Browse our Cell Sourcing Products and Services
GUIDEBOOK
How Much Do You Really Know About Leukopaks?
Find out what they are and why they are important for immune research and cell therapy development. Our comprehensive guide covers best practices, considerations, applications, and in-depth characterizations for leukopak collections.
Download Guidebook
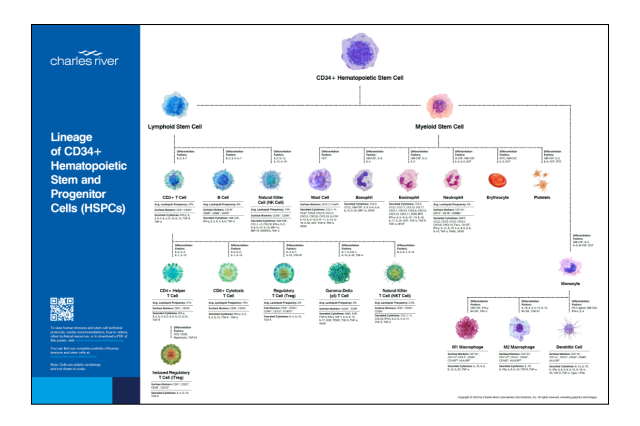
POSTER
Lineage of CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs)
Receive a complimentary poster to access information on immune cell markers, differentiation factors, cytokines, technical protocols, media recommendations, how-to videos and much more.
Download Poster
RESOURCES
Human Immune and Stem Cell Technical Resources
Explore our human immune and stem cell technical resources, which include protocols, media recommendations, how-to videos, and other useful resources to enable basic research and the development of advanced therapies.
View Resources
Quality Standards and Ordering Information
-
Licensure and Accreditation
Charles River complies with documented procedures governing the sourcing, handling, processing, storage, preserving, packaging, and distribution of products. Charles River is FDA registered (Blood and HCT/P) and meets all applicable GMP/GTP standards (21 CFR 210, 211, 606, 1271 and 21 CFR part 11 and part 58). Charles River maintains compliance with the State of California Laws and Regulations for production of biologics and tissue banking, meets CLIA requirements to ensure the accuracy and reliability of quality control testing and is AABB-accredited, which verifies that an appropriate quality management system and controlled collection processes are in place. In addition to the FDA, Charles River complies with European Medicines Agency (EMA) guidelines and our GMP-compliant facility is routinely audited by the FDA, State of California, and AABB.
-
How to Order
Please see How to Order – Cell Solutions.
-
Discounted Pricing
We are pleased to offer GSA MAS pricing discounts for government institutions located in the United States. Qualifying academic institutions are eligible for discounted pricing. Learn more
-
Terms & Conditions
Pricing is subject to Terms and Conditions – Cell Solutions.