Featured Resource
GUIDEBOOK
How Much Do You Really Know About Leukopaks?
Find out what they are and why they are important for immune research and cell therapy development. Our comprehensive guide covers best practices, considerations, applications, and in-depth characterizations for leukopak collections.
Download Guidebook
- LYMPHOID CELLS
- MYELOID CELLS
- CELL MEDIA RECOMMENDATIONS
- TECHNICAL PROTOCOLS
- HOW-TO VIDEOS
- WHITE PAPERS
- SCIENTIFIC POSTERS
- WEBINARS
- BLOGS
Lymphoid Cell Lineage
Cell lines in the table below are available for purchase in addition to Peripheral Blood Mononuclear Cells (PBMCs).
| Cell Type | Surface Markers | Avg. Leukopak Frequency | Differentiation Factors | Secreted Cytokines |
| CD34+ Hematopoietic Stem Cells | CD34+ | Download White Paper | — | — |
| CD3+ T Cell | CD3+, CD45+ | 47% | IL-2, IL-7 | IFN-γ, IL-2, IL-3, IL-4, IL-10, IL-12, TNF-α, TGF-ß |
| CD4+ Helper T Cell | CD4+, CD45+ | 31% | IL-2, IL-4, IL-7, IL-12 | IFN-γ, IL-2, IL-4, IL-5, IL-6, IL-12, IL-21, TGF-ß |
| CD8+ Cytotoxic T Cell | CD8+, CD45+ | 14% | IL-2, IL-5, IL-7, IL-15 | IFN-γ, IL-2, IL-4, IL-12, TGF-ß, TNF-α |
| Regulatory T Cell (Treg) | CD4+, CD25+, CD45+, CD127-, FOXP3+ | 2% | IL-4, IL-7, IL-10, TGF-ß1 | IL-4, IL-10, TGF-ß |
| Induced Regulatory T Cell (iTreg) | CD4+, CD25+, CD45+, CD127- | — | CD3, CD28, Rapamycin, TGF-ß1 | IL-4, IL-10, TGF-ß |
| Gamma-Delta (γδ) T Cell | CD45+, TCRδ | 3% | IL-1, IL-2, IL-7, IL-15, IL-33, TNF-α | ANG, EGF, FGF-9, IFN-γ, IGF-1, IL-4, IL-10, IL-17, KGF, PDGF, TGF-ß, TNF-α, VEGF |
| Natural Killer T Cell (NKT Cell) | CD3+, CD45+, CD56+ | 2 – 5% | IL-2, IL-7, IL-15 | CCL-1~5, CXLC8, IFN-γ, IL-2, IL-4, IL-17, TGF-ß, TNF-α |
| Natural Killer Cell (NK Cell) | CD45+, CD56+ | 13% | IL-2, IL-12, IL-15, IL-18 | CCL-1~5, CXLC8, GM-CSF, IFN-γ, IL-5, IL-8, IL-10, IL-13, MIP-1α, MIP-1ß, RANTES, TNF-α |
| B Cell | CD19+, CD20+, CD22+, CD45+ | 9% | IL-3, IL-4, IL-7 | GM-CSF, IFN-γ, IL-2, IL-4, IL-6, TNF-α |
Myeloid Cell Lineage
Cell lines in the table below are available for purchase in addition to Peripheral Blood Mononuclear Cells (PBMCs).
| Cell Type | Surface Markers | Avg. Leukopak Frequency | Differentiation Factors | Secreted Cytokines |
| CD34+ Hematopoietic Stem Cells | CD34+ | Download White Paper | — | — |
| Monocyte | CD14+, CD16+, CD45+ | 20% | GM-CSF, IL-3, IL-6, M-CSF, SCF | IL-1ß, IL-6, IL-12, IL-15, IL-18, IL-23, TNF-α |
| M1 Macrophage | CD11b+, CD11chi, CD14+, CD45+, CD163low, HLA-DRhi | — | GM-CSF, IL-1ß, IL-4, IL-10, IL-13, TGF-ß1 | IL-1ß, IL-6, IL-12, IL-23, TNF-α |
| M2 Macrophage | CD11b+, CD11clow, CD14+,CD45+, CD163hi, HLA-DRlow | — | IL-1ß, IL-4, IL-10, IL-13, M-CSF, TNF-ß1 | IL-1ß, IL-1Ra, IL-6, IL-10, TGF-ß, TNF-α |
| Dendritic Cell | CD11b+, CD11c+, CD14+, CD45+, CD86+, HLA-DR+ | — | Flt-3 Ligand, GM-CSF, IFN-α, IL-4 | IL-1α, IL-1ß, IL-1Ra, IL-6, IL-8, IL-12, IL-18, IL-23, TGF-ß, TNF-α, Type 1 IFNs |
| Mast Cell | CD117, FceR1 | — | SCF | CCL1~5, CCL7, CCL9, CCL10, CCL17, CXCL2, CXCL8, CXCL10, G-CSF, IL-1ß, IL-2~6, IL-9~11, IL-13, IL-16, IL-33, SCF, TGF-ß, TNF-α, VEGF |
| Basophil | — | — | GM-CSF, IL-3, IL-4 | CCL3, CCL5, GM-CSF, IL-3, IL-4, IL-6, IL-13, IL-25, MIP-1α, VEGF |
| Eosinophil | — | — | GM-CSF, IL-3, IL-5 | CCL3, CCL5, CCL11, CCL13, CCL17, CXCL1, CXCL5, CXCL8, CXCL9, CXCL10, CXCL11, EDN, EPX, IFN-γ, IL-3~6, IL-10~13, IL-16, IL-17, IL-25, SCF, TGF-α, TGF-ß, TNF-α, VEGF |
| Neutrophil | CD11b+, CD15+, CD16+, CD66b+ | 5% | GM-CSF, EPO, GM-CSF, IL-3, SCF | BAFF, CCL2, CCL3, CCL5, CXCL1, CXCL8, CXCL10, Fas-L, G-CSF, IFN-γ, IL-1α, IL-1ß, IL-4, IL-6, IL-8, IL-10, TNF-α, TRAIL, VEGF |
| Erythrocyte | — | — | SCF, EPO,IL-3, GM-CSF | — |
| Platelets | — | — | GM-CSF, IL-3, IL-6, SCF, TPO | — |
Media Recommendations for Culturing Immune Cells
Download a copy of our media recommendations for culturing immune cells.
| Cell Type | CRL Recommended Medium | Alternative Medium Option 1 | Alternative Medium Option 2 | Additional Cytokines Needed for Survival, Differentiation, or Activation |
| CD34+ Hematopoietic Stem Cells | RPMI complete | — | — | SCF, IL-3, IL-6 |
| Mononuclear Cells (MNCs) | RPMI complete | AIM V | — | — |
| CD3+ Pan T Cells | X-VIVO | RPMI complete | AIM V | IL-2, IL-7 |
| CD4+ Helper T Cells | X-VIVO | RPMI complete | AIM V | IL-2, IL-7 |
| CD8+ Cytotoxic T Cells | X-VIVO | RPMI complete | AIM V | IL-2, IL-7 |
| CD4+/CD25+/CD127- Regulatory T Cells (Tregs) | X-VIVO | RPMI complete | — | IL-2, IL-4, IL-10, TGF-ß |
| CD3+/CD56+ Natural Killer T Cells (NKTs) | X-VIVO | RPMI complete | — | IL-2, IL-7, IL-15 |
| CD19+ B Cells | RPMI complete | α-MEM or DMEM | X-VIVO | IL-4, IL-7 |
| CD56+ Natural Killer Cells (NK Cells) | RPMI complete | DMEM | SCGM | IL-2, IL-15, IL-21 |
| CD14+ Monocytes | RPMI complete | DMEM | — | GM-CSF (differentiation to M1 Macrophages), M-CSF (differentiation to M2 Macrophages), GM-CSF + IL-4 (differentiation to Dendritic cells) |
| Macrophages (M1 & M2) | RPMI complete | X-VIVO | — | M1: GM-CSF M2: M-CSF |
| Dendritic Cells | RPMI complete | X-VIVO | — | GM-CSF, IL-4 |
| Neutrophils | RPMI complete | — | — | G-CSF, GM-CSF |
| Mesenchymal Stem Cells (MSCs) | α-MEM + 10% BSA | — | — | — |
Notes
X-VIVO is used without extra serum in standard culture, but 1~5% (v/v) of human AB serum can be added to enhance cell activity or survival depending on specific application.
RPMI Complete medium = RPMI 1640 + 10% Fetal Bovine Serum (FBS) + 2mM Glutamine. Heat-inactivated FBS can be used in the RPMI complete medium depending on specific application.
SCGM = Stromal Cell Growth Medium
Alternative medium must be used only when cell culture protocol has previously been developed for specific application.
Isolation and Cryopreservation of Immune Cells
| Isolation of Peripheral Blood Mononuclear Cells (PBMCs) Using a Density Gradient Reagent |
| Cryopreservation of Human Primary Cells (Target Cells) |
How to Thaw
How to Differentiate
| Differentiate Human CD14+ Monocytes into Dendritic Cells In Vitro |
| Differentiate Human CD14+ Monocytes into Macrophages (M1/M2) In Vitro |
Pluripotent Stem Cell Protocols
| Instructions for Use (Pluripotent Stem Cells) |
Miscellaneous Protocols
| The Activation of Human Neutrophils |
| How to Remove HypoThermosol® from Fresh Cells |
Troubleshooting Protocol
| How to QC Cryopreserved Cells (Vials) This protocol is recommended when the thawed cells are intended for use in QC testing applications. |
VIDEO
How to Thaw Cells in a Cryopreserved Leukopak
This "how to" video closely follows the technical protocol and shows step-by-step instructions for properly thawing cells in a cryopreserved leukopak, which is crucial to ensuring the viability and functionality of the cells.
VIDEO
How to Thaw Cryopreserved Cells (Vials)
It is essential to thaw cryopreserved cells correctly to maintain the cells' viability and functionality. This "how-to" video provides step-by-step instructions for thawing cryopreserved cells intended for use in downstream culturing applications and when testing a representative sample for quantification.
CASE STUDY
Using Cryopreserved Isolated Cells as a Starting Material for CAR-T Development
In this case study, we highlight the benefits of working with isolated cells as starting material for the development of effective cellular therapies.
Download Case Study
| A Comprehensive Analysis of CD34+ Stem Cell Mobilization Regimens Edward Scott, MD, Principal, Factotum Consulting – Tennessee, LLC Wassim Basheer, PhD, Senior Product Manager, Charles River |
| CD34+ Hematopoietic Stem and Progenitor Cells From Different Tissue Sources For Cell Therapy |
| A Comprehensive Analysis of Fresh Apheresis Collections: Conclusions and Best Practices Dominic Clarke, PhD, Global Head of Cell Therapy, Charles River Brad Taylor, PhD, Global Marketing Director, Charles River Neal Lawrence, R&D Scientist, Charles River |
| A Study Assessing Fresh Leukopak Stability Neal Lawrence, R&D Scientist, Charles River Wassim Basheer, PhD, Senior Product Manager, Charles River Keith Hansen, Global Marketing Director, Charles River |
| A Comparative Study: Are Cryopreserved Leukopaks a Viable Alternative to Fresh? Brad Taylor, PhD, Global Marketing Director, Charles River Wassim Basheer, PhD, Product Manager, Charles |
| A Reliable Donor Network is the Foundation for Development of Successful Cell-Based Therapies Brad Taylor, PhD, Global Marketing Director, Charles River Wassim Basheer, PhD, Senior Product Manager, Charles River |
| Best Practices in Collection of Human Bone Marrow Aspirate for Pre-Clinical and Clinical Use Brad Taylor, PhD, Global Marketing Director, Charles River Darren Pelio, MHS, PA, Bone Marrow Aspiration Specialist |
| GMP-Compliant Human Bone Marrow for Cell Therapy Applications Dominic Clarke, PhD, Global Head of Cell Therapy, Charles River Brad Taylor, PhD, Global Marketing Director, Charles River |
| Regulatory T Cell Modulation of Immune Suppression |
POSTER
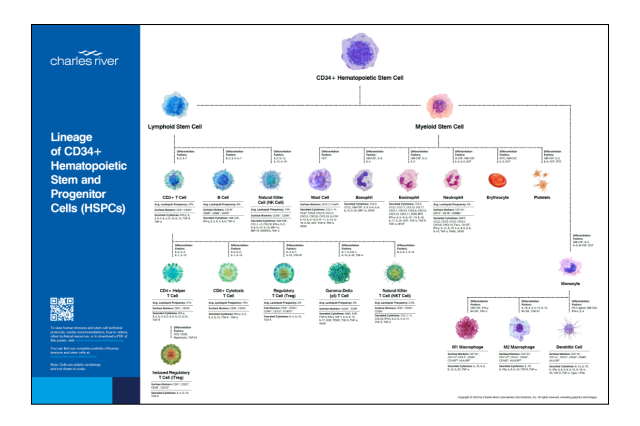
Lineage of CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs)
Receive a complimentary poster to access information on immune cell markers, differentiation factors, cytokines, technical protocols, media recommendations, how-to videos and much more.
POSTER
Patient-derived Organotypic Tumor Spheroids (PDOTS) Optimize Donor Selection in Allogenic Cell Therapy Development
Authors: Chunxiao Cui, Raluca Dumitru, Danielle Hagee, Anthony Attardo, Kirti Khardenavis, Wassim Basheer, Sabrina D. Munnik, David C. Lopez, Gemma Moiset, Ketki Bhise, Sinal Patel, Alyssa Martin, Michael A. Perricone, Aaron Goldman
POSTER
Characterization and Function of Donor-Derived Antigen-Specific and Regulatory T Cells
Authors: Arif Azam Khan, Jaehyung Park, Nathan LaVoy, Jeff Bellinder, Chris Tompkins, Tisha San Miguel, Neal Lawrence, Brianna Schoen
POSTER
Characterization and Function of Human T Regulatory Cells – Suppression of Proliferation and Antigen-Specific Cytotoxicity
Authors: Arif Azam Khan, PhD, Nathan Lavoy, Jaehyung Park, PhD, Chris Tompkins, Brianna Schoen, PhD
POSTER
Generation of Multi-Functional and Tumoricidal NKT-like Antigen-Specific T cells
Authors: Jaehyung Park, Jeff Bellinder, Tisha San Miguel, Neal Lawrence, Arif Azam Khan, Nathan LaVoy, Chris Tompkins, Brianna Schoen
POSTER
Optimization of Seeding Density for Enhanced T Cell Expansion and Correlation to Metabolite Production in G-Rex Bioreactors
Authors: Jeff Bellinder, Tisha San Miguel, Jaehyung Park, Andrew Scheffler, Brianna Schoen
POSTER
Reduction of Immunosuppressive Function of Regulatory T Cells Enhances Antigen-Specific T Cell Mediated Cytotoxicity Against Tumor Cells
Authors: Arif Azam Khan, Jaehyung Park, Nathan LaVoy, Chris Tompkins, Jeff Bellinder, Tisha San Miguel, Neal Lawrence, Brianna Schoen
WEBINAR
Safeguard Your Stem Cells: Strategies to Preserve Bone Marrow Stability
This webinar will explain strategies to preserve fresh bone marrow stability by comparing the results of a recent study where bone marrow aspirate was stored at controlled room temperature (CRT) and cold temperature (2 –8 °C) over a three-day period.
WEBINAR
Optimize Cell Therapy Manufacturing Efficiency with Cryopreserved Cells
This webinar examines how using purified and cryopreserved cell populations, such as GMP-compliant PBMCs and isolated T cells, can streamline CAR-T manufacturing. Learn best practices and strategies to reduce variability and support consistent, high-quality outcomes in both autologous and allogeneic therapies.
WEBINAR
Induced Pluripotent Stem Cells: Case Studies of Effective Differentiation and Regulation
We explore challenges and solutions in using iPSCs for cell therapy development, focusing on effective differentiation protocols and addressing safety concerns. Experts share insights on successful methods for differentiating and regulating human iPSCs.
WEBINAR
Have the End in Mind: Starting Material Considerations for Cell Therapies
View this webinar to explore the key factors of starting material and how they can impact the success of your cell therapy.
WEBINAR
Finding Effective CAR Drivers Using Ex Vivo Efficacy Models for Successful Cell Therapies
In this webinar, we provide insights on donor management strategies to improve cell therapy outcomes, the impact of source material variability on downstream cell therapy efficiency, dynamic ex vivo preclinical efficacy testing models, and the impact of differential gene expression between donors on cell therapy potency.
WEBINAR
Managing Donor Variability to Develop Robust PBMC Humanized Models for Cancer Research
Our scientific experts share data showcasing the impact of donor variability on the development of PBMC humanized NCG mouse models used for cancer studies.
WEBINAR
Comparing CAR T Cells Across Novel Cryopreservation Containers and Platforms
Depending on how CAR T cells are cryopreserved and shipped, their fitness and function can change. This webinar will discuss current methods in use and present a novel cryopreservation process and container that is both more robust and adaptable for these potentially lifesaving cells.
WEBINAR
How Fresh is Fresh? A Leukopak Stability Study
We share valuable data on how different storage temperatures may impact cell yields over multiple time points, and discuss the results of our investigation into the optimal temperature for preserving fresh leukopak stability.
WEBINAR
Leukopaks or iPSCs? Determining Your Ideal Cellular Starting Material
While leukopaks and induced pluripotent stem cells (iPSCs) are both viable options for cellular starting material, it can be challenging to select the most suitable one for your specific requirements. We compare these two different cellular starting material sources and providing impactful data to help you make an informed decision.
WEBINAR
Characterizing Donor-derived Cellular Starting Material for Advanced Therapies
We discuss how sourcing cellular starting material as well as a highly characterized donor network can contribute to maintaining regulatory compliance while advancing your cell or gene therapy program.
WEBINAR
CD34+ Stem Cells: Which Mobilization Regimen is Right for You?
Hematopoietic stem cells have unique properties that make them extraordinarily valuable in CGT development. It's important to know which method of cell mobilization and collection will provide the right yield and quality for your program's success. We cover what those available methods are, their key differences, and how those differences could affect the development of your cell or gene therapy.
WEBINAR
Utility of Antigen-Specific T Cells (ASTCs)
This webinar highlights the usage of antigen-specific T cells when developing immunomodulatory therapeutics to a wide range of diseases caused by viruses, bacteria, tumor cells, and autoimmunity.
WEBINAR
T Cells From Beginning to End: Optimizing Leukopaks and Drug Product Cryopreservation
Learn strategies for cryopreserving leukapheresis material for T cell processing and how to extend leukopak stability for added flexibility in manufacturing T cell therapies.
WEBINAR
CARrying Your CAR-T to the Clinic
Chimeric Antigen Receptor (CAR)-based therapies are rapidly developing in oncology and other fields. In this webinar you will learn how to screen multiple CAR constructs, test engagement and optimize scFv for earlier identification of optimal constructs, how to test the efficacy and safety of your CAR cell product in vitro, and which animal models can be used for preclinical testing of your CAR cell product.
WEBINAR
Building Better CARs: Advancing Dual-Targeting Discovery
Learn how Century Therapeutics was able to target cancer cells and deliver patients allogenic treatments with reduced side effects and improved outcomes through design and development of a novel, dual-targeting CAR for their allogeneic IPS cell-derived iT platform.
WEBINAR
Demystifying Cell Therapies: A Stroll Through Cell Therapy Drug Development
Cell therapies have shown tremendous clinical success and benefits for patients. In this webinar you'll get updates on the significant advances in immunology, genetics, and advanced modalities, and explore cell therapies and the revolutionary role they play in disease treatment and obtain a roadmap to help overcome pitfalls along your journey.
Ask a Scientist Blogs
| Antigen-Specific T Cells Arif Azam Khan, PhD |
| Proteoglycans vs. Glycoproteins Anne Lodge, PhD |
| What's the Difference Between Human Monocytes and Macrophages? Anne Lodge, PhD |
| What's the Difference Between Naïve and Memory T Cells? Anne Lodge, PhD |
| What's the Difference Between M1 and M2 Macrophages? Anne Lodge, PhD |
| What's the Difference Between Serum and Plasma? Anne Lodge, PhD |
| When Should I Use NK Cells or Cytotoxic T Cells? Anne Lodge, PhD |
Immunology/Oncology Blogs
| A Tale of Two Assays: Recall Antigen vs. Antigen-Specific T Cell Anne Lodge, PhD |
| Hot Tumors vs. Cold Tumors Anne Lodge, PhD |
| How Does the Leukapheresis Collection Procedure Work? Anne Lodge, PhD |
| Immunology for Non-Immunologists: Cytokine Measurement Anne Lodge, PhD |
| Immunology for Non-Immunologists: MHC and HLA Explained Anne Lodge, PhD |
| Immunology for Non-Immunologists: Proliferation Assays Anne Lodge, PhD |
| Immunology for Non-Immunologists: T Cell Antigen Recognition Anne Lodge, PhD |
| Surface Antigen Expression Anne Lodge, PhD |
| Tumor-Infiltrating Lymphocytes: A New Weapon Against Melanoma Arif Azam Khan, PhD |
| Useful Tools in Immunology: OMIM.org Anne Lodge, PhD |
| Using Anti-MART-1 T Cells for Melanoma Immunotherapy Research Anne Lodge, PhD |
| Using PBMC as a Quality Control for Flow Cytometry Anne Lodge, PhD |
| What Are Irradiated PBMCs? Anne Lodge, PhD |