum-free and protein-free GMP grade CryoStor™ CS10 cryopreservation medium (BioLife Solution, Seattle, WA). For cryopreserved DC cells, either prepare cells for long-term storage in Liquid Nitrogen vapor phase or thaw for use. Storage in liquid phase nitrogen is NOT recommended. Short-term storage of cells (less than two weeks) at -80°C is acceptable but should be minimized to ensure maximum stability. Once thawed, samples must be used immediately.
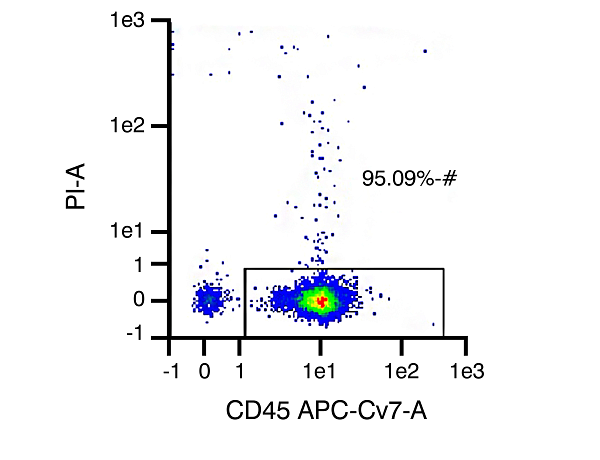
A
B
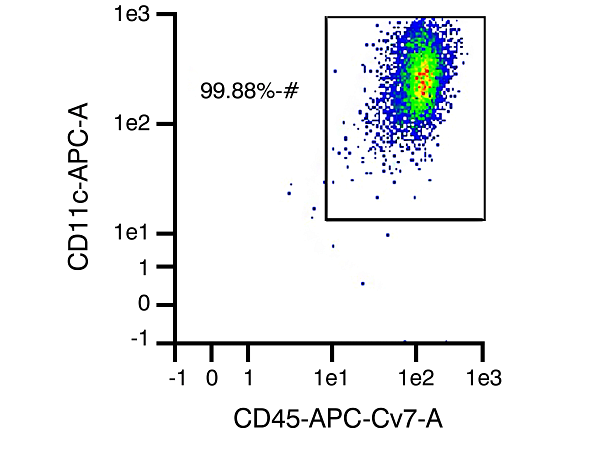
Figure 1. In Panel A, CD45 and Propidium Iodide (PI) are used to assess purity and viability of PBMCs. CD45 is expressed on all leukocytes and is used to indicate purity of the white blood cell (WBC) component. Because PI cannot pass through the membrane of live cells to stain DNA, it serves as an indicator of viability. Representative data shown here indicates viability of 95.09% – meaning >95% of cells in this sample are viable WBCs (may not be indicative of received samples). In Panel B, CD11c surface marker is targeted for immunomagnetic labeling to identify DC cells present following differentiation of monocytes in culture. When comparing levels of CD11c versus expression of CD45, the purity of the sample for dendritic cells can be assessed. This graph shows a representative data set with dendritic cell purity of 99.88% (actual purity may vary somewhat from sample to sample).
Dendritic Cell Clinical Applications
Dendritic cells have been used to develop in vitro assay methods for immunological reactions including T-cell stimulation, antigen presentation, contact sensitization, and more. The unique functions of these antigen-presenting cells are being investigated for their therapeutic potential in several different cell therapy clinical trials.
- Dendritic cell cancer vaccines - Autologous (collected from the person being treated) dendritic cells which have been genetically engineered to carry a cancer cell-related antigen is being evaluated for their efficacy in treating leukemia.
- Dendritic cells in combination therapies - Several different cancer immunotherapy strategies are being developed in hopes of eliciting long-term anti-tumor immune response. Scientists are investigating the efficacy of dendritic cell vaccines as combination therapies to be used alongside more traditional methods such as chemotherapy and monoclonal antibodies.
Lineage of CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs)
Receive a complimentary poster to access information on immune cell markers, differentiation factors, cytokines, technical protocols, media recommendations, how-to videos, and more.