What are Human T Cells?
Human T cells are lymphocytes that play a central role in the adaptive immune response to pathogens or diseased cells. These cells are used as critical starting materials throughout the research and therapy development spectrum including cell and gene therapies, oncology, vaccine development, and immunotherapies (CAR-T). Human T cells are also responsible for immune responses that lead to rejection of a transplanted organ, most autoimmune diseases, and some allergic reactions.
T cell subtypes are distinguished by the presence or absence of specific cell surface proteins, most commonly known as CD45, CD3, CD4, and CD8. These markers play a crucial role in T cell activity and are used as confirmatory markers for distinguishing functional T cells. We provide a wide selection of human T cell subtypes to meet your research requirements.
Buy T Cells For Your Research
Our human T cells are isolated from fresh leukopaks by positive or negative selection using immunomagnetic cell separation procedures. Flow cytometry analysis is used to validate purity and viability. Leukopaks are collected at our donor center from healthy donors that have consented under an IRB approved protocol. Our donor center is FDA-registered, state-licensed, and certified with the AABB and CLIA.
-
CD3+ Pan T Cells
CD3+ Pan T Cells – Available in 5M, 10M, 25M, and 50M vial sizes with positive or negative selection options. CD3 is the primary T cell surface biomarker that is critical for the signaling cascade activating T cells and is used to distinguish T cells from other immune cells.
-
CD4+ Helper T Cells
CD4 Helper T Cells – Available in 10M and 25M vial sizes with positive or negative selection options. CD4+ helper T cells perform multiple functions, including activation of B cells and CD8+ cytotoxic T cells. CD4+ helper T cells mediate anti-tumor immunity and are thus critical to cancer immunotherapy research.
-
CD8+ Cytotoxic T Cells
CD8+ Cytotoxic T Cells – Available in 5M, 10M, and 25M vial sizes with positive or negative selection options. CD8+ cytotoxic T cells (also known as cytotoxic T lymphocytes, or CTLs) are the main effector cells of the adaptive immune system attacking invasive target cells. Cytotoxic T cells are important for killing cancerous or virally infected cells and play a key role in cancer immunotherapy.
-
Naïve and Memory T Cells
Naïve CD3+ T Cells
Human T cells which have not yet been activated and can therefore respond to antigens from unfamiliar pathogens that have not previously been encountered.Naïve CD4+ T Cells
Available in a 5M vial size with negative selection option. Naïve CD4+ T cells are activated after interaction with antigens. These cells play a key role in regulating the immune system and are known to suppress the immune response in cases of chronic infection.Memory CD4+ T Cells
Available in a 10M vial size with negative selection option. These are CD4+ T cells that have already encountered an antigen. They lie dormant in tissues and protect the body from the re-emergence of potentially pathogenic or harmful cells.Memory CD8+ T Cells
Available in a 5M vial size with negative selection option. These cells persist long-term and are capable of quickly proliferating and regaining their former cytotoxic function following an immune challenge. CD8+ memory T cells are being investigated as a therapeutic strategy for increasing the strength and longevity of human T cell therapy treatments. -
Gamma-Delta (γδ) T Cells
Available in 2M and 5M vial sizes. Gamma-delta (γδ) T cells represent a relatively small subset of the human T cell population. They are unusual in that their activity is not dependent on the major histocompatibility complex (MHC) to recognize foreign antigens which makes them an important target for immunotherapy research. Gamma-delta T cells are known to recognize and attack diverse cancers.
-
Regulatory T Cells (Treg Cells)
Natural Regulatory T Cells (nTregs)
Available in a 2M vial size. Natural Regulatory T cells are isolated directly from a leukapheresis collection and immediately cryopreserved. The most widely accepted phenotype for natural Tregs is the co-expression of CD4 and CD25 surface markers and the absence of CD127 expression on the cell surface with additional intracellular expression of FoxP3. Treg cells are critical to the maintenance of immune cell homeostasis as they can suppress the activation and expansion of other T cells. Regulatory T cells are being investigated for their potential role in the treatment of autoimmune diseases such as arthritis and lupus.Induced Regulatory T Cells (iTregs)
Available in a 2M vial size. Induced Treg cells are differentiated in-vitro from CD4+ T cells. Induced regulatory T cells do not show a significant intracellular level of FoxP3 expression, but they retain their immunosuppressive capability similar to the natural Tregs (Tregs isolated directly from peripheral blood).Expanded Regulatory T Cells
Available in a 2M vial size. This Treg population is obtained by in-vitro expansion of natural Tregs (Tregs isolated directly from blood). Expanded Tregs retain the immunosuppressive activity of natural regulatory T cells as well as the classical Treg immunophenotype i.e., CD4+, CD25+, and CD127- with FoxP3 intracellular expression. -
Antigen-specific T Cells
Antigen-specific T cells are human T cells that have been activated by a specific antigen derived from viruses, bacteria, tumor cells, and in the case of autoimmune disease, peptides derived from normal tissues. ASTCs offer a unique tool for studying the activation and specificity of the immune system. Antigen-specific T cells are used to study cancer-intrinsic evasion of cytotoxic T cells, assess tumor-specific cytotoxicity in response to antigen modification, evaluate immunomodulatory activity to novel molecules, and examine cytotoxic response after microfluidic delivery of antigen to less conventional antigen-presenting cells.
Learn More -
Natural Killer T cells (NKT Cells)
Human Natural Killer T cells (NKTs) are very rare T lineage cells that share morphological and functional characteristics with both T cells and NK cells. Human Natural Killer T cells are crucial to studying autoimmunity, examining the link between adaptive and immune responses, and developing immune cell therapies.
How Much Do You Really Know About Leukopaks?
Find out what they are and why they are important for immune research and cell therapy development. Our comprehensive guide covers best practices, considerations, applications, and in-depth characterizations for leukopak collections.
Download Guidebook
Human T Cell Quality and Purity Powers Your Research
Human T cells can be distinguished from other lymphocytes by the presence of a T cell receptor (TCR) on their cell surface. Human T cells originate as precursor cells in the bone marrow and then migrate to the thymus, where they differentiate into several different cellular subtypes with distinct functions. We understand that our product quality directly impacts your research quality. Our primary isolated human CD3+ T cells are guaranteed to have >90% purity and >95% viability as assessed by flow cytometry.
Our human T cells are either positively or negatively selected via immunomagnetic bead separation from a healthy donor leukopak collection. During positive selection, the desired cells are targeted with the beads for isolation. Conversely, during negative selection, unwanted cell types, rather than target cells, are labelled for removal. Although negative selection may result in lower T cell yields, it is still the preferred method of isolation to avoid T cell activation. Following immunomagnetic isolation, human T cells are tested for purity and viability by flow cytometry. Serological testing and infectious disease screenings are performed for all samples. The human T cells are isolated same day and onsite from a leukopak collection at our donor center and then cryopreserved immediately in CryoStor® CS10 (BioLife Solutions, Seattle, WA). T cells are stored in the vapor phase of liquid nitrogen prior to shipping.
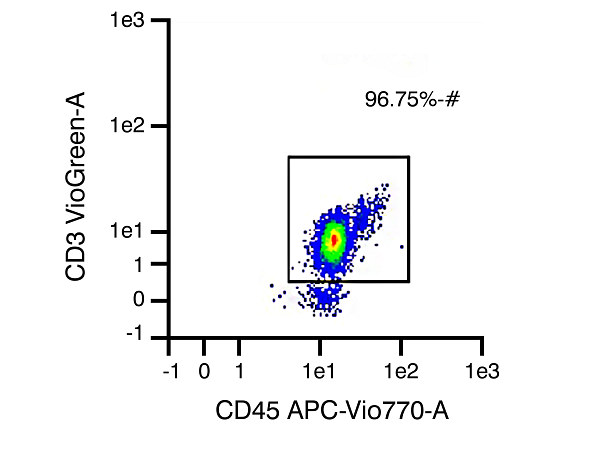
CD3 Pan T cells
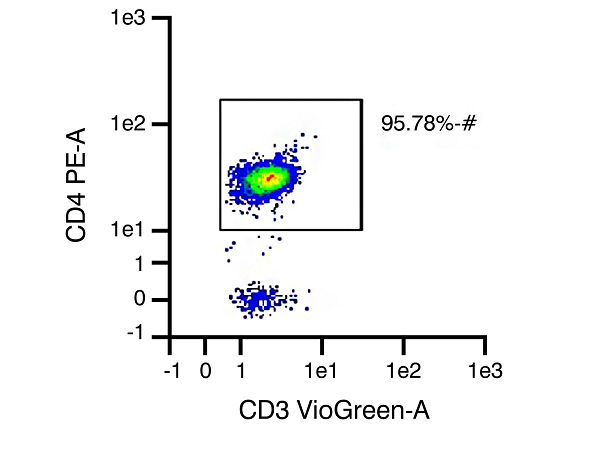
CD4 Helper T cells
CD8 Cytotoxic T cells
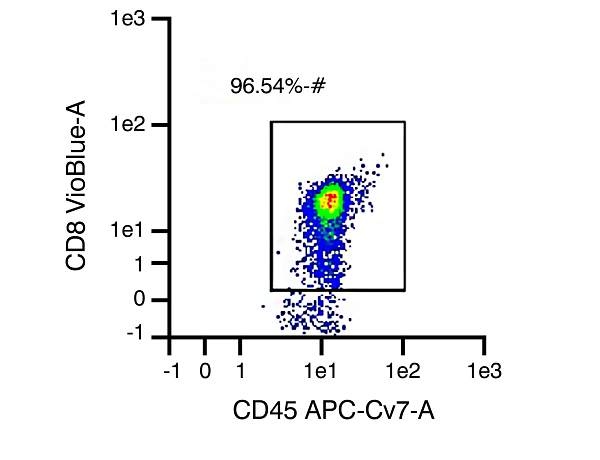
Figure 1. Representative data set from different isolations of human T cell subtype populations, with T cell purity of 96.75% CD3 expression (pan T cells), 95.78% CD4 expression (helper T cells), and 96.54% CD8 expression (cytotoxic T cells), respectively, after isolation.
Validated Human T Cell Yield
Process yield is a critical starting material criterion that directly impacts the quality of the final product. Successful cell therapy candidates must demonstrate consistent purity and efficacy. Therefore, starting materials for these products must contain a sufficient yield of therapeutic target cell type to withstand processing losses and meet stated efficacy values. Cell therapy starting materials must also demonstrate consistent target cell yields across multiple collections.
In a comprehensive study to characterize T cell yields and assess inter-donor variability in leukopak collections, Charles River analyzed a cohort of 69 healthy donor collections by examining the different human T cell subtypes through flow cytometric analysis. On average, the CD3+ T cell population represented about 47.2% of the total WBC population, while the CD4+ and CD8+ T cell subsets represented on average about 31.6% and 14.6% respectively of the total WBC population.
The study also emphasizes the existence of inherent donor-to-donor variability. This variability may be beneficial, as patient populations are diverse, and manufacturers prefer a product with demonstrated efficacy for the highest number of patients possible. There are instances when limiting donor variability is essential; for example, during development of allogeneic therapies to demonstrate process consistency, or during long-term clinical trials. In these cases, access to reliable and recallable donors is beneficial.
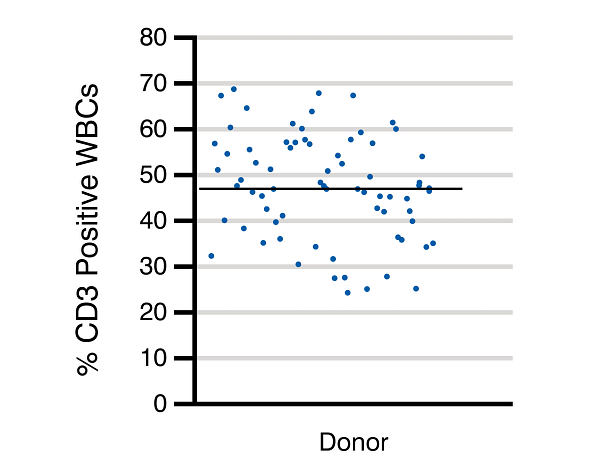
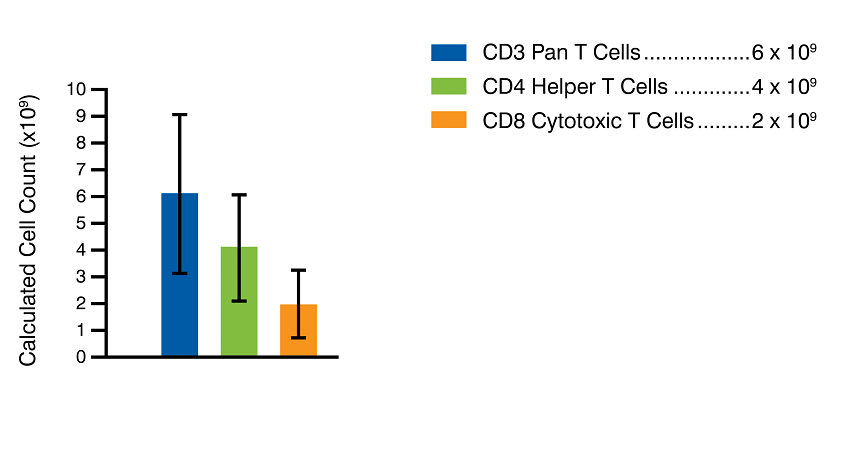
Figure 2. Despite donor variability, CD3+ T cells average about 47% of the total WBC population in a leukopak collection (Panel A, black line). In an average leukopak of 12.7 x 109 cells, this would correlate to an anticipated average of CD3+ T cell counts equaling roughly 6 x 109 cells, and CD4+ and CD8+ T cell counts equaling around 4 x 109 and 2 x 109 cells, respectively (Panel B). Mean post-collection WBC viability of the 69-donor cohort was 99%, with 1% standard deviation. CD3+ T cell viability post-isolation is guaranteed to be >95% as assessed by flow cytometry.
Consistent Human T Cell Products for Consistent Results
We are dedicated to providing optimal and consistent starting material to support cell therapies. Even though inherent donor-to-donor variability will always exist, with proper training, adherence to best practices, and standardized methods, collection-to-collection variability can be minimized. One strategy to minimize variability for certain applications is to utilize recallable donors.
We analyzed data from five separate collections from the same donor in order to ascertain the relative consistency of human T cell subtypes across multiple collections. Highly consistent profiles were observed in the percentage of CD3+ T cells as well as the CD4+ and CD8+ T cell subtypes per total white blood cell population (Figure 3 A). The ratio of CD4+ to CD8+ T cells was also highly consistent from one collection to the next from the same donor (Figure 3 B).
3A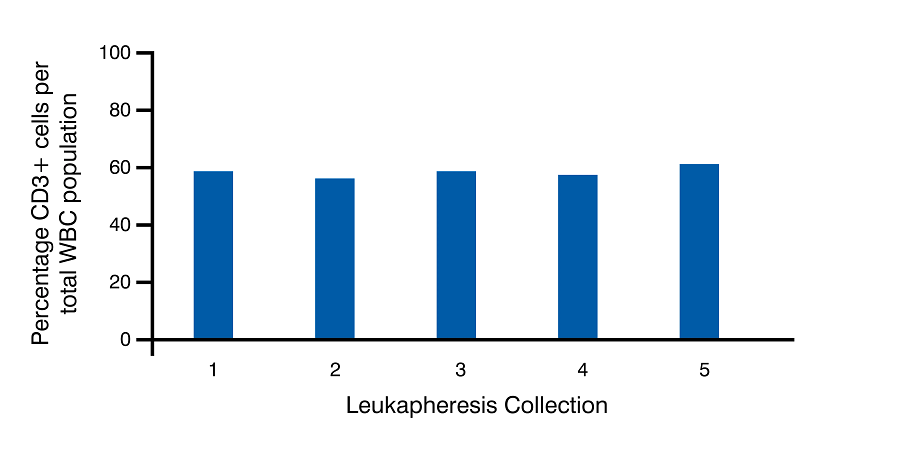
3B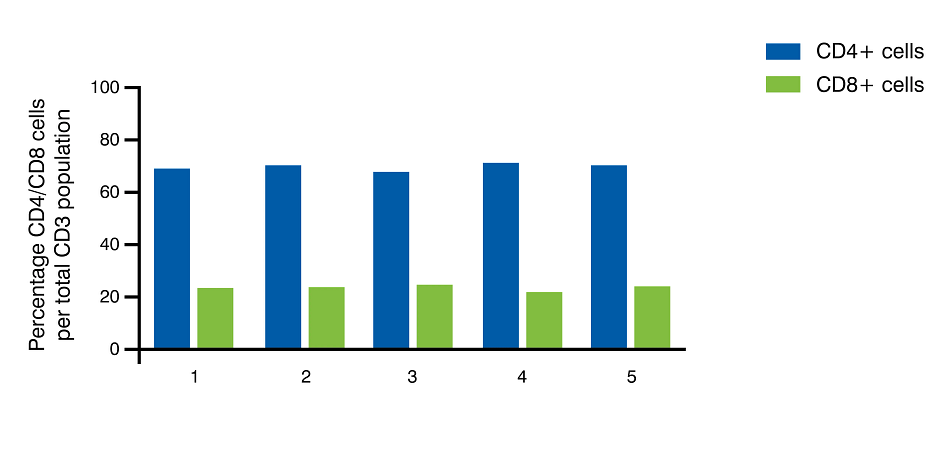
Figure 3. CD3+ values are reported as the percentage of CD3+ cells per total WBC yield. CD4 and CD8 cells are reported as percentage of CD3+ cells. Representative data from multiple collections from the same recallable donor is shown.
Optimize Cell Therapy Manufacturing Efficiency with Cryopreserved Cells
View this webinar to learn how purified and cryopreserved cell populations streamline logistics, reduce variability, and enhance transduction efficiency in CAR-T production.
Watch on Demand