Cell Microarray Data in Regulatory IND Submissions
To help our team and our clients better understand how our cell microarray data can support BLA and IND submissions, we carry out an ongoing feedback process to follow up on specificity screening studies and find out how those molecules are progressing. We received responses from over 80 clients surveyed. Many of our sponsors were unable to disclose, and therefore, these numbers represent a sample of the true dataset.
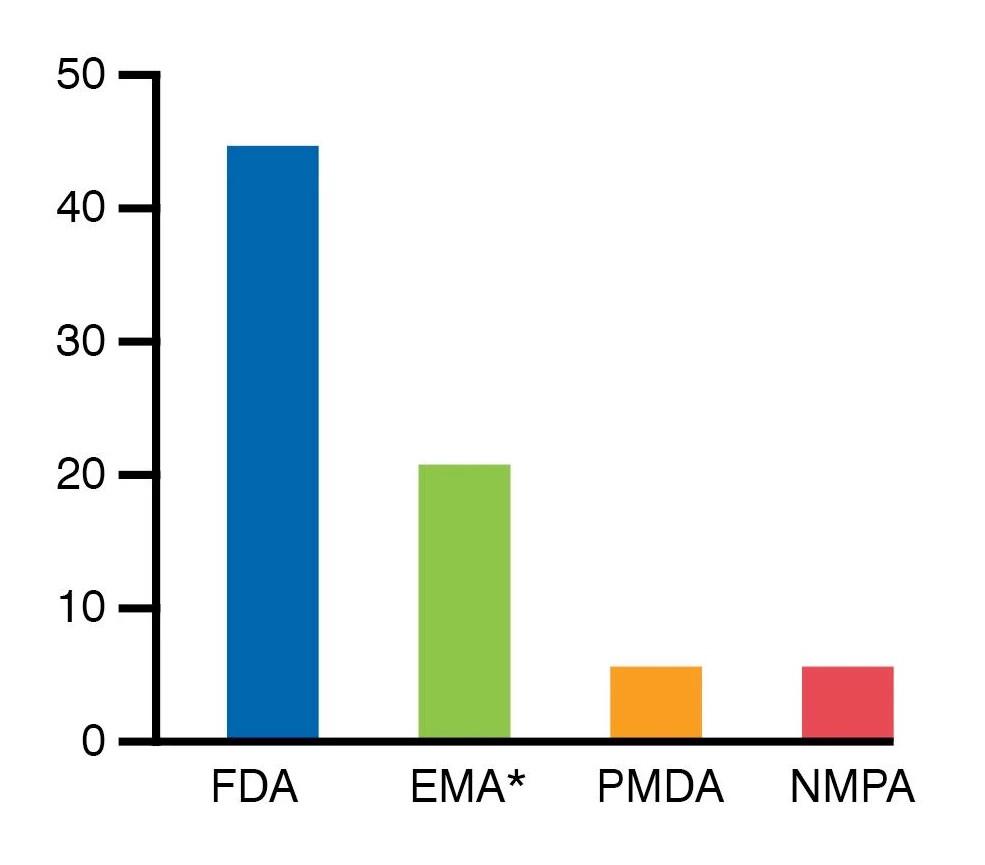
Overall IND Submissions
Retrogenix® IND-enabling specificity data have been extensively used in at least 78 IND submissions to major regulators around the world and other European regulatory agencies.
WEBINAR: Maximize Safer, Targeted Biologic Development with Smarter NAMs-Based Off-Target Screening
This webinar showcases how the Retrogenix® platform empowers smarter, earlier decisions across biologic formats. You’ll also learn how this platform, recently accepted into the FDA’s ISTAND Pilot Program, aligns with evolving regulatory support for NAMs and the shift toward reduced animal use.
Watch the Replay
IND Submission Success Rate
Across the board, for all IND submissions that used our generated data, 95% of submissions were approved, with 5% of the applications still in the process of submission.
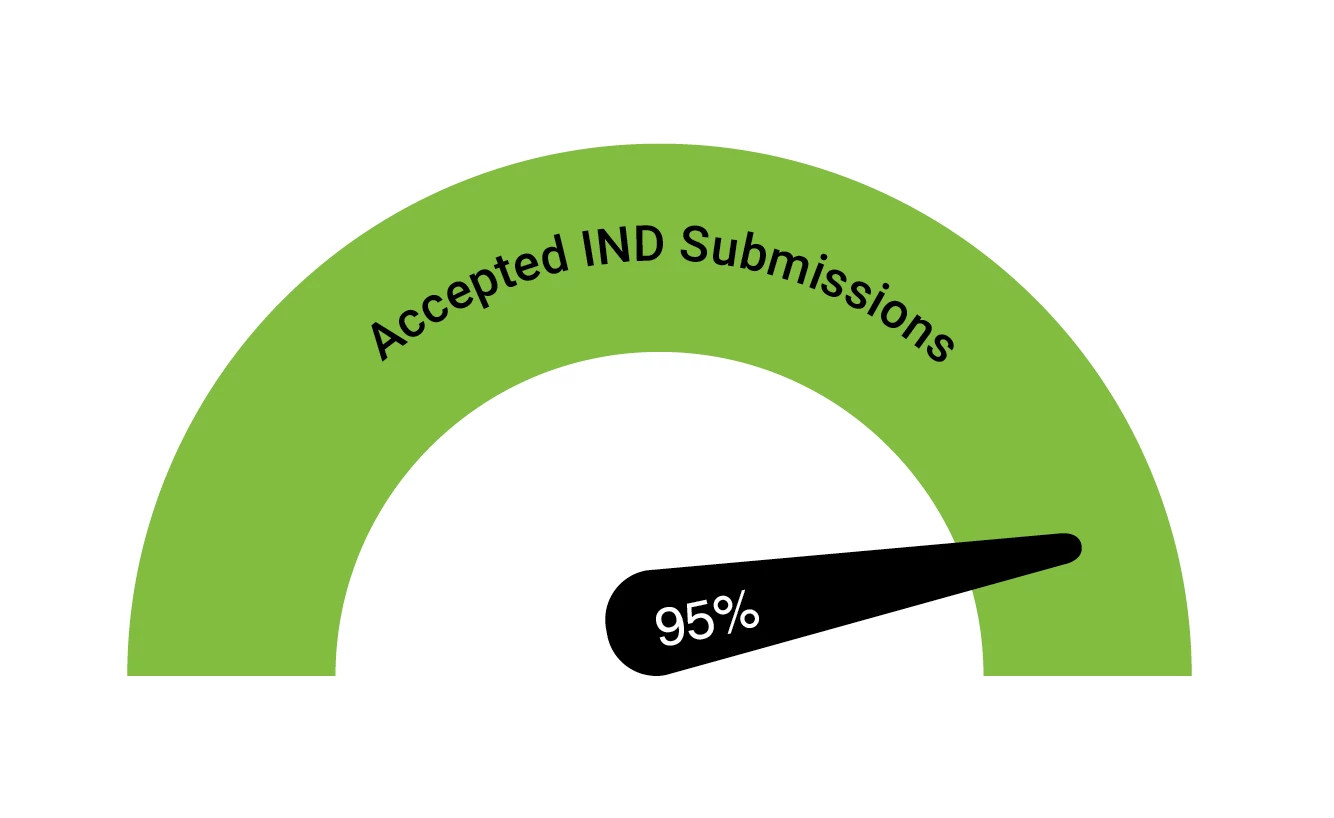
Accepted IND Submissions
95% of IND submissions containing Retrogenix® cell microarray data have been accepted.
Cross-Reactivity Screening Methods
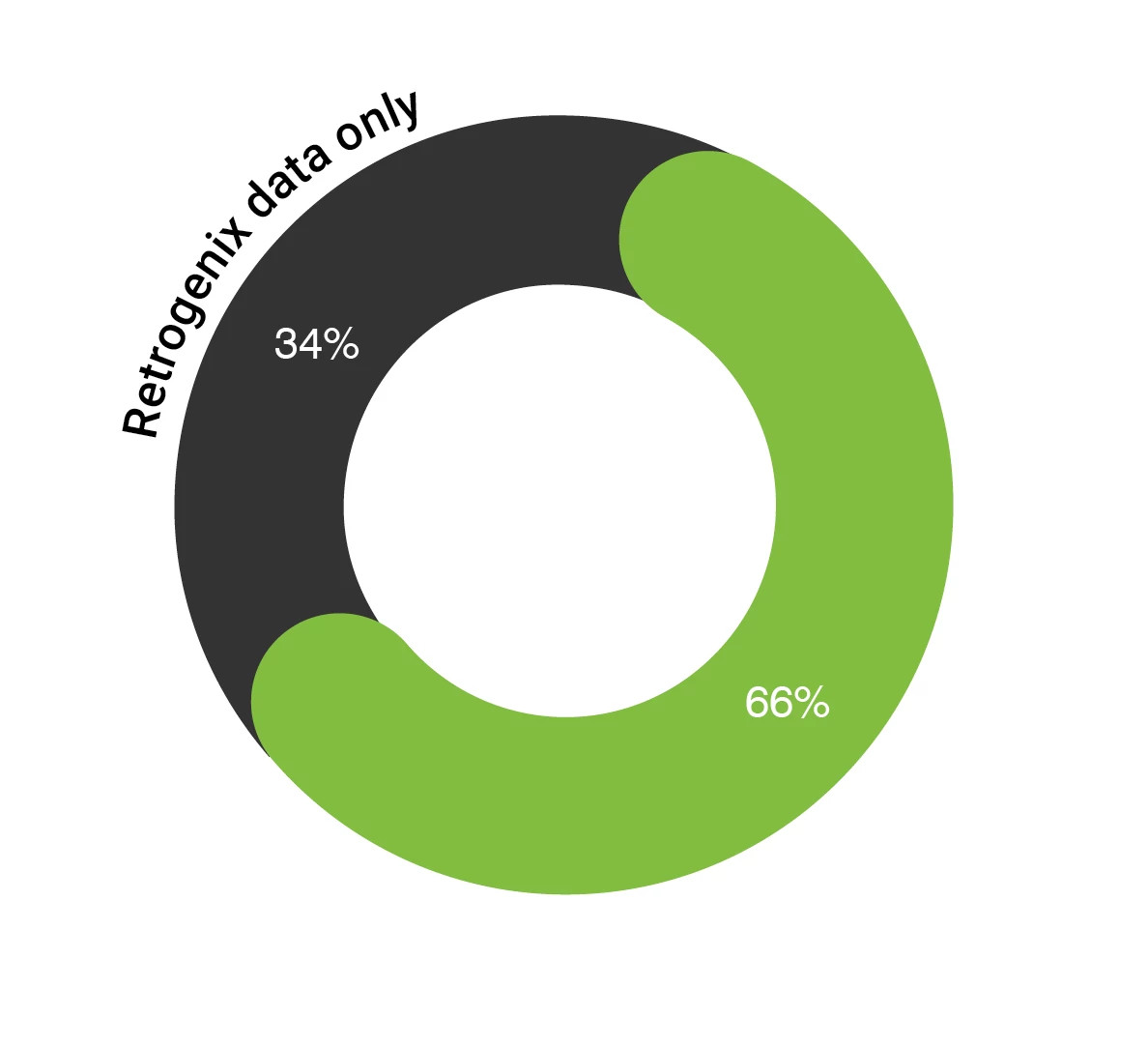
Retrogenix® cell microarray data is utilized as both an alternative to and complementary to immunohistochemistry-based (IHC) tissue cross reactivity (TCR). Until recently, standard toxicology target-binding assessment relied heavily on IHC-based TCR. More recent FDA guidance states that other methods can be used. We observed that 34% of the molecules put forward for regulatory approval relied on our data only for target specificity screening, with a further 67% using Retrogenix® cell microarray alongside IHC-based TCR and other cross reactivity data.
34% of IND submissions contained only Retrogenix® Cell Microarray data for cross-reactivity assessment
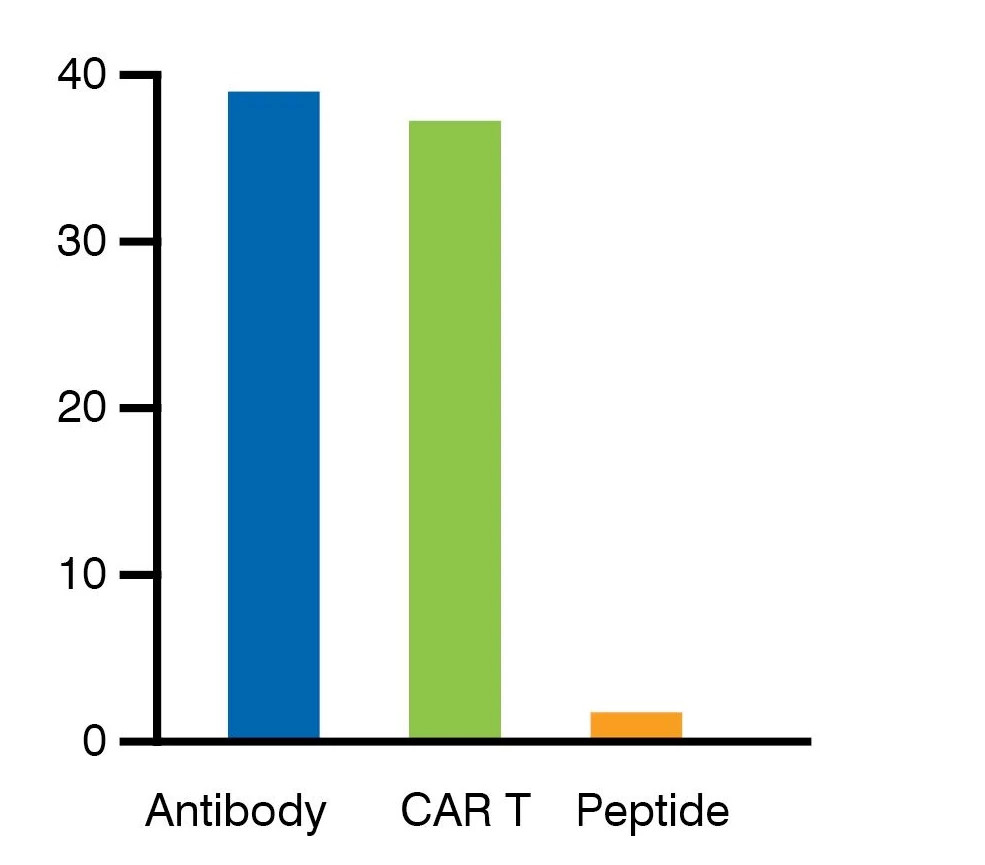
IND Submissions and Molecule Types
Due to the wide variety of specificity screening projects performed using our cell microarray platform, we have seen many different molecule types being submitted to the regulators. However, antibodies or antibody-related molecules and CAR Cells or scFvs are the predominant candidates, with at least 39 antibody and 37 cell therapy applications being put forward for IND approval by our respondents.
Number of each type of biotherapeutic submitted across all regions
If you would like a more detailed analysis of how our data has been used in NDA or IND submissions, or would like to discuss a potential study, please get in touch with us using our contact form.
eGuide for Off-Target Screening via the Retrogenix Platform
Download the eguide for all the information you need on how the Retrogenix platform can be leveraged to screen for specificity in vitro against a physiologically relevant human protein library from early-phase discovery onwards, to aid candidate selection, downstream study design, and IND approval.
Download now
Frequently Asked Questions (FAQs) for Cell Microarray Data in IND Submissions
-
How can I use the Retrogenix® Cell Microarray data in my IND package?
Depending on the study plan, clients receive a full IND-enabling report. We have worked with multiple pharmaceutical toxicology groups to optimize the report, and it is designed to be included in an IND safety package.
-
Should I combine Retrogenix® Cell Microarray data with other tox studies before submitting an IND application?
Retrogenix® Cell Microarray data has been used successfully as a powerful compliment to other IHC-based cross-reactivity studies, and in some cases as an alternative, in IND submissions to worldwide regulatory agencies. We would recommend looking at the specific requirements of each regulatory body for your biotherapeutic. Please contact us to discuss the best strategy for your program.
-
What are the advantages of performing both a Retrogenix® Cell Microarray assay and an IHC-based study at the pre-IND stage?
The Retrogenix® Cell Microarray Technology can provide data on what specific human plasma membrane and secreted proteins your biotherapeutic is binding to, while IHC-based approaches can provide histological context on where in the body any cross-reactive binding may be found. By generating and combining these two datasets, along with further toxicological interpretation, we can provide the most well-rounded assessment of a biotherapeutic’s specificity to reduce the risk of adverse effects caused by off-target binding in patients.