What is a Leukopak?
A leukopak is an enriched apheresis product collected via leukapheresis, the act of extracting white blood cells (WBCs) from the peripheral blood. They contain a high concentration of peripheral blood mononuclear cells (PBMCs) – which consists of T cells, B cells, NK cells, and monocytes. Mobilized leukopaks are collected from healthy donors following an approved regimen that stimulates mobilization of CD34+ cells into the peripheral blood supply.
How Many Cells Are in a Leukopak?
Charles River leukopaks typically range between 6.4 to 22.9 billion white blood cells (WBCs), depending on the donor. On average, we see between 10 to 13 billion white blood cells per collection.
In addition to white blood cells, our leukopaks also contain plasma, some platelets, and very few red blood cells. Leukopaks typically consist of approximately 47% T cells, 20% monocytes, 9% B cells, 14% natural killer cells, 10% of other cell types (granulocytes, platelets, hematocrit cells).
Why Purchase Leukopaks from Charles River?
- Research use and GMP leukopaks to support you from discovery to manufacturing
- Low granulocyte and low red blood cell contamination
- High mononuclear cell viability
- We are a trusted provider of blood products and cellular starting material, with more than 45 years of combined experience in apheresis expertise
- 400,000+ successful apheresis collections performed
- We are the market leader in cryopreservation; we were the first to commercially market cryopreserved leukopaks
- Our nationwide donor center is FDA-registered, AABB- and CLIA-certified, and state-licensed
- Our large, diverse, and recallable donor network can support even the most stringent inclusion and exclusion research criteria
Explore Leukopak Products from Charles River
-
Fresh Leukopaks
Available in three SKU sizes: one bag, ½ bag, or ¼ bag. We also offer local delivery for fresh leukopaks to clients in Los Angeles and surrounding areas.
Explore Fresh LeukopaksYour company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Cryopreserved Leukopaks
Advance your project needs from basic research and R&D to process development and preclinical work and eliminate any challenges around logistics with our cryopreserved leukopaks. Long-term storage is possible in liquid nitrogen vapor phase. Short-term storage (less than two weeks) at -80 °C is also acceptable. Explore Cryopreserved Leukopaks
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Fresh Mobilized Leukopaks
Reduce donor-to-donor variations and access a more robust source of CD34+ hematopoietic cells with our fresh mobilized leukopaks. Our experienced medical team at our donor centers can safely conduct mobilized leukapheresis following six different dosing regimens. Explore Fresh Mobilized Leukopaks
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Disease State Leukopaks
Enhance your disease-state research with high-quality, well-characterized Research Use Only (RUO) human disease-state leukopaks collected from patients recruited using your inclusion / exclusion criteria (I/E) to meet your project requirements. Our disease state blood-derived products encompass a wide range of diseases. Explore Disease State Leukopaks
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
GMP-compliant Fresh and Cryopreserved Leukopaks
Ensure consistency and continuity in the development and commercialization of your advanced therapy with our GMP-compliant fresh and cryopreserved leukopaks. We offer phase-appropriate GMP-compliance through our fully developed and up-to-date quality systems, CGMP-trained personnel, and documentation provided during batch release. Explore GMP Leukopaks
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
GUIDEBOOK
How Much Do You Really Know About Leukopaks?
Find out what they are and why they are important for immune research and cell therapy development. Our comprehensive guide covers best practices, considerations, applications, and in-depth characterizations for leukopak collections.
Download Guidebook
Frequently Asked Questions (FAQs) About Leukopaks
-
How are Charles River leukopaks collected?
Charles River leukopaks are extracted using continuous flow centrifugal technology (Spectra Optia® apheresis system) directly into a sterile collection bag containing ACD-A anticoagulant, using the Continuous Mononuclear Cell (CMNC) collection protocol.
All leukopaks are collected under rigorous SOPs at our donor center, which is FDA-registered, fully accredited by AABB and CLIA, and licensed in accordance with applicable state regulations.
-
What’s the difference between leukopaks and whole blood?
The difference between leukopaks and whole blood is significant. Leukopaks have a much higher concentration of white blood cells (WBC) and a superior purity and quality compared to whole blood or even buffy coat samples.
In fact, leukopaks typically contain 20 times more mononuclear cells, which are highly sought after for their use in important research areas such as immunotherapy, vaccine development, and advanced therapy process development. With this increased WBC concentration and improved quality, leukopaks provide researchers with a valuable resource for obtaining accurate and reliable results in their studies.
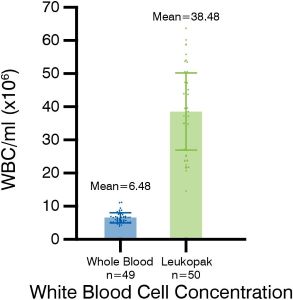
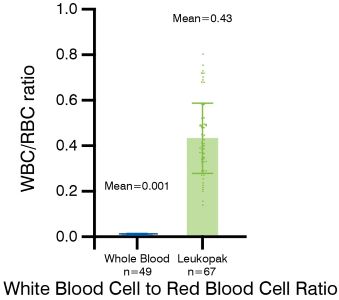
Figure 1 - WBC concentration is significantly higher in a leukopak, (mean of 38.48 x 106 WBCs/mL) than whole blood (mean of 6.48 x 106 WBCs/mL). Additionally, red blood cell (RBC) concentration is significantly lower in a leukopak with a mean of 0.43 WBC for every RBC (about 1 WBC for every 2 RBC), whereas in a whole blood unit, it is a mean of 0.001 WBC for every RBC (about 1 WBC for every 1,000 RBCs). -
How do you ensure consistency in a leukopak collection while maximizing yield?
A high-quality leukopak is a perfect balance of purity and yield. Donor-to-donor variability is inherent and often unpredictable. Achieving balance in purity and yield requires not only apheresis expertise, but also a commitment to donor management. With donor nurturing and dietary guidance, the potential quality of leukopaks from donors is optimized. However, even high-quality leukopaks can be variable in terms of yield as shown in Figure 2A.
A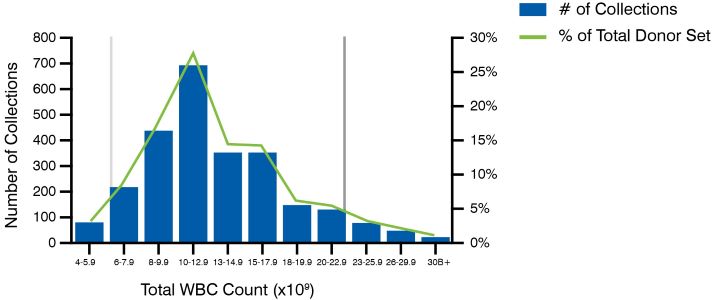 B
B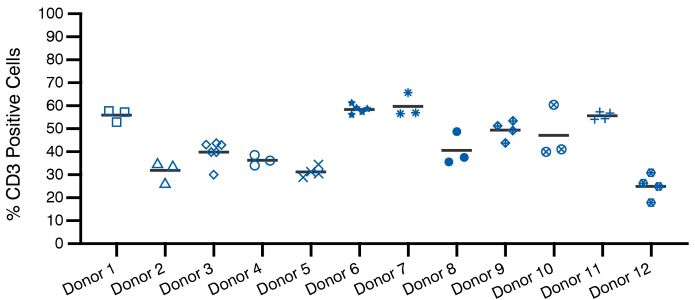
Figure 2 – Figure 2A demonstrates natural variability across a sampling of 2,490 leukopaks collected in our donor facility. The mean WBC yield of this analysis is 12.7 x 109 cells/donor and 5th (light grey line) and 95th (dark grey line) percentiles of 6.4 and 22.9 x 109 (cells/donor), but the distribution of normal WBC yield is quite broad. However, even high-quality leukopaks can be variable in terms of yield. Figure 2B demonstrates the relative consistency of CD3+ cellular populations collected from recallable, reliable donors at multiple timepoints. This consistency is also observed for CD4+ and CD8+ T cell populations. One strategy to minimize variability for certain applications is to utilize recallable donors.Although donor-to-donor variability certainly exists, with proper training, adherence to best practices, and standardization, collection-to-collection variability can be minimized. One strategy to minimize variability for certain applications is to utilize recallable donors (see Figure 2B).
Recallable donors are often assigned to a research program because they meet specific physiological and demographic traits and have self-identified as being willing to make repeat donations. Recallable donors are those who are responsive to outreach, tend to donate regularly, are punctual and communicate well, plus have good eating and hydration habits.
-
Can I select donors with particular physiological or demographic traits relevant to my research?
Definitely! In addition to standard collections, our donor center team can recruit donors with specific characteristics to meet your unique criteria. A key advantage of partnering with Charles River is our extensive and recallable donor network at your disposal. This diverse and well-characterized donor pool allows for swift and efficient recruitment of donors matching your specific research needs.
-
What type of leukopak is best for my research?
Choose the ideal leukopak for your research by browsing through our Charles River product portfolio, consisting of fresh, cryopreserved, mobilized, disease state, and GMP leukopaks.

