Rodent MRI as an In Vivo Imaging Tool
MRI is the medical application of nuclear magnetic resonance (NMR); the physical phenomenon where atomic nuclei absorb and re-emit electromagnetic radiation when exposed to a high magnetic field. The contrast between different tissues may be generated with varying radio wave pulses, based on the differences in the relaxation properties of atoms within the tissue. This technique has been refined over several decades and now rodent MRI is one the most specialized in vivo imaging techniques used in the preclinical assessment of drugs, enabling detailed pictures of internal organs and tissues, such as the brain.
Below, you’ll find descriptions of our MRI in vivo preclinical imaging capabilities and details of how these technologies can support your research and drug development.
Improving In Vivo Preclinical Imaging Speed with Artificial Intelligence
Preclinical neuroimaging, MRI in particular, is one of the most robust and objective readouts used for efficacy studies in animal models of CNS diseases. See how Charles River is utilizing AI to speed up the analysis of MRIs.
Read the Article
-
Anatomical MRI
Anatomical or structural MRI is used to study the shape, volume, integrity, and developmental changes in brain tissues and the blood brain barrier (BBB). Some of the applications for structural MRI include studying brain atrophy in neurodegenerative disease models, and ischemia or hemmorhagia in stroke models.
Our imaging team performs routine high-throughput measurement and longitudinal phenotyping studies. We also offer high resolution T2-weighted imaging. Ex vivo 3D anatomical imaging supports high resolution anatomical phenotyping and allows long scan times to maximize image resolution in complete 3D.
Figure 1: Fast spin-echo sequence, 12 min scan, 18 x 0.7 µm slices, 78 mm in plane.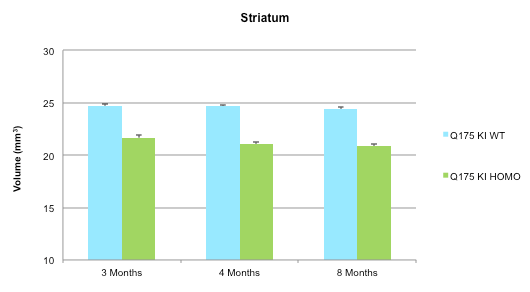
Figure 2: Increased atrophy in the striatum of a Huntington’s Disease homozygous mouse model compared to wildtype. -
Diffusion MRI
Diffusion-Weighted MR Imaging (DWI)
Diffusion-weighted MR imaging (DWI) is the gold standard to determine the age and severity of lesions in acute infarction both in clinical applications and in preclinical research. DWI can quantitatively assess the direct water movement due to diffusion and is utilized as a valuable tool in disease models closely related to altered tissue water environment such as stroke and traumatic brain injury. Additionally, DWI is used to monitor therapeutic response in brain tumors.
Diffusion Tension MRI
Diffusion tensor imaging enables the study of myelinated white matter in the brain. Water diffusion is anisotropic in highly organized cellular structures. By utilizing this, we are able to quantitatively characterize parameters such as fractional anisotropy and radial, axial, and mean diffusivity. These parameters allow investigation of demyelination in animal models, such as the cuprizone mouse model and LPC rat model of multiple sclerosis.
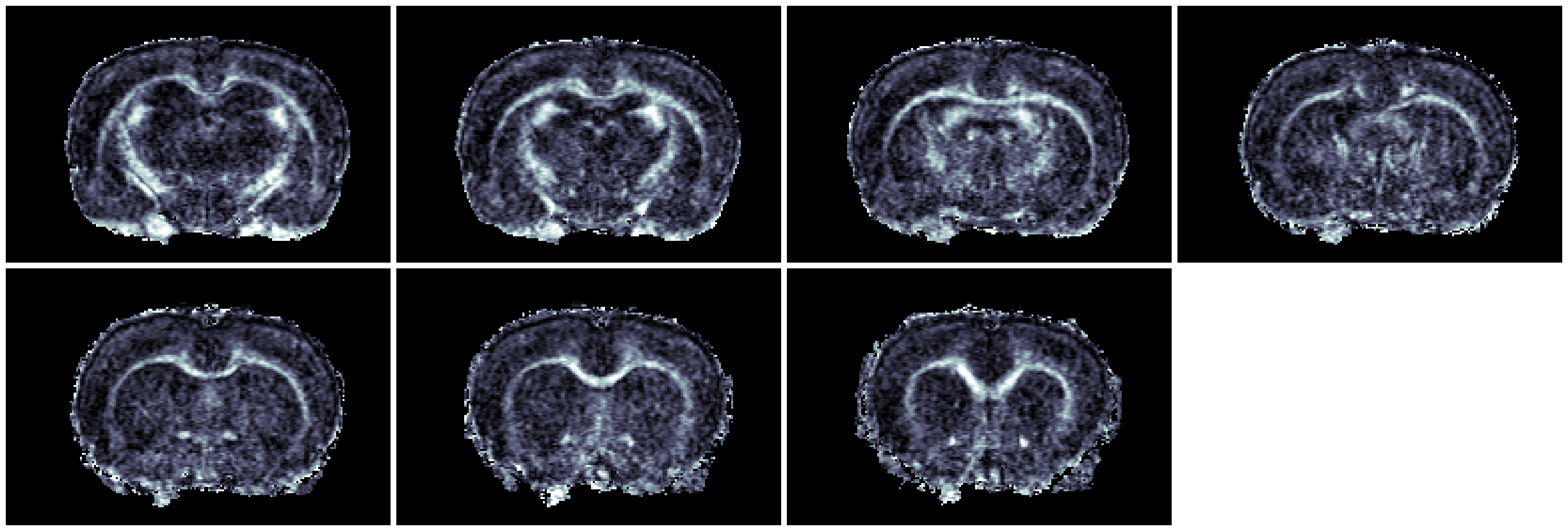
Figure 1: Fractional anisotropy map of 4-segment spin-echo diffusion tensor imaging of a lesion induced by unilateral injection of lysophospatidylcholine (LPC) into the corpus callosum. -
Functional and Pharmacological MRI
Functional MRI (fMRI) and pharmacological MRI (phMRI) are valuable tools to quantitatively measure changes in brain physiology and response to pharmacological modalities. Watch the video to view real-time measurement of seizure progression as an example of how phMRI can noninvasively measure changes in brain function.
Changes in brain tissue activity in response to stimuli, such as by a compound or sensory stimulus, can be quantitatively measured with MRI-based applications to provide evidence of neuronal engagement. Neurovascular activity and coupling (hemodynamic response to metabolic activity) may be detected utilizing blood oxygenation level-dependent (BOLD) effect in fMRI studies. Fast imaging sequences detect well-localized activations (e.g., at somatosensory cortex after forepaw stimulation). Cerebral activity following drug administration and CNS adaptation to medication may be detected using BOLD paradigm or relative cerebral blood volume (rCBV) via contrast enhancement methods, such as superparamagnetic iron oxide MION for MRI-based activity measurement. Results provide insight into brain physiology and neuropharmacological mechanisms.
phMRI is a technique that allows researchers to noninvasively map brain function in response to the hemodynamic changes brought on by the introduction of pharmaceutical stimuli. Analogous to fMRI but employing pharmacological methods of simulation, phMRI maps the time course and neurological response to specific pharmacological stimuli and gives insight into the pharmacokinetic and pharmacodynamic properties of drugs aimed at neurological, psychiatric, and neuromuscular diseases.
-
Hemodynamic MRI
Hemodynamic MRI comprises a broad range of modalities that may be utilized in any indication with a vascular component, including naïve animals, models of stroke or traumatic brain injury, neurodegenerative diseases like Alzheimer’s disease, hypoperfusion, and study of ventricular assist devices (VADs).
Charles River’s capabilities include perfusion imaging to measure relative cerebral blood volume (rCBV) and relative cerebral blood flow (rCBF). Imaging CBV uses contrast agents like monocrystalline iron oxide nanoparticles. This method generates gradient-echo and spin-echo sequences that are sensitive to different blood vessel diameters. CBF uses Gadolinium to track blood flow, blood volume, and mean transit time. Additionally, we offer time-of-flight magnetic resonance angiography (ToF-MRA) to detect vascular structure malformations using 2D and 3D imaging. This method requires the use of contrast agents.
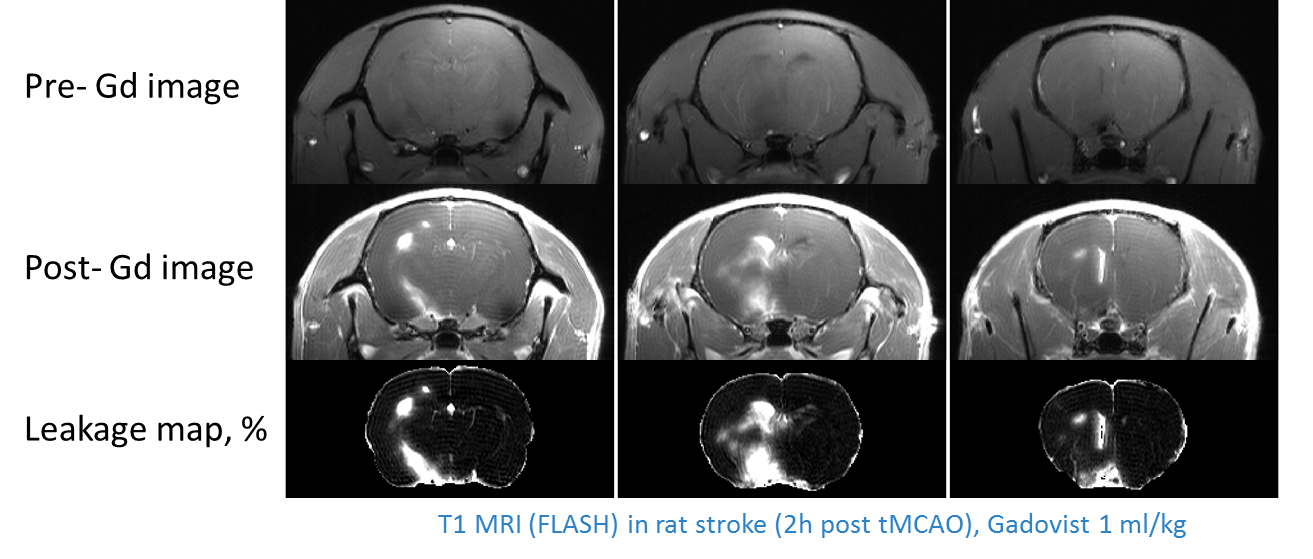
Figure 1: Gadolinium (Gd; Gadovist) contrast agent enhanced MRI used to detect blood brain barrier leakage via full brain mapping. When the blood brain barrier is compromised, the large Gadolinium molecule can leak into the brain. -
Proton Magnetic Resonance Spectroscopy (MRS)
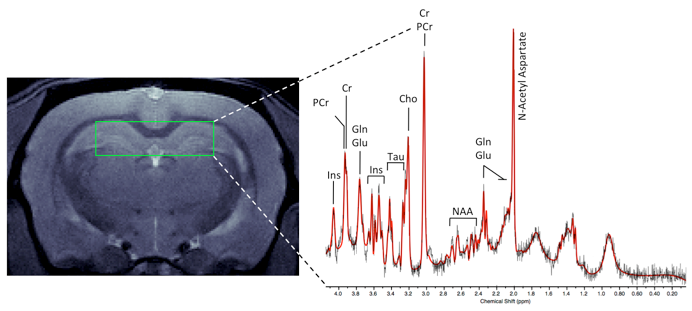
1H-Magnetic Resonance Spectroscopy (MRS) is used to quantitatively evaluate metabolic markers in localized brain structures such as striatum, hippocampus, or even in-brain tumors. This method allows longitudinal in vivo metabolic profiling of disease progression as well as response to drug therapies and is ideally suited for chronic long-term studies to comprehensively characterize disease rat and mouse models with multiple readouts and timepoints.
Charles River has implemented pulse sequences for localized 1H-MRS, enabling quantitative determination of 15-18 metabolite concentrations that are detected using LCModel software. Our imaging team also offers 31P MRS to detect changes in energy metabolism including ATP, phosphocreatine, and inorganic phosphate levels. As it is non-invasive, MRS enables collection of data from multiple time points prior to endpoint sampling, allowing for longitudinal metabolic biomarker analysis.
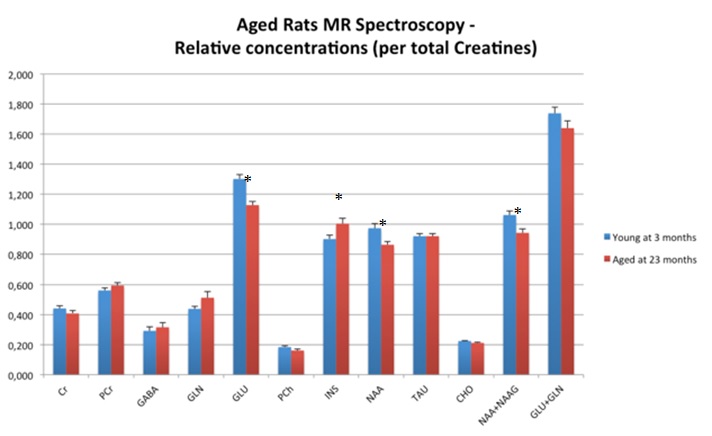
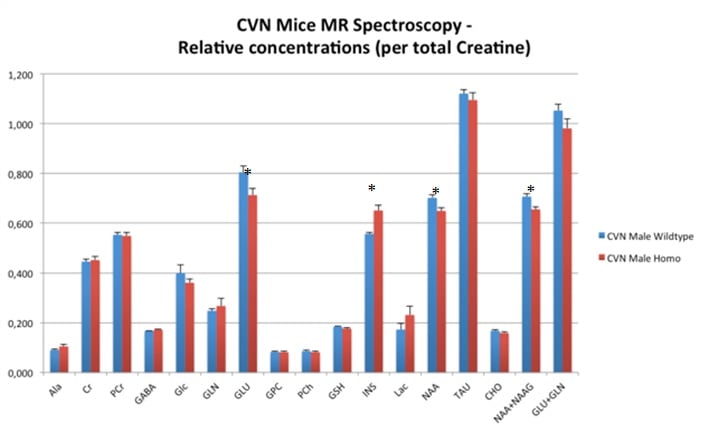
Image gallery: 1H-MRS in aged female Fisher rats and CVN Alzheimer’s disease mouse models. In both models, N-acetyl aspartate (NAA) and Glutamate (GLU) are decreased while myo-inositol (INS) is increased.
Using Preclinical MRI to Accelerate Discovery
Preclinical in vivo magnetic resonance imaging (MRI) can improve your efficiency and smooth the path from laboratory concept to commercial product. With more than 75 years of in-house collective expertise with MRI modalities, our team can help you to design and execute studies that meet your needs.
Our preclinical in vivo imaging experts who specialize in neuroscience research will help you find the right modality to address any of your specific questions – whether you seek data on mode of action, functional target engagement or something else – across all disease areas, e.g., acute neurology, neurodegeneration, demyelination, pain, seizures, and more.
We perform in vivo imaging with MRI using state-of-the-art systems and a plethora of models:
- Dedicated high field 11.7T in vivo imaging system
- Structural and functional imaging modalities
- Vast portfolio of neurological models
Frequently Asked Questions (FAQs) About MRI Preclinical In Vivo Imaging
-
How do healthcare costs relate to preclinical in vivo imaging using MRI?
Over a decade ago, both industry and FDA regulators recognized that a new approach, taking advantage of advances in preclinical in vivo imaging using MRI was needed to improve efficiency from in vivo pharmacology through drug development by recognizing the failure of molecules’ efficacy and safety earlier than Phase II clinical studies.
-
What species are you able to perform MRI on?
Preclinical in vivo imaging using MRI is performed on mice, rats, and large animals.
-
Are there resolution differences between brain and non-brain in vivo imaging using MRI?
While the measured organ, surrounding tissue environment, and imaging sequence can make a difference, it is possible to achieve high resolution brain and non-brain tissue MRI data. The brain does have the advantage of minimum motion that allows higher resolution compared to other organs.
-
Which software does Charles River use for brain volumetric analysis?
Currently, we use MATLAB-based software that was developed in-house and for brain volumetric analysis, the freely available AEDES software package is used.