DMD Studies in MDX Mice
Duchenne muscular dystrophy (DMD) is a form of muscular dystrophy that occurs primarily in males and causes progressive weakness due to the atrophy of skeletal and heart muscles. While DMD is a rare disease, it is the most common form of muscular dystrophy with an incidence of 1 per 3,500 live male births. The first medication approved for DMD uses an exon-skipping strategy and is under continued evaluation. However, there remains an unmet clinical need for new therapies targeting Duchenne's and and other forms of muscular dystrophy.
We have extensively validated a well-known model for Duchenne muscular dystrophy studies, the X-linked muscular dystrophy (MDX) mouse. Validation endpoints include classic outputs like creatine kinase levels and histopathological analysis of muscle tissues as well as translational methods such as quantitative gait analysis using fine motor kinematics and changes in muscle structure and metabolism using multiple imaging modalities.
-
Duchenne Muscular Dystrophy Validation Data
The MDX mice show significant differences across various tested endpoints. For example, fine motor kinematic analysis showed strong alterations in lower body posture, which can be seen in increased hip, knee and ankle extensions, and also in increased overall hip height and decreased forelimb toe clearance compared to wild-type mice. The quantitative kinematic signature is confirmed by open field locomotor activity tests. Detailed imaging analysis also revealed significant changes in muscle structure, including muscle tissue damage at five weeks of age and fibrosis at 25 weeks of age. Additionally, the effect of exercise on muscle structure can be measured using MR imaging. This approach has been well documented. The baseline analysis of the mouse model for kyphosis and increased creatine kinase levels are used to confirm the disease phenotype.
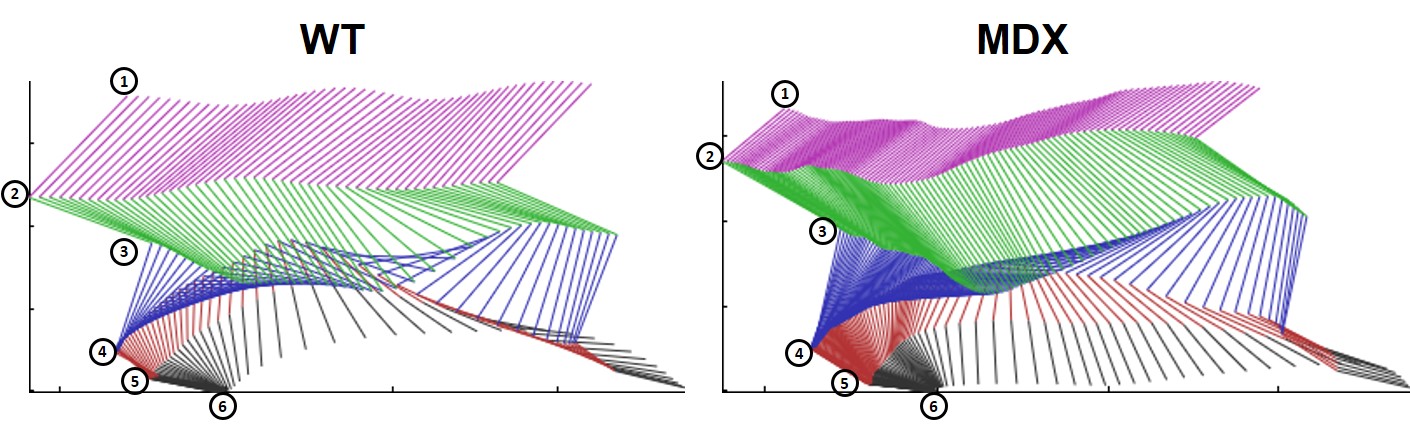
Figure 1
Fine motor kinematic analysis showed alterations in lower body posture, as seen in increased hip, knee and ankle extensions and hip height as well as decreased toe clearance in MDX mice compared to control mice. The different limb points represented are iliac crest (1), hip (2), knee (3), ankle (4), heel (5) and paw (6).
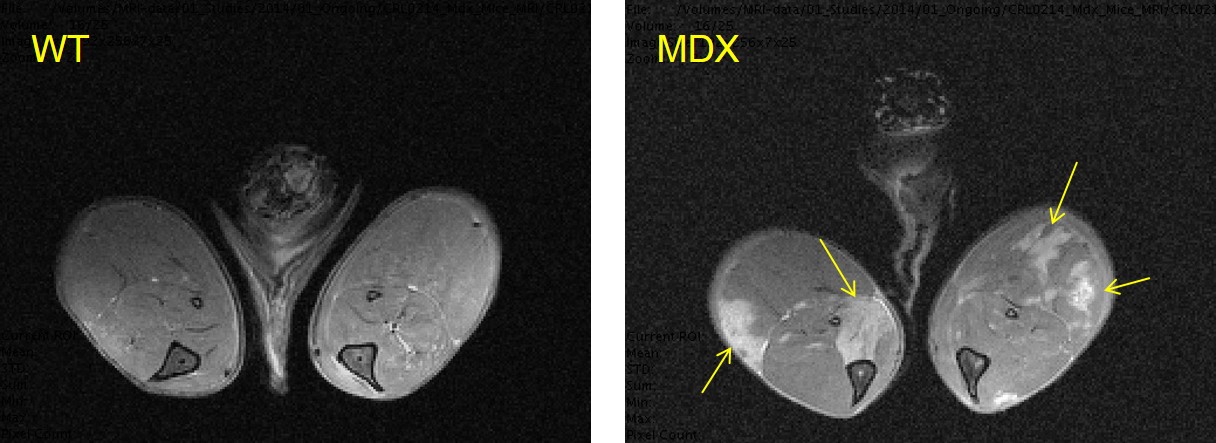
Figure 2
Muscle tissue damage due to oedema (marked with yellow arrows) in MDX mice at 55 weeks of age measured using T2-MRI mapping
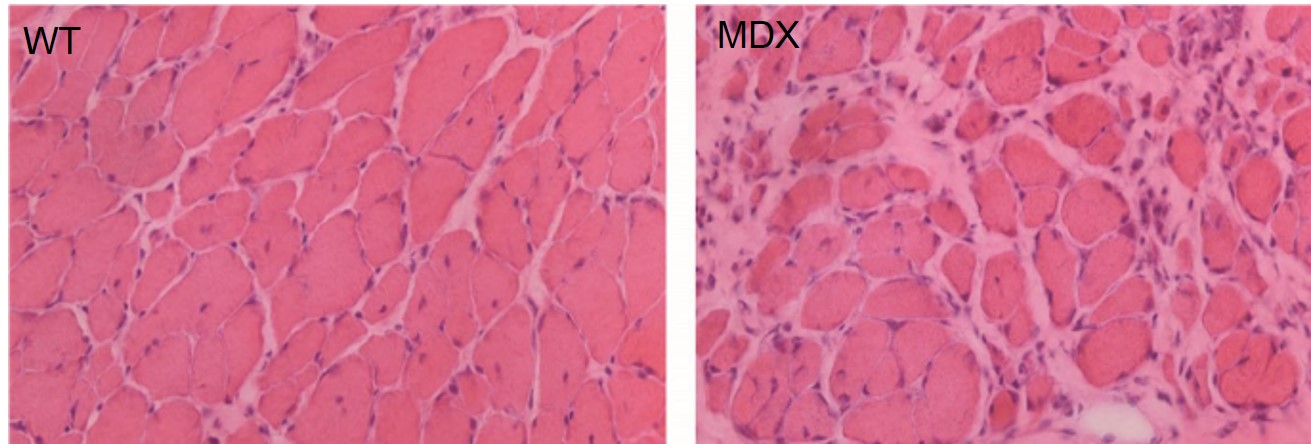
Figure 3
Histopathology of WT and MDX mice muscle showing less uniform muscle fibers with inflammation and clustered nuclei
Next-generation ASOs for Duchenne's Muscular Dystrophy
Learn how one company used a funnel-like antisense oligonucleotide (ASO) screening study, encompassing chemical modification screening, in vitro models, and in vivo studies to develop and profile next-generation ASOs.
Read the story
We offer an efficient platform for DMD services and candidate selection combined with noninvasive imaging modalities, robust sensory-motor behavior testing, and biochemical assays to comprehensively evaluate various therapeutic modalities for the nine known forms of muscular dystrophies. Our portfolio of tests for muscular dystrophy studies includes:
Behavioral testing to assess locomotion, balance, and posture
- Fine motor kinematic analysis
- Treadmill
- Open field
- Rotarod
- Grip strength
Imaging to assess neuromuscular degeneration and metabolic changes
- FDG PET imaging
- TSPO SPECT and PET imaging
- Kyphosis index measurement
- Muscle T2-MRI
- Magnetic resonance spectroscopy (MRS)
- Gadolinium-enhanced MRI
Frequently Asked Questions (FAQs) for Muscular Dystrophy Studies
-
How do you assess movement changes in the MDX mouse model for muscular dystrophy studies?
Movement is a key clinical endpoint in DMD pathophysiology and should be included in a efficacy study for therapies targeting muscular dystrophies. We gait and balance changes are measured using a quantitative translational method called fine motor kinematic analysis that analyzes high speed film using a proprietary data analysis package.
-
What is the MDX mouse model?
The X-linked muscular dystrophy (mdx) mouse model is used to study Duchenne muscular dystrophy. In this model, the dystrophin gene is truncated so the mouse does not express dystrophin, which is expressed in skeletal muscles, thus mimicking the DMD disease phenotype.