Efficient and Flexible Cell Therapy Platforms
Bringing a new drug to market can be fraught with challenges, which is why our cell therapy platforms offer a streamlined approach to process development while providing the flexibility and customization you need for your product and quality attributes.
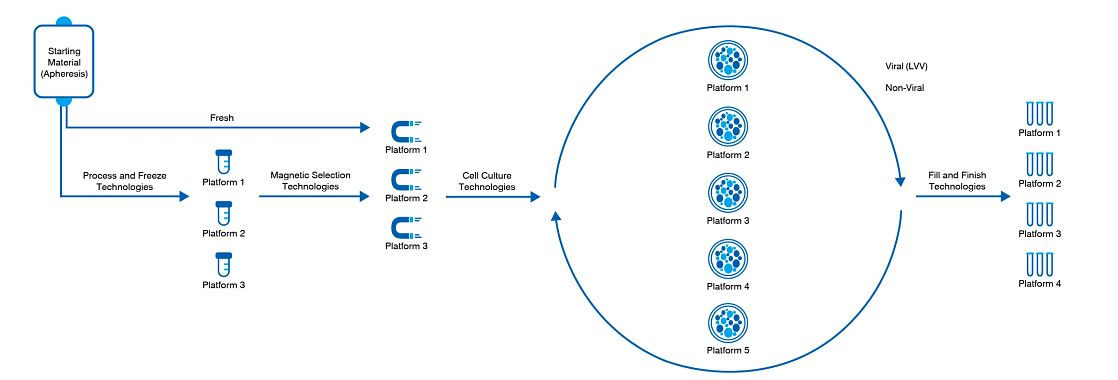
With our standardized processes, you can benefit from ready-to-use platforms that have been validated and developed for autologous CAR-T and TCR-T cell therapies. As an experienced contract development and manufacturing organization (CDMO) in cell and gene therapy, we have developed protocols that provide faster knowledge transfer and process crossover to reduce bottlenecks and speed development. These protocols range from cell selection and expansion to electroporation and viral vector transduction to wash/concentration and fill and finish methods.
CAR-T Platform Offerings Designed to Significantly Reduce Cost and Time in CAR-T Cell Therapy Process Development
Cell Therapy Flex Platform Advantage
When it comes to bringing a new drug therapy to market, errors and unplanned setbacks can be costly. Our CAR-T platforms incorporate strategic solutions to minimize risks, improve process robustness, and reduce costs in capital investments and batch runs.
With automated and closed system manufacturing leveraged as appropriate, we built our cell therapy platforms with state-of-the-art equipment and modalities that can flex from small projects with short timelines to large batches that span several weeks.
Additionally, our inline analytics and process monitoring of pH, dissolved oxygen, cellular metabolites, and cell health and viability make real-time decisions and process optimization a reality through machine learning.
This streamlined approach takes variability out of the equation and instead identifies and digitally captures processes that fit your specific drug development needs. With our tech transfer to new product introduction, we can then accurately repeat that process for lab and site transfers for cell therapy manufacturing.
Looking for support for your upcoming cell therapy manufacturing program?
Cell Therapy Flex Platform
Accelerate your CAR-T program from first run to IND with Charles River’s Cell Therapy Flex Platform.
Read the Case Study
“Cell therapy is transforming medicine, but the economics of manufacturing remain a major barrier. Our work with Charles River and the Flex platform allows us to tackle these challenges head-on, delivering healthier cells, faster processing times, and scalable separations that reduce complexity.”
Brandon McNaughton, CEO of Akadeum
Transform Drug Production with a CAR-T Platform
Our CAR-T platforms can be operationally customized depending on your needs. We have developed platforms for both CAR-T and TCR-T cell therapies for viral and non-viral vectors, including the gene-editing approach currently offered at our Memphis facility to manufacture cell therapies using CRISPR.
While the modality is already developed to streamline the drug development process, specific steps, such as genetic engineering, thawing, and filling, can be customized. As part of our intake, we look at your cell culture media, what reagent is needed, as well as different raw materials before selecting a series of cell therapy platforms that accommodate your needs.
Next-gen Immunotherapy: A Developer's Guide
Uncover the latest techniques to recognize, target, and eliminate cancer cells and tumors. Examine each cell type, plus autologous and allogeneic options, to better understand these evolving therapies.
Get the Insights
Advance Your Process with Cell Therapy Technology
Your innovation with new drug therapies continues to evolve, and so do we. At Charles River, we are constantly working to bring you CAR-T platform solutions that elevate and speed your commercial readiness. Learn more about our CDMO capabilities for CAR-T cell manufacturing.
In addition to our CAR-T platforms, we are working on validating other cell therapy platforms for NK cells, MSCs, and IPSCs to expand our cell therapy technology offerings in the future.