12
IND Submissions Supported
>100
RNA Scientists
~450
RNA Therapeutics Citations
ASO Drug Development
ASOs represent a highly specific and cutting-edge approach to treating diseases with unmet need and can address “undruggable” targets. They are short, synthetic single strands of nucleotides designed to bind specific mRNA sequences, enabling precision targeting of disease-related mutations. Several ASOs have been approved, and a large amount of research and drug discovery effort is currently dedicated to expanding their application into diseases with larger patient populations, including metabolic diseases and neurodegenerative disorders.
Expert Guidance for ASO Development Programs
Due to their mechanism of action and oligonucleotide structure, ASOs have unique challenges in drug discovery and development. We have developed comprehensive ASO development services and a deep understanding of the challenges faced by ASO drug developers.
Our RNA experts and Scientific Advisory Services (SAS) team support your ASO development with guidance and program design, reducing risk and ensuring a smoother path to the clinic.
Our teams can help:
- Provide guidance on the most appropriate ASO mechanism of action for your target and disease (e.g., RNase H recruitment, splicing modification, or miRNA targeting)
- Design screening cascades to evaluate ASO mechanism of action
- Select disease-appropriate cell and in vivo models for lead selection, efficacy, and preliminary toxicity studies
- Construct programs designed to reflect your clinical objectives
Comprehensive ASO Development Services
From target validation to IND-enabling studies, our comprehensive range of services, combined with the deep expertise of our scientists, ensures that your ASO drug development programs are optimized for success from the earliest stages.
ASO Design and Target Evaluation
Evaluating your target in the context of disease helps you select the most appropriate mechanism of ASO action such as RNA knockdown, splicing modulation, or upregulation of protein expression. Our ASO design algorithms consider RNA secondary structure, unspecific and toxic motifs, and ASO specificity. At this stage, ASO length, chemical modifications and delivery to the target cell should also be considered.
ASO Screening and Hit Identification
High-throughput ASO screening is conducted in 384-well assay formats to identify ASOs with efficacy and potency. Screening cascades are specifically designed to evaluate your ASO mechanism of action at the RNA and/or protein level and can involve between 20 and over 1000 oligonucleotide sequences, depending on the cell model and readout chosen. The target and disease also dictate the specific cell model to be used, selected from rodent primary cells, human cell lines, commercially available human iPSC-derived disease cells, and patient-derived cells.
ASO Efficacy Studies
Once lead ASOs have been selected, studies can be conducted in disease-relevant cell and in vivo models to demonstrate efficacy, investigate off-target effects, explore delivery mechanisms, assess tolerability, and identify translational biomarkers.
Our expert teams can advise on the most appropriate models and readouts for your program, considering disease, model phenotype, and translation into the clinical setting. We have a wide range of models for the most common areas for which ASOs are being developed, including neuroscience, cardiovascular and metabolic disorders, and rare genetic disorders.
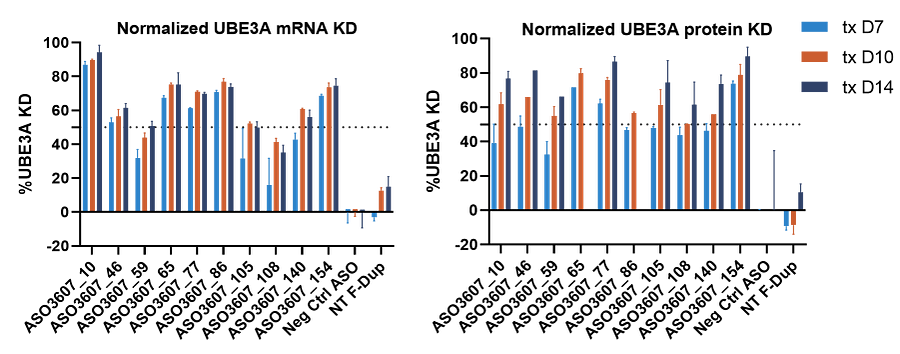
The following data shows the efficacy of a range of ASO sequences in patient-derived cells. Dup15q syndrome is a neurodevelopmental disorder caused by duplication of the Prader-Willi/Angelman critical region on chromosome 15. Expression of the ubiquitin-protein E3 ligase (UBE3A) is thought to cause most of the disease phenotype. A panel of ASOs was developed for a single Dup15q patient, and their effect on UBE3A gene expression and protein levels was examined in patient iPSC-derived glutamatergic neurons.
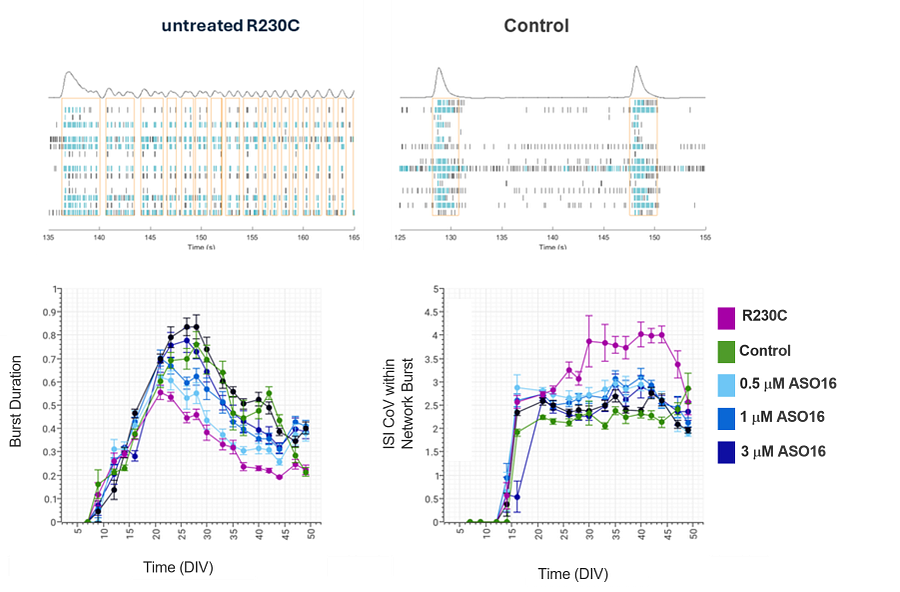
Phenotypic or functional assays in appropriate cell models are also crucial in demonstrating the efficacy of novel ASOs. Where “off-the-shelf” models are not available, for example for N=1 disorders, cell models can be generated from patient cells. A screening campaign was undertaken to identify a patient-specific ASO for a rare disease patient with R230C mutation in KNCQ3 (a subunit of voltage-gated potassium channels responsible for regulating neuronal activity). To test the efficacy of lead ASOs identified from a screening campaign, a co-culture of patient iPSC-derived glutamatergic and GABAergic neurons was established. Using MEA electrophysiology a disease phenotype was detected with R230C mutant neurons developing a seizure-like bursting pattern, shown by increased burst duration and high variation in inter-spike intervals (ISI) with each burst. ASO16, identified as a high-potency and low-immunotoxicity ASO from initial screening activities, reversed this increased burst duration and ISI variation in a dose-dependent manner in the patient-derived cell model.
In Vitro Toxicity
Early assessment of the toxicity potential of any lead ASO is vital to ensure that you proceed through drug discovery with the most suitable candidate. ASOs are known to induce a proinflammatory response through stimulation of toll-like receptors (TLRs), which can be assessed in human peripheral blood mononuclear cells (PBMCs).
Similarly, for ASOs intended to be delivered to the brain, it is key to assess neurotoxicity potential. For this purpose, our experts have developed mixed cultures of neurons, astrocytes and microglia, with neurotoxicity readouts including changes in biomarkers, morphological features, and functional readouts including neuronal activity measured by multi-electrode array.
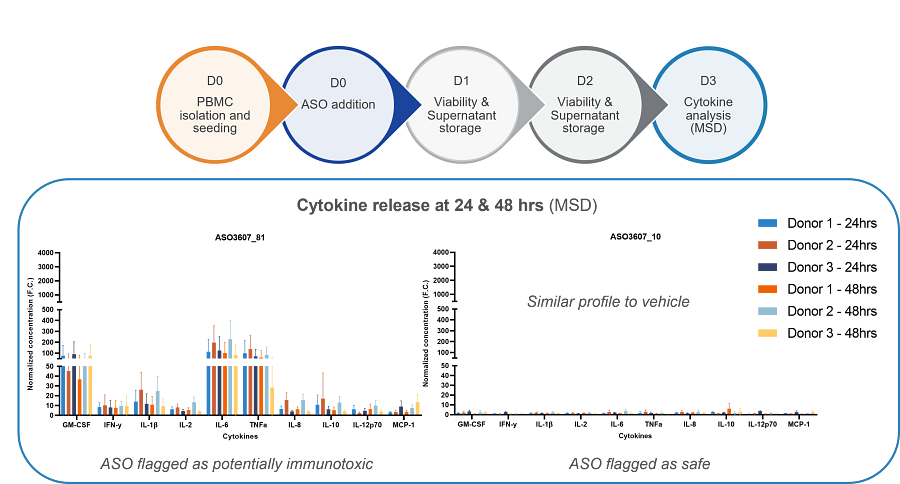
The example data above shows cytokine release profiles from PBMCs from three donors, measured by MSD, in response to a safe and immunotoxic ASO.
PK/PD and Bioanalysis
Assessment of ASO pharmacokinetics, metabolism, and distribution in vitro and in vivo minimizes compound failure and supports progression of an efficacious and safe drug into clinical trials. Multiple assay formats and technologies are available to investigate ASO properties, including in vitro ADME and drug transporter assays. In vivo pharmacokinetics and distribution can be assessed using microdialysis and subsequent detection of ASOs in dialysates, or by analysis of terminal tissue samples. Various bioanalytical platforms such as hybridization ELISAs or LC-MS can be used for ASO detection.
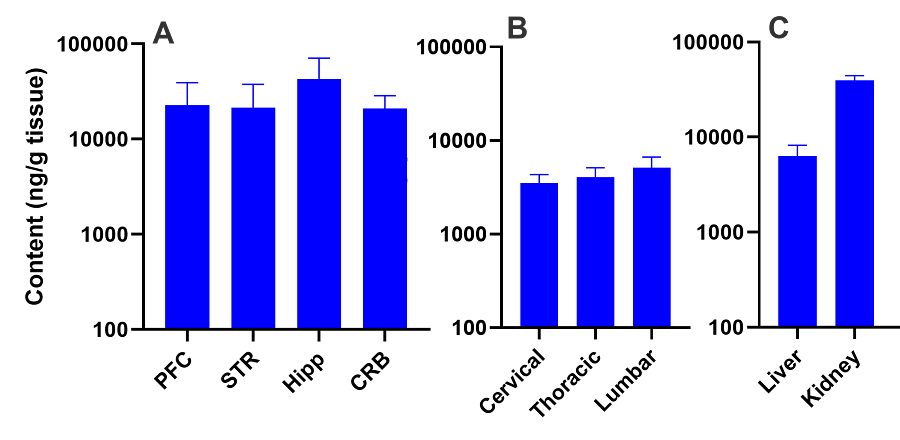
The data below show the detection of Nusinersen by LC-MS following intracerebrovascular administration, showing distribution through various areas of the brain (A), spinal cord (B) and peripheral tissues (C).
ASO Delivery
There are two aspects to ASO delivery – the chemical modification and/or packaging of ASOs to enhance stability and improved delivery to target cells, and the physical mechanisms and technologies used to deliver an ASO to its therapeutic site of action. Chemical modifications to improve stability, efficacy and cell penetration, and reduce degradation should be explored in the design and efficacy stages of ASO development.
The physical mechanism of ASO delivery to a patient generally depends on the indication and target tissue. For example, intravenous or subcutaneous injection can be used for systemic delivery to the kidneys or liver, or direct intravitreal injection may be used for direct delivery into the eye. For delivery to the central nervous system, intrathecal injection can be used, or there are various options for guided surgical delivery. Our teams can advise on the most appropriate and translatable delivery method for your ASO.
IND-Enabling Studies
Filing an investigational new drug (IND) application is a vital regulatory step in ASO development. Our RNA experts and SAS teams can support you in planning, launching, and completing IND-enabling studies, with nonclinical studies designed to reflect the objectives of your clinical program.
Charles River ASO Services
![]()
Expert Scientific Guidance
Advisory services and program design guidance.
Comprehensive Services
End-to-end services to support your ASO development
Strategic Guidance for ASO Development
Considered screening, efficacy and toxicity strategies can significantly accelerate and optimize ASO development timelines. Our on-demand webinar explains how a holistic approach supports an efficient path to get your therapy to patients quickly.
Watch On Demand
Frequently Asked Questions (FAQs) About ASO Development
-
How do ASOs work as therapeutics?
ASOs bind to mRNA sequences to alter RNA functions in various ways, with the aim of preventing disease-causing proteins from being produced, or to increase or restore the production of missing endogenous proteins. ASO binding can physically block the translation of mRNA to a protein to decrease the expression of mutated proteins. Binding can also direct cellular machinery to degrade mRNA to reduce protein production.
The majority of FDA-approved ASOs induce exon skipping by altering the splicing of pre-mRNA, leading to the translation of alternative or corrected splice variants that produce a fully functional or corrected protein. Unlike gene therapies, ASOs have a transient effect with no permanent changes to the genome.
-
What are some examples of FDA-approved antisense therapies?
ASOs have been approved by the FDA for the treatment of several disorders with genetic origin including Duchenne’s muscular dystrophy, spinal muscular atrophy and specific mutations causing amyotrophic lateral sclerosis (ALS).
For Duchenne’s muscular dystrophy, a muscular disorder caused by mutations or deletions in one of 79 exons of the dystrophin gene, exon skipping ASO have been approved, including Eteplirsen (exon 51 skipping), Golodirsen (exon 53 skipping), Viltolarsen (exon 53 skipping), and Casimersen (exon 45 skipping).
Other FDA-approved ASOs are:
- Nusinersen – Approved for the treatment of spinal muscular atrophy
- Inotersen – Approved for the treatment of familial hypercholesterolemia
- Tofersen – Approved for the treatment of SOD1 associated ALS
- Milasen – A novel patient-specific therapy approved for a specific individual with Batten’s disease
-
What are the key milestones in ASO development?
The science behind the use of ASOs as therapeutics stems from work in the 1970’s that showed that synthetic “antisense” oligonucleotides can block gene expression. From then modifications to ASOs were investigated to reduce their degradation by endogenous proteins and make them more suitable for therapeutic use. The first ASO approved by the FDA occurred in 1998, with the approval of Fomivirsen for cytomegalovirus retinitis. Second and third generation ASOs followed as advances were made in pharmacokinetics and stability of ASOs as drugs. Since the 2010s, several ASOs have been approved by the FDA, paving the way for future ASO approvals.