Research Models
|
Regina Kelder
Mighty Mouse Models: Becoming Potent Weapons in the Fight Against SARS-CoV-2
The evolution of mouse models for COVID-19 research; moving into the fast lane
Lab mice—most often of the species Mus musculus—have long been the mainstay of biomedical research. They are easy to handle and to breed, and their genomes are easily manipulated for genetic studies.
But when it comes to SARS-CoV-2 infections, mice have not, until recently, been the optimal choice. This is because the chief target of the virus, a protein receptor called angiotensin-converting enzyme 2 or ACE2 is significantly different in mice vs. humans. In the mouse, 11 of 29 amino acids of a critical domain on the virus’ surface differ from the human version, meaning the spike protein of the virus has significantly lower binding affinity. The net effect of this is lower SARS-CoV-2 infection susceptibility, meaning that mice can shrug off the virus while humans are dying of COVID-19 in droves.
In 2020, with a global pandemic taking hold, scientists turned to other animals, notably the LVG Golden Syrian hamster, ferrets, and some large animals, to learn about the mechanisms of SARS-CoV-2, but they also didn’t abandon the mouse. Using different genetic modification strategies, they made the virus more adaptable to mouse ACE2, and eventually generated humanized mice that contain human ACE2. The upshot: In addition, several mouse models for the study of mild and severe SARS-CoV-2 infections have been described or are under development, while a unique humanized mouse on a triple-immunodeficient background has the potential to be a gamechanger.
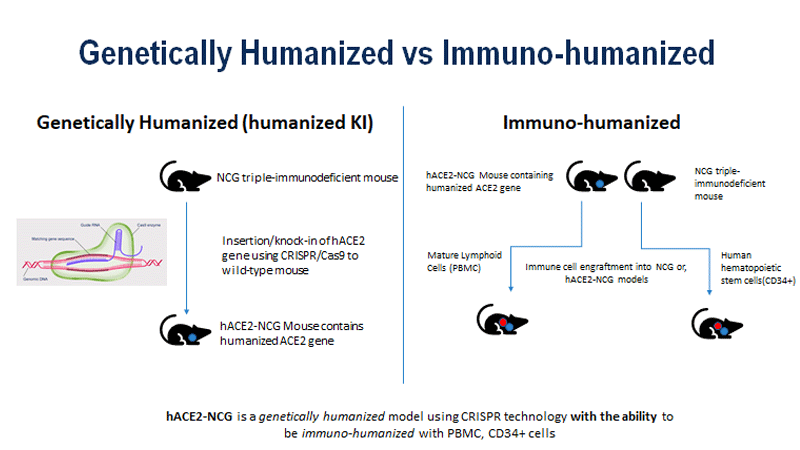
Charles River’s hACE2-NCG mouse model , a humanized knock-in model was developed specifically for COVID-19 research. It relies on CRISPR/Cas9 technology to introduce the hACE-2 gene into the designated position on an NCG background, thus increasing infection susceptibility. Through the engraftment of human cells, part of the immune system can be reconstituted prior to SARS-CoV-2 infection. The advantage of studying SARS-CoV-2 infections through human reconstitution could allow for the development of models used to study the mechanisms of different immune cells during infection.
Of importance, the novel, engineered hACE2-modified mouse model is the first and thus far only commercially available humanized ACE2 mouse model on a triple-immunodeficient background capable of being immuno-humanized for COVID-19 research.
Overall, mouse models have more than proven their value in the evaluation of vaccines and antiviral agents, disease pathogenesis as well as puzzling conditions like ARDS and long-COVID-19, a puzzling syndrome that afflicts some recovering patients. Below is a retrospective of some of the key studies that have led the way, including some exciting new developments on the humanization front.
Can SARS-CoV-2 Adapt to Mouse ACE2?
One of the early efforts by scientists to make mice better models for disease was to modify the spike protein of SARS-CoV-2 so that it binds more effectively to the mouse ACE2. One way they did this was by sequential passaging of SARS-CoV-2—a technique dating back to Louis Pasteur’s time that researchers use to force diseases to be transmitted between species. A team of scientists from Beijing found that the rapid adaption of SARS-CoV-2 in BALB/c mice enabled them to infect both aged and young wildtype BALB/c mice efficiently, resulting in moderate pneumonia and inflammatory responses. The findings were published in May 2020 in the journal Science. Meanwhile, a team of scientists from the University of North Carolina at Chapel Hill adapted the SARS-CoV-2 strain to infect mouse cells by using reverse genetics to modify the receptor binding domain—the key part of the virus’ spike protein that allows it to dock to the mouse’s body receptors, and gain entry to infect mouse cells via the mouse ACE2 protein. While the mice were sensitized for infection, they developed only mild disease, the team’s findings in Nature showed. The study was published in August 2020.
Can We Express Human ACE2 in Transgenic Mice?
Researchers have also tried, with some success, to engineer mice to express human ACE2. There are currently three transgenic mouse models that are susceptible to SARS-CoV-2, but because they express human ACE2 (hACE2) differently, they do not exhibit the same severity of disease; some are mild and some are lethal.
A recent 2020 study led by researchers at the Icahn School of Medicine at Mt. Sinai in New York compared one transgenic model modified to express hACE2 to a mouse model where the hACE2 is delivered directly to the mouse lung via a viral vector. The study, published in Emerging Microbes & Infections , found that the transgenic model replicated the virus to such high titers in the lung and brain that it killed the mouse. Interestingly, a 2007 study in the Journal of Virology, from the University of Iowa studying SARS-CoV—the virus that triggered the Severe Acute Respiratory Syndrome (SARS) global outbreak in 2003—showed similar findings.
The same transgenic mouse model—K18-hACE2—also showed that SARS-CoV-2 infection in the lungs of the mice cause severe inflammation and compromised respiratory function, and death in some, though brain infection was not evident. The findings from Washington University in St. Louis were published last year in Nature Immunology . A separate study carried out by researchers at the University of Iowa, the same K18-hACE2 mice developed severe lung disease and, in some cases, brain disease. Evidence of thrombosis and vasculitis were detected in the mice with severe pneumonia. The study was published in Nature in January 2021.
Researchers are also substituting mACE2—the mouse gene that expresses ACE2 protein—with the human ACE2 (hACE2) gene. This had been done successfully in the study of Middle East Respiratory Syndrome (MERS) – a cousin of SARS-CoV-2—and last year a team of scientists from the Beijing Institute of Microbiology and Epidemiology generated a mouse model expressing human ACE2 using CRISPR/Cas9 knock-in technology. In comparison to wild-type mice, the genetically modified mice demonstrated high viral loads in the lung, trachea and brain following intranasal infection. The findings were published last year in Host Cell Microbe .
Scientists have also attempted to sensitize the respiratory tracks of transgenic mice exposed to SARS-Cov-2 using adenovirus and other viruses that also expresshACE2. Research groups from the University of Iowa and University of North Carolina used this approach to rapidly generate a mouse model to study MERS , and found that the mice developed widespread infection of the lungs, and tissue changes consistent with viral pneumonia. Still, this delivery system did not achieve quite the same results in mice following SARS-Cov-2 exposure. Following transduction, the virus did not replicate as rapidly in the animals, a Journal of Experimental Medicine study found .
Will immuno-humanized mice be the optimal model for COVID-19 research?
In the study of infectious disease—whether its modeling pathogenesis or testing vaccines or treatments—the ideal scenario, and presumably the most translatable result, would be to use an animal model that mimics part of the human body which protects against infections. So, it’s not surprising that humanized mice are emerging as an important story in the COVID-19 fight.
Humanizing the murine immune system by taking human immune cells and engrafting into immunodeficient mouse models is not a new technology. Experiments in transplanting human tissue into mice began around three decades ago, and about 14 years ago researchers finally succeeded in creating mice that could successfully harbor human immune cells and be infected with human viruses that the mice shrug off.
Humanized mice have been worked with to study HIV and certain cancers and have been an important tool in studying the translatability of immuno-oncology drugs. Humanized models are broadly applicable to cancer cell lines common in the study of cancer oncology as well as patient-derived xenograft models (PDX models) where tissue or cells from a cancer patient are implanted into a humanized mouse.
But until recently, a commercially available humanized model didn’t even exist where you could study human immune system responses in SARS-CoV-2 infections. That began changing this past year.
A team from Columbia University has adapted its humanized model —which contains hematopoietic stem cells (HSCs) and thymus tissue in mice that lack immune systems of their own—for COVID-19 by introducing the human ACE2 receptor, plus a class I HLA molecule, into the lung epithelium of mice that receive human HSCs and pig or human thymus tissue.
Scientists at Yale University are adapting their 5-year-old MISTRG humanized mouse model—so named for the five human genes that replaced the mouse genes to mimic a human immune system—to study chronic diseases caused by SARS-CoV-2. A preprint of Yale study that came out this year described how their model provided a live view into the “aberrant macrophage response” that is thought to drive Acute Respiratory Disease Syndrome among those infected with SARS-CoV-2, a common condition in those critically ill with COVID-19.
And Chinese scientists have built a triple immunodeficient humanized mouse model that engrafts CD34+ cord blood and supports hematopoiesis of human immune cell types known to express ACE2. Charles River has been assessing the susceptibility of the HuNCG model to SARS-CoV-2 infection, with the model being viewed as a starting point that will hopefully provide a vehicle for labs around the world to study how the immune system and inflammatory responses of some COVID-19 patients inflicts devastating damage on a significant subset of patients. Having a model like this could also lead to a better understanding of SARS-CoV-2 infection and to the development of drugs for the treatment and prevention of the disease that the virus causes.
Humanized models begin with a strain of immunodeficient mice, but how “humanized they are” depends a great deal on how many human parts can replace the mouse parts of the immune system. The NCG mouse (NOD SCID-like CRISPR II2R Gamma mouse) is a triple immunodeficient mouse that lacks functional or mature T cells, B cells and natural killer cells, and reduced macrophage and dendritic cell function. They can be immuno-humanized using mature lymphoid cells (PBMC) or human hematopoietic stem cells (CD34+) cell engraftments. The model has applications in oncology, immunology, diabetes, regenerative medicine, and most recently COVID-19.
The Charles River hACE2-NCG mouse model , referenced earlier, uses an optimized genetic modification strategy that knocks in human ACE2 at the mouse ACE2 locus. This introduces the hACE2 gene into the designated position on the NCG background, to increase susceptibility to SARS-CoV-2 infection. In this model, the intracellular domain of the mouse contains ACE2 under the transcriptional regulation of endogenous sequences, designed to mimic the physiological expression pattern of ACE2 in various tissue types, including the kidney, lungs, and intestines. There are currently no other commercial models like this capable of being immuno-humanized for COVID-19 research ( see chart below for the difference between genetically humanized and immuno-humanized.)
“Humanized knock-in models, like our new hACE2-NCG mouse, represent the next generation of animal models enabling researchers to study human immune response more effectively after exposure to different viral variants”
Conclusion
Researchers have come a long way since the first cases of COVID-19 surfaced in China. So has the mouse model. Looking ahead, we will likely see refinements to all of these mouse models, given the current statistics: Close to 240 million cases and nearly 5 million dead. The mice are important now and will be when the next pandemic hits.
