DMD In Vitro Models and Assays
Our in vitro expertise in DMD and other muscular dystrophies focuses primarily on cell-based assays in human primary myotubes, iPSC-derived skeletal myocytes or cell lines, which are differentiated into myotubes using established protocols. These disease-relevant in vitro assays can be used for the profiling of activator/ inhibitor test articles such as small molecules, siRNA, shRNAs, and antisense oligonucleotides (ASOs), and include myoblast to myotube differentiation assays as well as assays for quantification of dystrophin, utrophin and creatine kinase.
Myoblast to Myotube Differentiation Assay
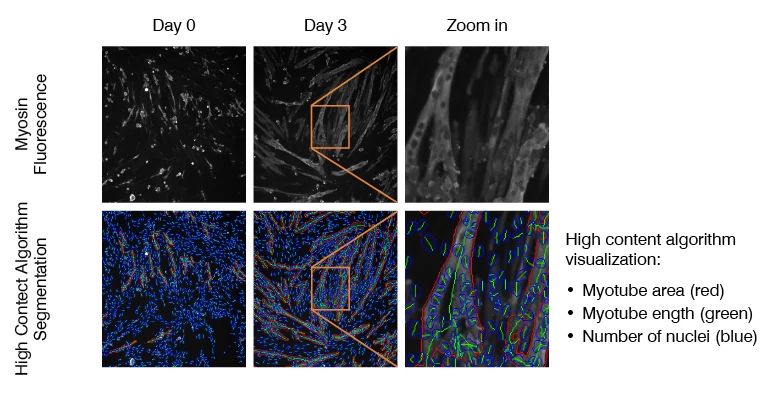
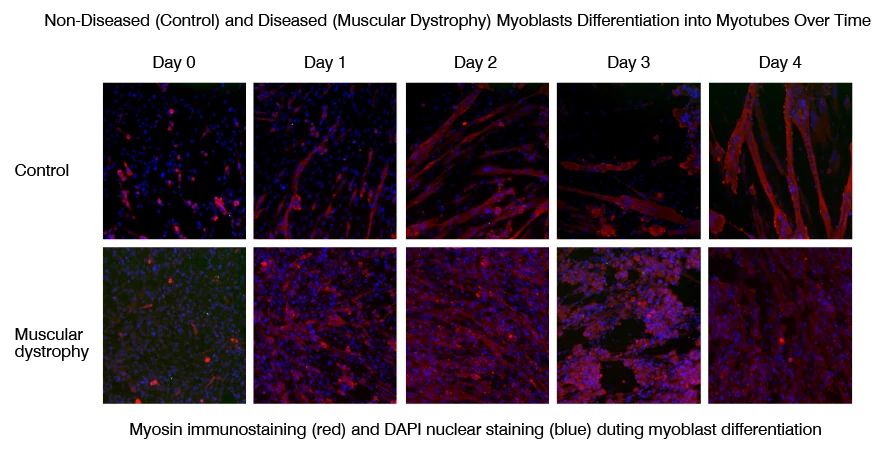
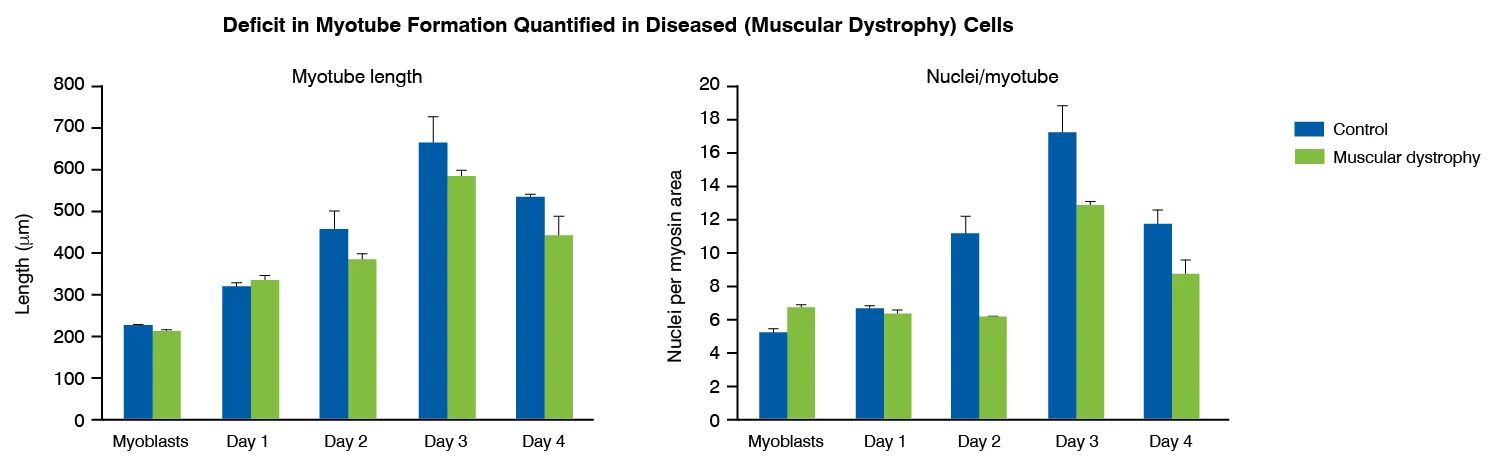
Myoblast to myotube differentiation assays uses high-content imaging to measure the modulation of myoblast differentiation by disease-relevant inflammatory stimuli, such as IL-6, IL-1 and TNF-α. This assay can be used to measure myoblast to myotube differentiation in healthy versus diseased (muscular dystrophy) skeletal muscle cells, and to assess potential phenotype restoration by therapeutics.
Dystrophin Quantification Assays
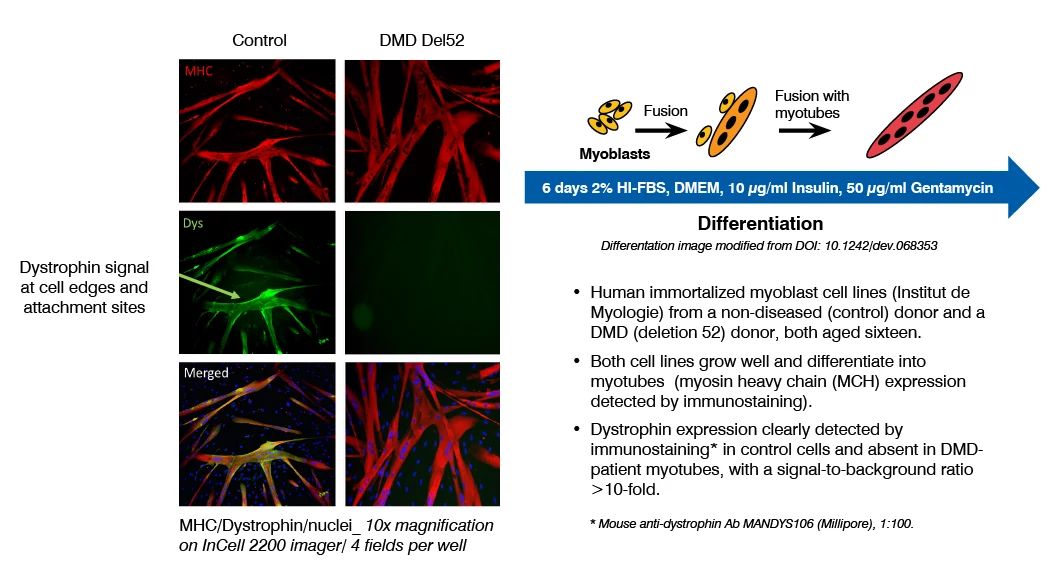
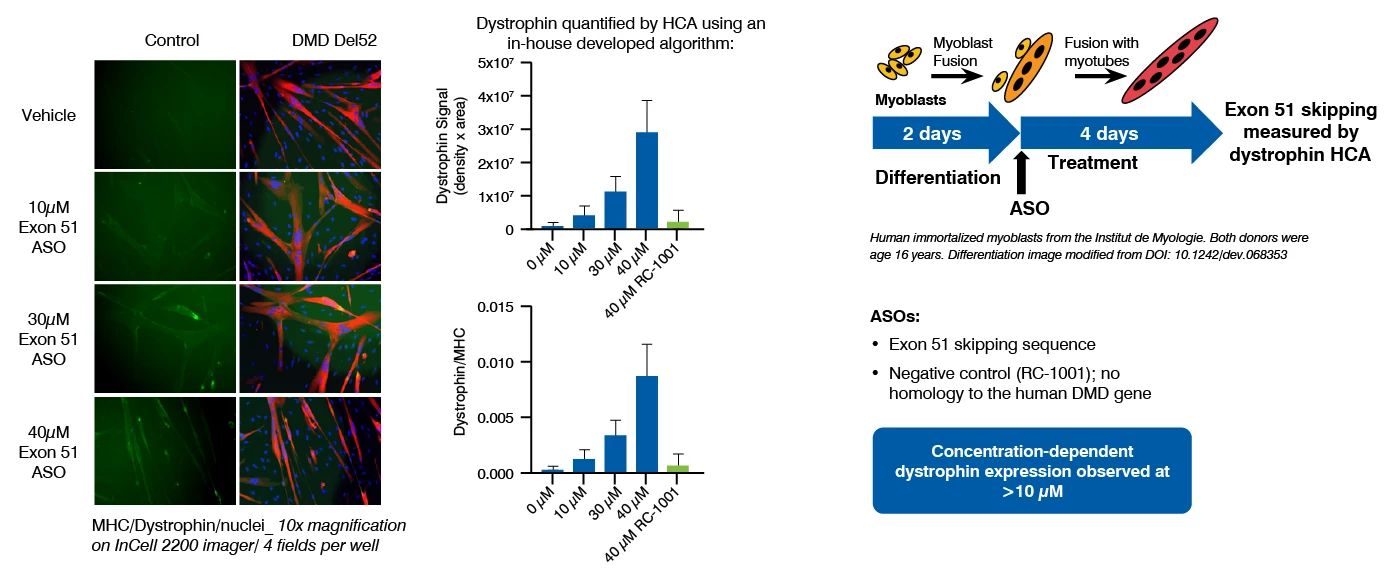
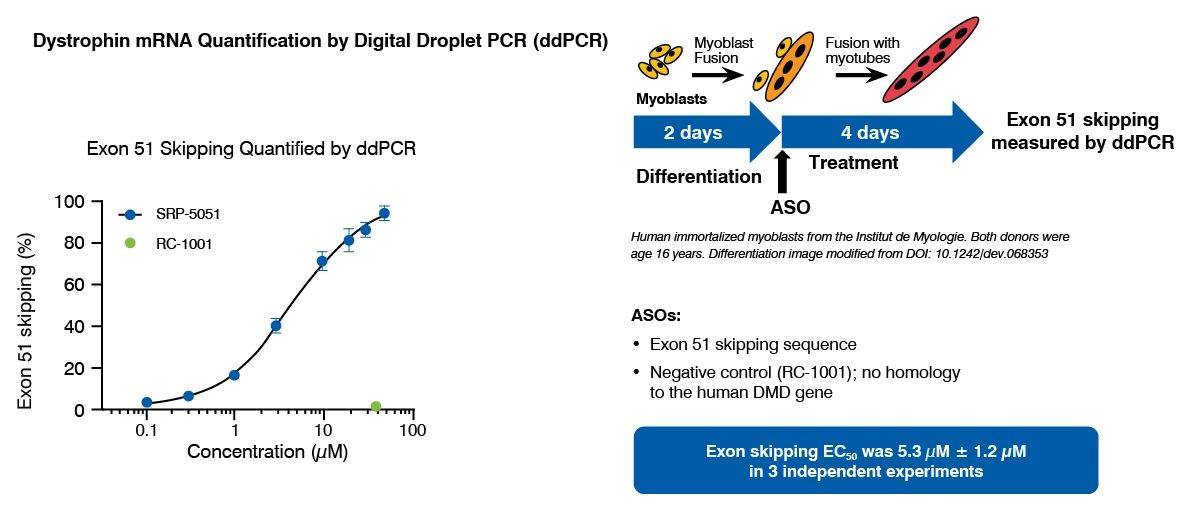
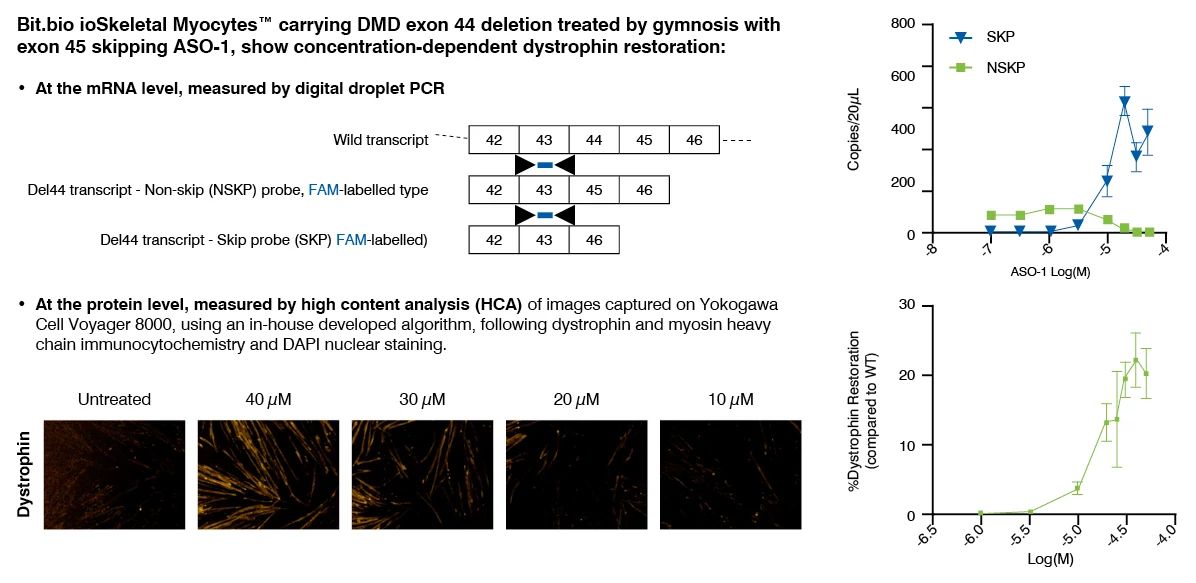
Dystrophin quantification in cell models is a valuable tool to characterize diseased versus non-diseased cells and measure dystrophin restoration in DMD cells, as potential therapeutic approach. At Charles River, we have established and validated this assay in human immortalized skeletal muscle cell lines (Institut de Myologie) and iPSC-derived skeletal myocytes (ioSkeletal Myocytes™ from bit.bio). This assay has been used to demonstrate the therapeutic potential of ASOs that induce exon 51 skipping.
-
Dystrophin Quantification Assay Principle and Data
Dystrophin can be quantified by immunostaining followed by high content analysis, or digital droplet PCR (ddPCR) for dystrophin mRNA quantification, following myoblast culture and differentiation.
This assay has been successfully used to demonstrate the restoration of dystrophin by ASOs that induce exon 51 skipping, in a human DMD (Del52) myoblast cell line and in DMD (Del44) Human iPSC-derived skeletal myocytes.
Creatine Kinase Quantification Assays
Creatine kinase (CK) is a well-established, translational serum biomarker for muscle injury in DMD, and has been shown to be elevated in DMD patients. This occurs due to the release of creatine kinase from damaged muscle fibers. Creatine kinase quantification assays can be used to assess the impact of therapeutics on creatine kinase in serum as a marker of muscle fiber damage.
-
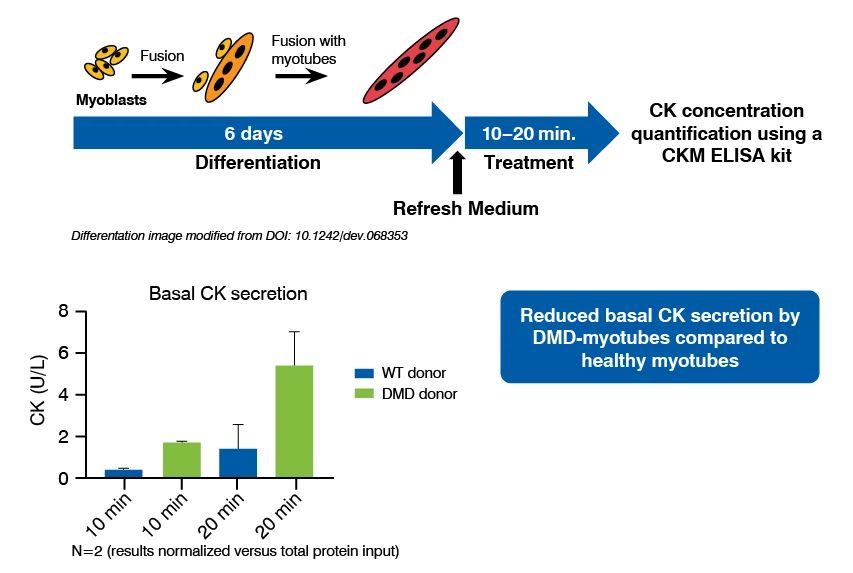
Creatine Kinase Quantification Assay Principle and Data
Creatine kinase quantification assays have been established in immortalized skeletal muscle cell lines originating from non-diseased and DMD age-matched donors. Following differentiation and treatment with investigative therapeutics creatine kinase is quantified in myotubes using a commercially available ELISA kit.
Utrophin Quantification Assays
Loss or failure to produce sufficient dystrophin is a key factor of muscle wastage in DMD patients. Utrophin, the fetal form of dystrophin, can act as a surrogate protein in the absence of dystrophin, as originally determined in DMD in vivo studies. Utrophin quantification assay can be used to identify therapeutics that upregulate utrophin expression in skeletal myotubes. As well as being used as a cellular assay for DMD therapeutics, utrophin quantification assay can be employed to examine utrophin levels in client-supplied samples.
-
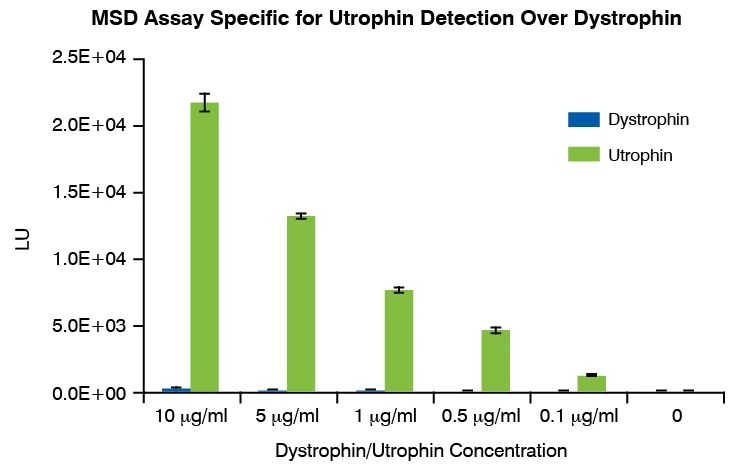
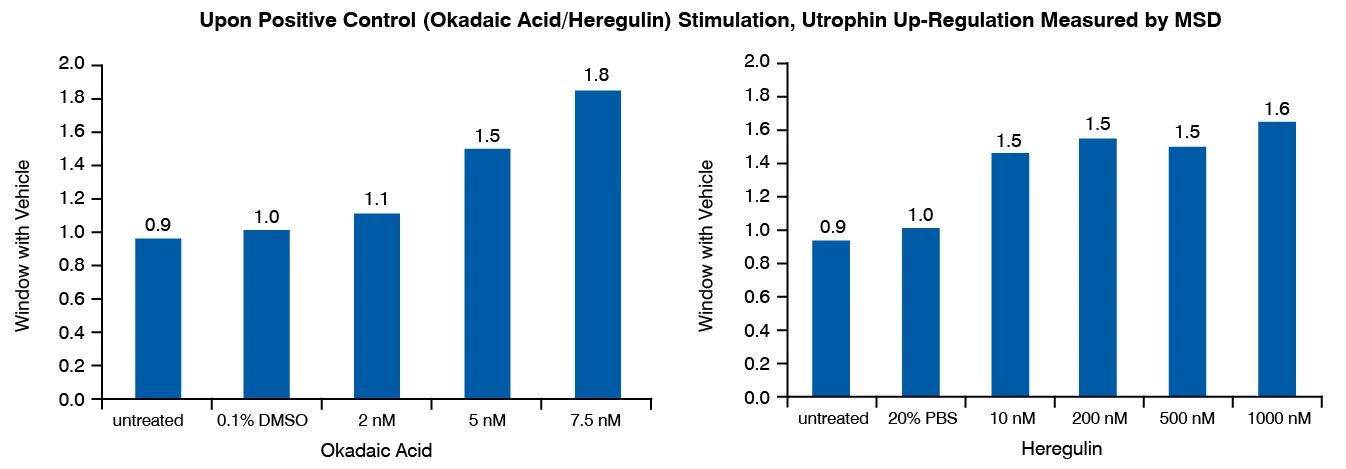
Utrophin Quantification Assay Principle and Data
Following incubation with a test therapeutic, utrophin protein is measured in cell lysates by MesoScale Discovery (MSD) immunoassay platform. The MSD assay is highly specific for utrophin over dystrophin. The model was developed in human primary skeletal myotubes (non-DMD); each new study will include a cell donor selection based upon upfront functional validation (out of up to three donors evaluated in parallel). This assay has been validated with heregulin, which has been shown to upregulate utrophin and improve the dystrophic phenotype of mdx mice.
We are currently working with key opinion leaders and clinical stage researchers in the muscular dystrophy field to expand our portfolio of in vitro assays and include analysis of other disease phenotypes (e.g., muscle breakdown, fibrosis) in patient-derived cells and human stem cells and to develop in vitro capabilities for other muscular dystrophies (e.g., DM1). This portfolio of in vitro assays complements our in vivo phenotyping and efficacy testing capabilities in the DMD MDX mouse model and other models of muscular dystrophy.