Confidently Navigate a Well-Managed Technology Transfer for Cell and Gene Therapies
As the field of cell therapy manufacturing continues to develop, the need for a systematic approach to technology transfer that keeps pace with the evolving landscape of cell and gene therapy (CGT) is more critical than ever. The complexities of the CGT market, combined with the challenge of adapting preclinical procedures to meet regulatory requirements, can lead to substantial program setbacks if not navigated properly.
To avoid delays, it’s essential to have a seamless handover of insights and methodologies from Research and Development (R&D) labs to manufacturing facilities. This technology transfer for CGTs is the cornerstone for scaling new cell therapies from a lab setting to commercial production, ultimately paving the way for treatments that improve or save lives.
Recognizing the importance of effective technology transfer, we provide focused project management to guide your innovations from concept to market. Acting as the crucial link between your novel discoveries and their real-world applications, we can help you transform your scientific contributions into viable commercial products.
Accelerate your cell therapy tech transfer to GMP with New Product Introduction
Without a robust new product introduction process, setbacks can occur in cell and gene therapy programs, increasing tech transfer timelines to GMP.
As a leading contract development and manufacturing organization (CDMO), we specialize in expediting the technology transfer of advanced therapy products, particularly in the realm of cell therapy manufacturing. Operating across 120 sites in 23 countries, we have contributed to 86% of FDA-approved therapeutics, including 93% of those approved for oncology.
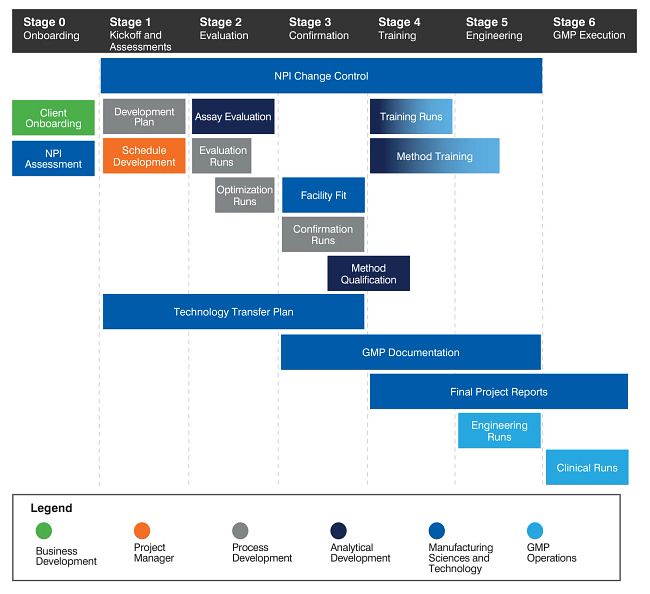
Technology Transfer Example
De-risking Cell Therapy New Product Introduction
Streamline your cell therapy program with a scalable, robust framework incorporating commercial readiness risk management strategies, standardized documentation, and integrated analytics.
De-risk Your Program
Cell Therapy Tech Transfer Process
Understanding your objectives
Ensuring a clear alignment with your objectives is important for successful tech transfer. Our cell therapy process and analytical development teams work closely with you to understand your project’s goals, processes, and products. By adhering to standardized protocols and rigorous risk management, we structure the transfer with a patient-centric vision, offering holistic solutions across all modalities. This methodology ensures a detailed evaluation of the technology transfer to GMP package, guaranteeing timely fulfillment of manufacturing requirements. An expert project manager will oversee coordination, monitor milestones and budgets, and maintain open communication among all stakeholders.
Streamlining data collection
To enhance the efficiency and accuracy of the technology transfer process, we utilize pre-designed document templates that serve as structured guides for each stage of the transfer. These templates allow our teams to consistently document key details, methodologies, and specifications across various phases and departments. It's crucial to move away from the notion that "the process is the product." Proper documentation eliminates the need for tribal knowledge, ensuring processes are scalable and transferable. This standardization not only streamlines data collection but also reduces the risk of errors, misinterpretations, and omissions. Additionally, the use of these templated documents speeds up the generation of reports and protocols, ensuring a smooth and efficient tech transfer process.
Providing a blueprint for your technology transfer package’s success
A detailed cell therapy technology transfer package serves as an essential roadmap, laying the groundwork for successful project execution. Similar to a blueprint guiding construction, this package directs the replication of critical knowledge. It covers everything from granular, step-by-step methodologies to quality standards and regulatory compliance, reducing ambiguities and errors at key checkpoints. Prioritizing capability over capacity, we ensure our development teams are equipped with the essential skills, experience, and technology platforms to de-risk tech transfer to GMP execution.
Facilitating manufacturing and scale-up
A robust technology transfer enhances both quality and safety, setting the stage for reliable and reproducible outcomes that minimize risks during the scale-up phase. We emphasize the importance of in-house analytics, helping to minimize delays and potential issues during GMP. This solid foundation for efficient, compliant manufacturing reduces the potential for expensive delays and builds credibility with internal teams as well as regulatory agencies. With a comprehensive technology transfer, the route to large-scale manufacturing of cell therapies is clear and achievable.
Partner with a CDMO offering expertise across the CGT landscape
-
Harness 75+ years of experience and a global network
When you join forces with a CDMO that understands every step of the CGT space, you get to enjoy an efficient and accurate cell therapy technology transfer to GMP. With over 75 years in the industry, we’ve built a well-established global network of cutting-edge CDMO facilities and testing sites with current GMP (CGMP) manufacturing capabilities across the CGT landscape. Elevate your tech transfer journey with a partner with a proven track record from early discovery to commercialization.
-
Fast-track your cell therapy technology transfer to market with proven commercial readiness
Benefit from our extensive experience in efficiently and effectively bringing cell and gene therapies to market. We’ve been involved in the development of 11 out of 32 US Food and Drug Administration (FDA)-approved CGT products, and more are in the pipeline. Notably, we recently achieved a milestone as the first North American CDMO to secure approval from the European Medicines Agency (EMA) for the commercial production of allogeneic cell therapy drug products at our Memphis facility.
Our continuous and systematic commercial readiness approach provides support from early research and discovery in your cell therapy product development lifecycle through manufacturing. Whether you’re starting at small-scale phase I CGMP manufacturing or are further in your product’s development phase, we have the capabilities to get you to commercial production.
-
Get regulatory guidance with in-house expertise
Boost your productivity by letting us take care of the regulatory matters for your cell and gene therapies. We provide Scientific and Regulatory Advisory Services for timely and accurate direction within the complex regulations landscape. Our regulatory guidance gives you access to the latest information about iPSC-derived therapies, CAR-T cells, and more. Having this service in-house offers clarity and actionable insights so you can make informed decisions about the tech transfer of your cell therapy solutions.
Frequently Asked Questions (FAQs) About Cell Therapy Technology Transfer
-
What is tech transfer?
Technology transfer, commonly known as tech transfer, is akin to passing the baton in a relay race of innovation. It’s the structured process by which scientific discoveries and technical know-how move from the laboratory (originating organization) to a new setting (receiving site) for further development and commercialization.
For a technology transfer in the CGT industry, the goal is to replicate a drug manufacturing process consistently, ensuring that the resulting therapeutic is both safe and effective.
-
What steps are involved in cell therapy technology transfer?
There are several steps involved in technology transfer:
- Invention is the creation of a new technology, process, or product.
- Invention disclosure is the formal sharing of the invention with an organization such as a technology transfer office or regulatory body.
- Evaluation occurs when the invention is assessed to determine its marketability and patentability.
- Protecting an invention is typically done by filing for a patent.
- Marketing an invention is done to attract licensing opportunities.
- Licensing is when the owner of the intellectual property grants permission to another party to use, produce, or commercialize the invention in exchange for agreed-upon terms.

-
How do you manage intellectual property during the transfer?
We manage and protect intellectual property (IP) with formal tech transfer agreements that outline rights and responsibilities for both parties while ensuring strict confidentiality.
-
What measures do you take to maintain the accuracy of the transferred technology?
To ensure accurate replication, we rigorously document and confirm the technology transfer through side-by-side comparisons and small-batch testing to maintain integrity during scaling.
-
What post-transfer support do you offer?
We provide ongoing support in the form of troubleshooting, additional training, and periodic review meetings to ensure the successful implementation and operation of the transferred technology.

