NA CDMO approved by EMA to commercially produce allogeneic cell therapy drug product
GMP-compliant products supporting development and clinical manufacturing of allogeneic cell therapies
FDA-approved cell and gene therapies supported
Accelerate and Scale With an Allogeneic Cell Therapy CDMO
Turning donor cells into a reproducible, scalable drug product that meets regulatory compliance standards, makes manufacturing allogeneic cell therapies uniquely complex. As a full-service allogeneic cell therapy CDMO with integrated assay development and comprehensive analytical testing, we provide comprehensive support throughout the entire development process from early phase to commercial production.
When working with T cells, B cells, tumor-infiltrating lymphocytes (TILs), hematopoietic stem cells (HSCs), dendritic cells, and more, we start with the end in mind, leveraging our Manufacturing Science and Technology (MSAT) team to ensure your drug product is tech transfer, scale-up, and GMP ready. Our 3D cell culture capabilities can scale your allogeneic cell therapy from one to 10 liters using stir tank or rocking bioreactors or one to 30 square meters with a fixed-bed bioreactor. With our Manufacturing Execution System (MES), efficiency and optimization is designed into our manufacturing processes for allogeneic cell therapies, By leveraging automated and closed-systems performed on leading platforms, our allogeneic cell therapy manufacturing processes are already validated from targeted donors, allowing us to shorten development timelines. Additionally, with our data-driven approach, our Enterprise Resource Planning (ERP) eliminates waste and keeps overhead for raw materials down.
As part of our continual testing of the latest technologies, our automated formulation and fill finish capabilities provide a consistent process for vials and bags that can easily scale to multiple batches to bring your life-saving therapy to more patients faster. Our team of quality experts also ensures your final drug product is particulate and contaminant free with visual inspection before labeling and packaging.
Uncover the latest techniques to recognize, target, and eliminate cancer cells and tumors. Examine each cell type, plus autologous and allogeneic options, to better understand these evolving therapies..
Get the Insights
“We are incredibly proud that our Memphis facility has received EMA approval to commercially manufacture an allogeneic cell therapy. We are looking forward to continuing close partnerships with our clients, and supporting future commercial projects, bringing these potentially curative therapies to patients, sooner.”
Birgit Girshick, Corporate Executive Vice President & Chief Operating Officer, Charles River
Streamlined Cell Sourcing Solutions from an Autologous Cell Therapy CDMO
Simplify your allogeneic cell therapy manufacturing with our in-house cell sourcing products and services, supported by a robust, recallable, and well-characterized donor network.
Advance your cell and gene-modified cell therapy with GMP-compliant leukopaks, PBMCs, and bone marrow. Choose your fit: CliniPrime™ cellular starting materials follow a standardized production process for speed and consistency, while GMPrime™ cellular starting materials offers a customizable production approach tailored to your program needs.
Cell Sourcing for Research and GMP Human Cells
- Leukopaks (fresh, cryopreserved, mobilized, disease state, GMP)
- Human Immune Cells (PBMCs, T cells, and other isolated and cultured immune cell types)
- Human Stem cells (bone marrow aspirate, cord blood, CD34+ and CD133+ HSPCs, embryonic stem cells, iPSCs, mobilized leukopaks)
- Human Blood Products (plasma, platelets, serum, whole blood)
- Cell Freezing Media (Cryostor®, Hypothermosol®)
- Disease State Biospecimens
- GMP-compliant leukopaks, PBMCs, and bone marrow
Accelerate your CGT's clinical and commercialization path with our GMP-compliant CliniPrime™ and GMPrime™ cellular starting materials, providing standardized or customized production processes to meet the specific needs of your program.
Shop our Products
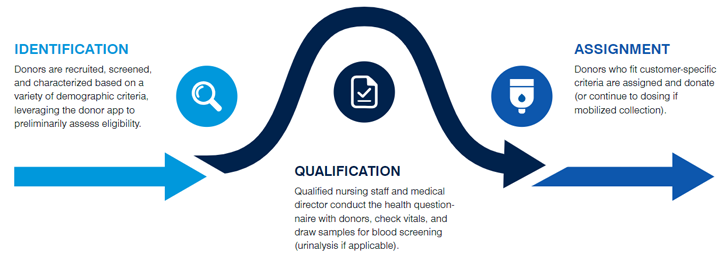
Built on long-standing donor relationships, our recruitment and management model supports a reliable, consistent supply of donor materials, helping keep your program on track from development through scale-out. From the outset, we align donor recruitment with your program needs through planned maintenance cycles, including donor recall, screening, and defined collection intervals. This continuity helps address donor variability and the operational challenges of scaling cell-based therapies. Supporting a clearer path to commercialization and reaching more patients. Explore Cell Sourcing Products and Services.
Innovative Solutions to Drive Your Allogeneic Cell Therapy Forward
-
Sartorius Ambr® 15 - Advanced Microbioreactor System
Stirred tank reactors like Sartorius’ Ambr® 15 are valuable in scaling up allogeneic cell therapy manufacturing. The Ambr® 15 is a multi-parallel bioreactor system that processes up to 48 parallel cultivations. It provides a high-throughput platform for selecting the best clones, media, and feeds, enabling efficient and cost-effective cell therapy manufacturing scale-up. Moreover, the Ambr® 15 controls critical process parameters, including temperature, pH, and dissolved oxygen levels, ensuring consistent and reproducible outcomes. With the Ambr® 15, we can help you accelerate manufacturing, lower costs, and expedite the delivery of your cell therapies to market.
-
Cell Selection and Electroporation
Cell selection and electroporation are crucial for the targeted delivery of therapeutic genes to specific cells in allogeneic therapy, reducing the risk of off-target effects and improving safety and efficacy. We are continuously exploring and testing new cell selection and electroporation systems to enhance the efficacy and safety of allogeneic cell therapy and can provide you with the most cutting-edge solutions by staying at the forefront of technological advancements and innovations.
The Memphis facility is the first North American CDMO to be approved by the EMA to commercially manufacture an allogeneic cell therapy drug product.
Get the Details
Frequently Asked Questions (FAQs) About Allogeneic Cell Therapy Manufacturing
-
What is allogeneic vs. autologous manufacturing?
Allogeneic manufacturing involves using cells from a donor source to create cell therapies that can be given to multiple patients. In contrast, autologous manufacturing involves using a patient's own cells to develop personalized cell therapies for that individual. Allogeneic manufacturing has the potential for greater scalability and cost-effectiveness, while autologous manufacturing offers the advantage of customized treatment with a reduced risk of immune rejection.
-
What is the process for sourcing donor cells for allogeneic cell therapy?
Sourcing donor cells for allogeneic cell therapy involves identifying a pool of suitable donors, screening them for safety and compatibility, and then collecting the cells for use in treatment. The donor cells must be carefully characterized to ensure they meet regulatory requirements and can be used safely in patients. The donor cells must also be collected to maintain viability and functionality for manufacturing the final cell therapy product. Once collected, the donor cells can be used as starting material for manufacturing, which typically involves expansion and modification of the cells before they are infused into the patient.