Discovery
|
Regina Kelder
Can Humanized Models Improve Huntington Translation?
How these advanced mouse models are being used to test small molecule splicing modulators designed to lower huntingtin protein. Plus, a closer look at gene therapies and ASOs for Huntington’s disease
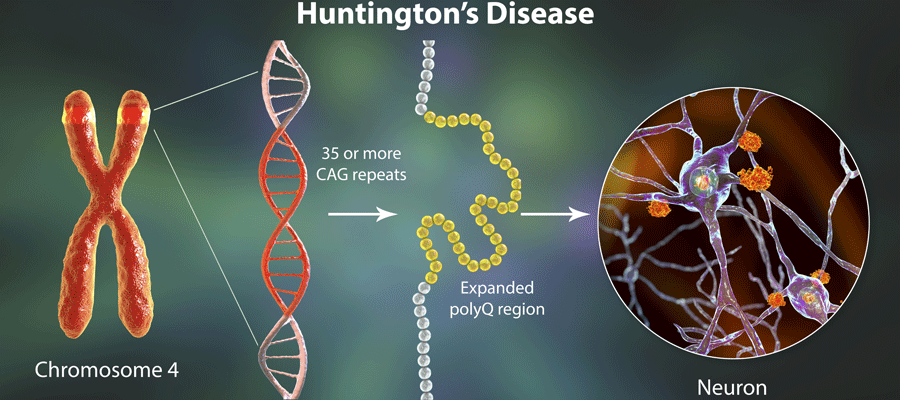
It’s difficult to imagine how a single genetic variant could be capable of causing so much neurological damage. But that’s what happens when someone inherits a mutation in the huntingtin or HTT gene found on chromosome 4. This anomaly puts people at extremely high risk of developing Huntington’s disease (HD), a neurodegenerative condition that leads to a range of devastating and worsening symptoms, from slurred speech and involuntary movement to mood swings and problems swallowing, speaking, and breathing. People with Huntington’s disease (HD) usually die within 20 years.
What causes the HTT mutation?
Everyone has a copy of the HTT gene, which encodes for the protein huntingtin. Near the beginning of the HD gene is a repetitive stretch of three letters – CAG. Normal HTT gene begins with 36 or fewer copies of this CAG repeat, but in people with the HTT mutation, there is an expanded repeat of CAGs, anywhere from 36 to over 100, that throws the gene’s function out of whack. The more copies a person has, the sooner symptoms will begin, and the more severe the physical and mental deterioration will be.
A major focus in Huntington’s disease research since the discovery of the HTT gene in 1993 has been on finding drugs that can lower the level of the harmful HTT protein, from antisense oligonucleotides and gene therapies to small molecule splicing modulators.
What are Splicing Modulators?
These small molecules are so-called because they interfere with a process call mRNA splicing. Under normal circumstances, mRNA splicing is a natural and beneficial process that removes introns—nucleotide sections within a gene sequence—that prevent the protein a gene codes for from getting formed correctly. But with HD patients the protein being formed by the mutated HTT gene is toxic, so you actually want to try and alter it.
Splicing modulators are kind of like the Spoiler Alert of molecular biology. They target the cellular machinery that processes these genetic messages, causing the messages to be read out of order and tipping off the cells what’s coming. With this burst of intel in hand, the cells ultimately destroy the message and do not make the toxic HTT protein.
After early preclinical studies showed that splicing modulators caused the levels of HTT RNA and huntingtin protein to drop exponentially depending upon the amount of drug given, the pipeline began filling up, with a few advancing to the clinic. However, last year the most promising and advanced candidate, developed by Novartis, was temporarily paused after early signs of peripheral neuropathy were seen in a few patients enrolled in the Phase II trial.
Researchers still don’t completely understand what triggered the adverse effects, but the good news is that a small early-stage clinical trial of a splicing modulator developed by PTC Therapeutics does show the drug appears to be well-tolerated. Interim data released in June 2023 by PTC also showed that huntingtin protein levels dropped in people receiving the modulator, and that the drop was greatest in people who received the highest dose. This trial is still ongoing, however.
Humanized Htt models to the rescue
Since splicing modulators only target human HTT pre-mRNA, scientists are turning from conventional large and small animal models to humanized models to try and improve the translation of efficacy and safety data to humans. CHDI, a large biomedical foundation whose sole focus is to rapidly develop new therapeutics that slow the progression of HD, has generated a humanized Htt mouse model that replaces one of the mouse (Htt) introns with the human (Htt) introns (which is directly targeted by the splicing modulators) in both the mutated and normal gene. “There are other HTT humanized models, but this model allows the targeting of the mouse Htt so that you can study on-target effects,” says Dario Magnani, PhD, a neuroscientist and Research Leader at Charles River’s Early Discovery lab in Saffron Walden, UK.
Charles River Laboratories, a CHDI partner, tested several splicing modulators in the humanized model with encouraging results, said Magnani. Initial findings showed a lowering of both normal and mutant HTT protein in a variety of tissues. Magnani said the ultimate goal of this ongoing project is to provide a better in vivo model suitable for preclinical safety and efficacy testing.
Indeed, the intronic region normally targeted by the splicing modulator has been replaced with the human sequence in a mouse model, which already contains the mutated native mouse huntingtin gene, rather than an additional human copy as more conventional models do. Thus, this model is representative of HD genetics and consistently displays associated motor symptoms and behavioral deficits, accompanied by brain atrophy and metabolite changes.
Magnani said using conventional animal models to test splicing modulators pose the challenge that you can’t test potential Htt on target toxic effects since the mouse gene can’t be targeted by the splicing modulators. This is particular important since the splicing modulators do not discriminate between the mutant and the normal HTT alleles.
Gene Therapy and ASOs
Two other major areas of research exploring ways of lowering HTT are gene therapies and antisense oligonucleotides (ASOs), which Charles River Discovery sites are supporting preclinically. Several small clinical trials by uniQure—developer of the world’s first gene therapy for Huntington’s disease—currently have a total of 40 patients enrolled in the US and Europe. The trials evaluated different doses of the gene therapy and different methods of administration.
The one-time gene therapy containing microRNA is packaged in a deactivated adeno-associated virus and administered into the brain. Once in cells, the miRNA is designed to recognize, bind to and non-selectively lower the human huntingtin protein in cells. Recent data released in 2023 included these findings:
- Using different behavioral rating scales, patients who received the gene therapy seemed to perform routine daily tasks better following treatment.
- The neurofilament light chain (NfL) spike—which is released when neurons degenerate and die—returned to normal within 18 months after surgery.
- In the low-dose group, HTT levels in spinal fluid dropped but rose in the high-dose group.
Based on these findings, uniQure plans to continue to enroll patients, and add an additional arm that will evaluate the gene therapy in combination with a steroid to try and reduce some of the side effects seen so far.
The role of advanced imaging in HD
Advanced imaging capabilities also signal a major shift in how we deliver gene therapies for some neurological disorders, including HD. Simon Authier, DVM, a Senior Director of Scientific Operations and Veterinary Science, said Charles River’s Laval site has completed more than 40 gene therapy studies in animals using intraoperative MRI. The technique is a more exact way of delivering viral vector-packaged gene therapies to different parts of the brain. The technique enhances drug delivery to desired brain regions while minimizing off-target side effects.
Authier said Charles River is also partnering with research centers to offer MRI-guided focused ultrasound developed by the company Insightec, to help treat neurological disorders like HD. The gene therapy can be administered intravenously, while the MRI-guided focused ultrasound transiently increases the permeability of the blood-brain barrier, allowing agents to cross it. “This really opens up therapies that were previously not conceivable,” says Authier.
The development of antisense oligonucleotides or ASOs for HD are also moving forward. In clinical development, Vico Therapeutics B.V recently announced the dosing of their first patient in a Phase I/2a trial using an ASO. This is the first ASO to reach the clinic that targets the CAG repeat in HD, as well spinocerebellar ataxia 1 and 2, the company said. These three conditions are all part of a family of diseases known as polyglutamine disorders, which are late-onset genetic disorders that lead to progressive neurodegenerative impairment.
Charles River will be presenting on its evaluation of the humanized model at the upcoming Society for Neuroscience Meeting Nov. 11-15. Check out all of our posters here.
