Screen and Profile with iPSC-Derived Human Cells
Now available through our exclusive partnership with bit.bio, and its precise opti-ox cellular reprogramming technology, human iPSC-derived cell models become consistent, scalable and reproducible. Generated in large quantities and varied tissue types, bit.bio’s reprogrammed cell types are an excellent tool to support your high-throughput screening campaigns. Combined with our extensive experience in stem cell culturing and differentiation, you now have access to robust assay quality for your HTS and cell-based assays.
See how MEA electrophysiology can be used with primary cells, human iPSC-derived cells, and ex vivo brain slices across different stages of drug discovery in our on-demand webinar.
Watch Now
Robust HTS with the New Generation of iPSC-Derived Human Cells
Induced pluripotent stem cells (iPSCs) can be used to generate large numbers of cells of varied tissue types, making them an ideal vehicle for HTS and cell-based discovery screening. bit.bio, formerly ElpisBiomed, has applied deep learning algorithms to accelerate the discovery of methods for the reprogramming of every single cell type in the human body. Reprogrammed from patient-derived iPSCs, bit.bio‘s library of validated human cells delivers consistency, purity, scale, and speed to support robust HTS screening.
Our discovery clients now have a distinct advantage. Combining the physiological relevance of bit.bio’s reprogrammed iPSC-derived human cells with Charles River’s extensive experience with stem cell culturing and differentiation, high content imaging, and assay development, you receive reproducible cell populations and robust assay quality for your HTS and cell-based assays.
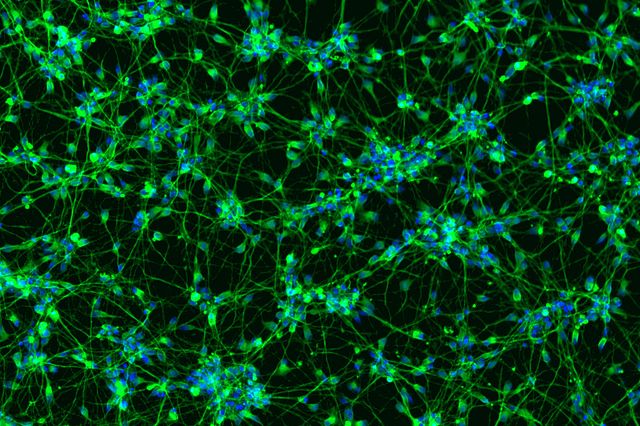
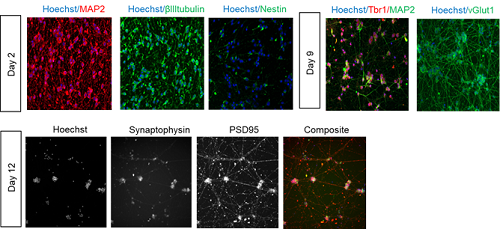
Case Study: bit.bio Rapid Differentiation into Functional Neurons
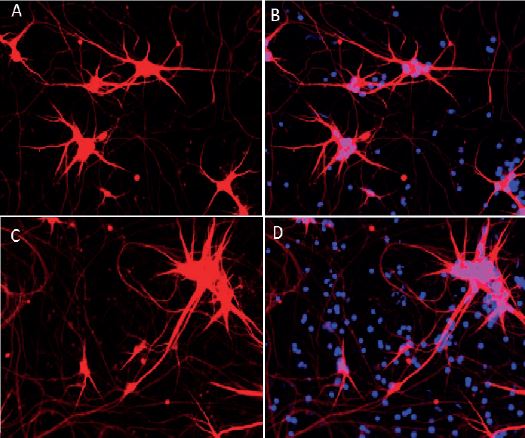
Within 12 days, bit.bio’s ioGlutamatergic Neurons convert into consistent, functional glutamatergic neurons. Cells exhibit neurite outgrowth and express numerous key neuronal markers, including Tbr1, MAP2, vGLUT1, synaptophysin and PSD95. ioGlutamatergic Neurons are programmed to rapidly mature upon revival in a 384-well plates without specialty differentiation media or protocols. This is unlike traditional methods which yield inconsistent numbers and purity of neurons, take over 30 days to achieve the same levels of maturity as the programmed ioGlutamatergic Neurons, and often cannot be performed in multi-well plates. Batch to batch reproducibility and homogeneity create a stable human model for excitatory neuronal activity and disease.
In addition to displaying protein markers consistent with neuronal differentiation, ioGlutamatergic Neurons form functional neural networks as measured by MEA after 2-3 weeks of maturation. This allows identification of compounds that show functional alteration of phenotypes relevant to diseased states. Before the bit.bio solution, this process was very difficult to establish at scale.
ioNEURONS/glut are Suitable for HTS Applications
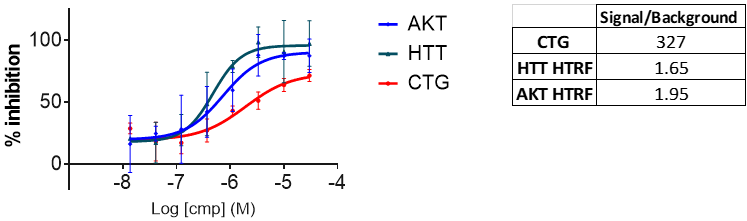
Displaying relevant markers for differentiation, ioGlutamatergic Neurons also differentiate in high density plates. This allows us to perform high-throughput screening in a physiologically-relevant cell type. Using an assay previously developed to identify the presence of huntingtin expressed at physiological levels, we were able to observe reproducible titrations of compounds shown to lower HTT. We were also able to perform functional follow-up assays, again on endogenous protein, to examine whether compounds were acting via toxicity or nonspecific protein degradation.
Robust and Scalable iPSC Cells for HTS
Learn more about the characterization of human iPSC-derived glutamatergic neurons.
Generating iPSC Cells for Disease-Relevant Cell-Based Assays
In another practical application of this technology, our Neuroscience Center of Excellence works with different suppliers to access patient-derived cells from neurological diseases such as Huntington's and amyotrophic lateral sclerosis to culture and differentiate them into neural cells for compound screening, target discovery, and validation. Using iPS stem cells, we offer high-throughput compatible assays with low variability and reproducible pharmacology observed in high-content and molecular biology approaches, including gene expression profiling for thorough quality control during culture and differentiation.
Stem cell-derived neurons are used in medium-throughput assays (e.g., hit compound deconvolution). We have developed arrays of high-content algorithms to allow measurement of cell morphology and toxicity in fixed-cell and real-time format and have electrophysiology capabilities for measurement of neuronal activity. Our CNS specialists have adopted specific stem cell differentiation protocols for production of other disease-relevant cells types to support other therapeutic areas of interest.
iPS Stem Cells for Neurological Diseases
Download this poster to see how disease relevant in vitro assays improve translation in vivo, leading to clinical success. Start your assays with ALS patient-derived iPS stem cells.
Frequently Asked Questions (FAQs) about iPSC Cells
-
What are stem cells?
Stem cells are unspecialized cells that have the potential to develop into many different cell types in the body. In research, these the cells can be induced to become specific cell types.
-
Why are stem cells important?
Stem cells are the building blocks of our body during development and are responsible for tissues homeostasis and regeneration during adulthood. Read a History of Neural Stem Cell Research and Its Clinical Application.
-
What are the different types of stem cells and their advantages?
Stem cells depend on their tissue of origin and differential potential. The three major types of stem cells are adult, embryonic, and induced pluripotent stem cells (iPSCs).
Adult stem cells are undifferentiated cells found among differentiated cells, in a tissue or in an organ. They can generate cells specific for the organ or tissue of origin; for this reason, they are defined as multipotent stem cells. Their major advantage is to maintain and repair the tissue or the organ where they are found. One example are neural stem cells found in the brain which can differentiate into nerve cells or glial cells.
hESCs are cells derived from an embryo at the earliest stages of development. As the least specialized stem cells, they can generate all cell types of the adult body. For this reason, they are defined as pluripotent. Pluripotent cells are a powerful tool to interrogate basic biological functions during early stages of cellular development.
Induced pluripotent stem cells (iPSC cells) are another form of pluripotent cells, derived from adult cells, (e.g., from fibroblasts or blood cells taken from a healthy donor or from a patient). The adult cell is genetically reprogrammed with specific genes to model embryonic stem cells, so they can then differentiate into many cell types. The main advantage of using induced pluripotent stem cells is that these cells carry the same genetic background of the adult cells where they originated, so they can be used to model a specific disease.
-
What drug discovery will benefit most from iPSC cells?
Drug discovery programs will benefit from the use of iPSC cells as those cells are able to generate unlimited numbers of cell types difficult to access, for example brain cells, to test new drugs and to identify targets.
-
Is stem cell research genetic engineering?
No. Stem cells can be maintained and differentiated without genetic manipulation. However, stem cells can be genetically engineered to modify or repair faulty genes that cause disease. Some research and discovery program may apply genetic engineering for the generation and differentiation of different stem cell types.
iPSCs for Research Use
We offer human wild-type induced pluripotent stem cells (iPSCs) as well as genetically modified iPSCs with the FailSafe® suicide gene system to support your research needs. Learn more