Hit Validation Via Flow Cytometry Analysis
Flow cytometry analysis is available for hit validation of results generated by Retrogenix® Cell Microarray Technology. A flow cytometry screen usually takes around 2-3 weeks and provides key additional data to support interpretation of the results from a full receptor identification, target deconvolution, or off-target screening.
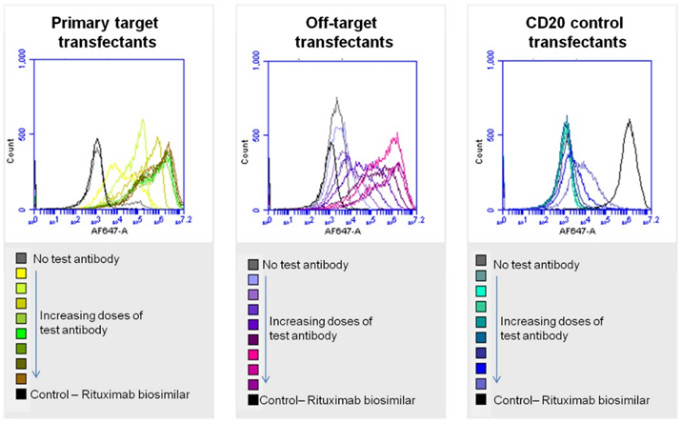
Expression vectors encoding the hits identified by cell microarray screening, along with appropriate control vectors, are used to transfect HEK293 cells. Live transfectants are then incubated with the test ligand and controls (and with any fluorescent detection antibodies that are required). Each set of transfectants is subjected to flow cytometry analysis and the results are interpreted by our scientists.
![]() Off-Target Screening
Off-Target Screening
Understand cross-reactivity to reduce toxicity-based attrition and aid lead selection.
![]() Target Deconvolution
Target Deconvolution
Accelerate phenotypic drug discovery through target deconvolution of phenotypic molecules.
![]() Receptor Identification
Receptor Identification
Uncover receptors for proteins, viruses, and more complex ligands such as whole cells.
![]() CAR Cell Specificity Screening
CAR Cell Specificity Screening
Safely utilize a patient’s own immune system by precisely recognizing specific tumors.
Dose Response Flow Cytometry
Results of titration experiments with increasing doses of the test ligand (compared with no-ligand or the positive control) showing the curve shift produced by varying degrees of ligand-receptor binding.
Hit validation via flow cytometry analysis of binding of a test antibody, an isotype-matched negative control antibody, and Rituximab biosimilar control to unfixed transfected cells expressing the known primary receptor, the putative off-target (identified by cell microarray screening), and a CD20 control.
WEBINAR: Maximize Safer, Targeted Biologic Development with Smarter NAMs-Based Off-Target Screening
This webinar showcases how the Retrogenix® platform empowers smarter, earlier decisions across biologic formats. You’ll also learn how this platform, recently accepted into the FDA’s ISTAND Pilot Program, aligns with evolving regulatory support for NAMs and the shift toward reduced animal use.
Watch the Replay
Cell Microarray Technology Case Study: Identification and validation of monoclonal antibody cross-reactivity
A single monoclonal test antibody – developed against a key GPCR target – was provided by the study sponsors along with an isotype-matched negative control antibody. An initial pre-screen was undertaken to determine the levels of background binding of the test antibody to untransfected HEK293 cells as well as its binding to cells over-expressing the known primary receptor. Binding was assessed using a well-validated AlexaFluor647-labelled anti-human IgG Fc detection antibody, followed by imaging for fluorescence. The pre-screen assessed the suitability of the test antibody for onward screening and helped Retrogenix® scientists to select the optimal dose to use throughout the study. Keep reading the case study...
eGuide for Off-Target Screening via the Retrogenix Platform
Download the eguide for all the information you need on how the Retrogenix platform can be leveraged to screen for specificity in vitro against a physiologically relevant human protein library from early-phase discovery onwards, to aid candidate selection, downstream study design, and IND approval.
Download now
Frequently Asked Questions (FAQs) for Hit Validation via Flow Cytometry
-
Can I request a follow-on validation study after results have been delivered?
Absolutely. We can typically use materials which are already available to us at the end of your main study to carry out validation, negating the need for you to produce and ship further samples to our High Peak Facility.
-
What further analyses can be performed by the wider CRL pipeline, following the Cell Microarray?
Many services across CRL complement the platform. For example, Retrogenix® Cell Microarray Technology, hit validation and specificity analysis can be combined with IHC-based Tissue Cross Reactivity (TCR) capabilities to provide a powerful dataset for IND submissions. The functionality of potential off-targets found with CAR cells on the platform can also be tested at Charles River.
-
Is additional validation always available?
Dependent on the study type, we have built in optional stages for hit validation interactions found with your test article, and to provide additional data points (particularly important for IND submissions. In our most in-depth study types, any hits found with your test molecule will be retested via the platform using both live and fixed cells, and you can even provide controls to further test the specificity of these interactions. In addition, specific hits will also be tested by flow cytometry.