Human Peripheral Blood Mononuclear Cells (PBMCs)
All PBMCs are isolated onsite from a fresh leukopak collection using Ficoll density gradient centrifugation and are then immediately cryopreserved using CryoStor CS10. Researchers looking to buy human PBMCs can trust our strict SOPs, which ensure the highest quality, yield and purity of mononuclear cells while greatly reducing contamination from unfavorable cells such as granulocytes, red blood cells and platelets. Flow cytometry is used to determine white blood cell yield, purity, and the percentage of various immune cell subsets. Our PBMC product undergoes post-thaw quality control testing to ensure recoverability, cell count, and viability prior to product release.
Optimize Cell Therapy Manufacturing Efficiency with Cryopreserved Cells
View this webinar to learn how purified and cryopreserved cell populations streamline logistics, reduce variability, and enhance transduction efficiency in CAR-T production.
Watch on Demand
Buy PBMCs for Your Research
-
Normal Cryopreserved PBMCs
Ideal for isolation of T cells, B cells, NK cells, and Monocytes to be used for basic research, development of advanced therapies, toxicology studies, and drug and vaccine development.
Size Product Code 10 x 10e6 cells PB009C-1 25 x 10e6 cells PB009C-2 50 x 10e6 cells PB009C-50 100 x 10e6 cells PB009C-3 Custom PB009C-KIT* Full Leukopak Kit PB009C-FL-KIT* Contact us if you don’t see a specific product, size, or format, or if you would like to customize your request.
*Product is made to order.Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
GMP PBMCs
We offer a convenient source of GMP-compliant isolated mononuclear cells for those advancing from pre-clinical to commercial stages of therapeutic development. This option allows for efficient access to necessary GMP starting materials, saving both time and resources.
Receive a high-quality, cryopreserved product that meets GMP standards within our fastest turnaround time. Explore GMP PBMCsYour company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Irradiated PBMCs
PBMCs that cannot proliferate. Ideal as stimulator, antigen presenting, or feeder cells. X-Ray irradiated PBMCs are a much safer alternative to using gamma rays from a Cesium 137 source since there is no radiologic material used and no chance of ionizing contamination. X-Ray irradiated human PBMCs are also a better alternative than using Mitomycin C treated PBMCs since Mitomycin C is toxic and requires extra handling steps leading to PBMC loss. Explore Irradiated PBMCs
Size Product Code 25 x 10e6 cells PB009C-IRRD-2 50 x 10e6 cells PB009C-IRRD-50 100 x 10e6 cells PB009C-IRRD-3 Custom PB009C-IRRD-KIT* Contact us if you don’t see a specific product, size, or format, or would like to customize your request.
*Product is made to order. -
Disease State Cryopreserved PBMCs
PBMCs isolated from immune-mediated (autoimmune) disease patients. Other diseases available upon request. Collected in accordance with an IRB-approved protocol. Our disease state cryopreserved PBMCs encompass a wide range of diseases, and are an invaluable resource in the research and development of antigen-specific T cells targeting disease-associated epitopes.
Contact us to check on availability or custom recruitment.
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Mobilized Cryopreserved PBMCs
PBMCs derived from mobilized apheresis collections with increased CD34+ yields.
Size Product Code 50 x 10e6 cells M009C-1 100 x 10e6 cells M009C-2 250 x 10e6 cells M009C-3 Contact us if you don't see a specific product, size, or format.
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Pooled Cryopreserved PBMCs
Produced by combining lots of PBMCs from different donors. Ideal for detecting pyrogen presence (Monocyte Activation Test), which is required testing for all injectable pharmaceuticals. Pooled PBMCs are also beneficial when doing antibody preclinical testing.
Size Product Code ≥10 X 10e6 cells MPBMC-1076 Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
Cryopreservation of PBMCs on Day 0 Simplifies Complex Logistics While Preserving Viability and Distribution Frequencies of the Different Cellular Subpopulations
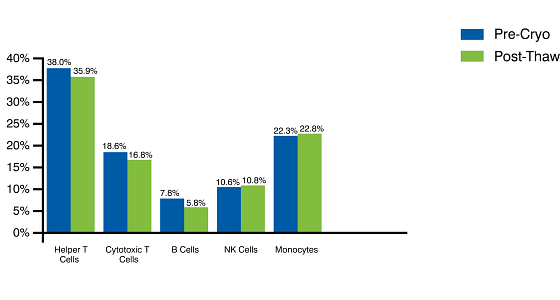
Cryopreserved PBMCs maintain subcellular distribution frequencies post-thaw.
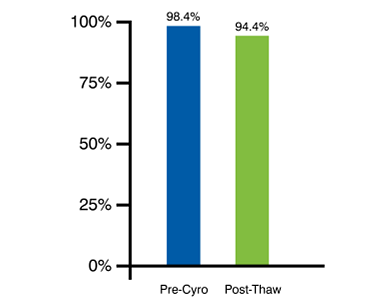
Day 0 freezing results in superior post-thaw viability of PBMCs.
Figure 1. Human peripheral blood mononuclear cells are examined to determine viability and cell type distribution profiles based on cell surface markers before and after cryopreservation. Our PBMC samples are guaranteed to have >95% viability and >90% purity pre cryopreservation as assessed by flow cytometry.
Characterization of Human PBMCs
The majority of human peripheral blood mononuclear cells, 70% on average, are lymphocytes. Lymphocytes fall into three main categories, namely T cells, B cells, and NK cells. Most lymphocytes are CD3+ meaning that they are T cells of some kind. Together the 3 main subtypes of T cells (CD3+, CD4+, and CD8+) make up the highest percentage of PBMC cell types (40-50% on average). T cells are currently very important for advanced therapies, particularly for cancer immunotherapy studies.
B cells (CD19+) make up on average about 10% of the PBMC population, and they play a high-profile role in cancer immunotherapy studies, as well as in studies focused on treating autoimmune diseases. NK cells (CD56+) make up on average about 14% of the human PBMC population. NK cells are a type of granular lymphocyte capable of directly attacking and killing invading pathogens and they play an important role in the development of cell-based immunotherapies such as CAR-NK therapies.
Monocytes (CD14+) make up on average about 20% of the PBMC population. Monocytes can differentiate into either macrophages, which are phagocytic, or into dendritic cells, which act as antigen-presenting cells.
Ready to Buy Human PBMCs?
Request a quote today and explore our full range of options, including normal, GMP-compliant, irradiated, disease-state, mobilized, and pooled PBMCs.
How Much Do You Really Know About Leukopaks?
Find out what they are and why they are important for immune research and cell therapy development. Our comprehensive guide covers best practices, considerations, applications, and in-depth characterizations for leukopak collections.
Download Guidebook
Frequently Asked Questions (FAQs) About Human PBMCs
General Information About Human PBMCs
-
What are human Peripheral Blood Mononuclear Cells (PBMCs)?
Peripheral blood mononuclear cells (PBMCs) are a mixed population of immune cells that circulate in human blood, primarily T cells, B cells, NK cells, and monocytes. Because they play central roles in immunity and inflammation, PBMCs are a cornerstone for cell therapy research, immuno-oncology studies, vaccine development, and infectious disease modeling.
PBMCs are typically collected through leukapheresis, a specialized process that enriches for mononuclear cells in a collection bag referred to as a leukopak. Charles River offers leukopaks and purified human PBMCs.
Leukopaks and PBMCs are collected from healthy donors who have consented under an IRB-approved protocol at our FDA-registered HemaCare Donor Center in the United States. In addition to standard collections, our donor center team can recruit donors with specific characteristics to meet your basic research or drug discovery needs.
-
What types of cells can be isolated from human PBMCs?
PBMCs can be further separated into specific subpopulations depending on research needs. The most common subsets include:
- T cells: Central in adaptive immunity, often used in CAR-T and TCR-T programs
- B cells: Antibody-producing cells for immune function studies
- Natural Killer (NK cells): Innate immune cells useful in cytotoxicity assays
- Monocytes: Precursors of macrophages and dendritic cells, used in immunology and regenerative biology
-
Can PBMCs be used as starting material for cell therapy development instead of leukopaks?
Yes. Using purified PBMCs as starting material can streamline cell therapy workflows more effectively while eliminating the concern of having contaminating granulocytes, which are typically found in leukopaks, that can affect the viability and functionality of the PBMCs. A case study highlights the benefits of working with PBMCs as a starting material for effective CAR-T development.
PBMC Collection, Isolation, and Processing
-
How are human PBMCs collected and isolated?
PBMCs are most commonly obtained from leukopaks collected via leukapheresis. They are then isolated using density gradient centrifugation (e.g., Ficoll) or other enrichment methods. Charles River provides ready-to-use PBMCs and protocols for in-house isolation from leukopaks, allowing flexibility depending on the research stage. For detailed step-by-step instructions on isolating PBMCs from a fresh leukopak, you may refer to our leukopak PBMC isolation protocol.
-
How are human PBMCs cryopreserved?
Charles River PBMCs are cryopreserved in a controlled-rate process using CryoStor® CS10 cryoprotectant to maintain high viability and functionality. Cells are stored in the vapor phase of liquid nitrogen until shipment, ensuring stability across global distribution.
-
How are the PBMCs characterized?
Each Charles River PBMC lot undergoes immunophenotyping by flow cytometry using the 8-color immunophenotyping kit from Miltenyi Biotec. Key subpopulations (T cells, B cells, NK cells, and Monocytes) are quantified, and results are reported in the Certificate of Analysis (CoA). This quality documentation provides confidence in cell composition and reproducibility for downstream experiments.
Cryopreservation, Handling, and Storage of PBMCs
-
How should I properly store and thaw PBMCs?
PBMCs should be transferred to liquid nitrogen vapor phase immediately upon arrival for long-term storage or thawed for immediate use. Long-term storage of cells at -80°C is NOT recommended. When ready for use and for best results, follow our step-by-step thawing protocol or instructional video.
-
What is the viability of the PBMCs?
When thawed according to our recommended protocol, Charles River guarantees a post-thaw viability of ≥ 90% as measured by NucleoCounter NC-200™. We also guarantee PBMC yields post-thaw as listed per vial size.
Safety, Compliance, and Donor Screening for PBMCs
-
What biosafety measures should I consider when handling PBMCs?
PBMCs should be handled under Biosafety Level 2 (BSL-2) procedures. Universal precautions should be utilized when working with PBMCs. Always use aseptic technique and personal protective equipment (PPE) and follow institutional biosafety guidelines when thawing or handling the cells.
-
Which donor screening tests are performed for the PBMCs?
Donor eligibility and testing follow FDA requirements for allogeneic blood donation. Standard screening includes:
- Syphilis (serological)
- Unexpected red cell antibody screening
- Hepatitis B (surface antigen and core antibody
- Hepatitis C antibody
- HIV 1/2 plus O antibody
- HTLV-I/II antibody
- HIV-1/HCV/HBV Nucleic Acid Testing
- West Nile Virus Nucleic Acid Testing
- Trypanosoma cruzi Antibody (Chagas Disease)
Results are reported in the CoA. Cytomegalovirus (CMV) testing is available upon request for programs that require CMV-specific donor status.
Ordering and Shipping
-
How quickly can customers expect to receive shipments of PBMCs after ordering?
All shipments are coordinated based on the terms specified in your Product Order Form (POF). Domestic orders are typically delivered the next business day via specialty courier, FedEx First Overnight (by 8:30 AM), or FedEx Priority Overnight (by 10:30 AM). International delivery times vary by destination and customs clearance requirements.