Your Trusted Partner for Cell Therapy GMP Manufacturing & Processes
As an end-to-end cell therapy CDMO, we excel in managing all phases of your product development lifecycle and associated supply chain, facilitating a seamless journey from cell therapy process and analytical development to Good Manufacturing Practices (GMP) and commercialization. With integrated quality control and biologics testing, this depth of knowledge allows us to ensure the safety, quality, and efficacy of products, while meeting the highest regulatory standards and maintaining efficient production timelines.
“Akron Bio is honored to support Charles River’s effort to improve manufacturing efficiency and patient safety by further closing the cell therapy manufacturing process with our CSS cytokines.”
David Smith, President and Chief Commercial Officer, Akron
Explore our Cell Therapy CDMO Facility
With 25 ISO 7 / Grade B US / EU compliant GMP suites available, we offer clinical through commercial production of cell therapies for global markets.
View the Facility
Accelerate Your Cell Therapy Manufacturing Program
An experienced CDMO, we offer cell therapy manufacturing services for various autologous and allogeneic cell types and starting materials, including marrow-infiltrating lymphocytes (MILs), dendritic cells (DCs), natural killer (NK) cells, T-cells, CAR-T, bone marrow-derived mesenchymal stem cells (BMSCs), mesenchymal stem cells (MSCs), whole blood, apheresis, leukapheresis, tumor isolates, and stem cells.
In addition, we have experience handling TILs, tumors (resections/biopsies/lysate), and apheresis material, including mobilized, whole blood, and buffy coat. Learn more about cell sourcing
Tech agnostic and experienced with leading-edge platforms, we provide a comprehensive, data-driven phase-appropriate approach that reduces risk and meets all cell therapy GMP manufacturing guidelines. By integrating cell therapy tech transfer and commercial readiness early in the product development lifecycle, we reduce late-stage CMC challenges and speed time to market.
With already validated systems, our teams have optimized and scalable cell therapy manufacturing processes and analytics on closed and automated systems for all or part of your drug development needs.
Navigating Complexity from Clinic to Market
In this article, our experts share how to de-risk the cell therapy development process in order to accelerate timelines and streamline CGT manufacturing.
Download Now
Our comprehensive cell therapy CDMO support includes:
-
Manufacturing Science and Technology (MSAT) team
A team comprised of experts in process development, technology transfer, and manufacturing support, offers optimized and scalable cell therapy manufacturing processes to bolster high-quality, efficient, and cost-effective manufacturing.
-
Quality management systems (QMS)
From early phase through commercial manufacturing our comprehensive QMS ensure therapies are safe, effective, and meet regulatory compliance standards.
-
Quality control/release testing
We uphold the safety, purity, and potency of cell therapy products through comprehensive analytical testing, meeting regulatory compliance, and maintaining quality throughout development and manufacturing.
-
Regulatory support
This team will help you navigate the complex landscape of cell therapy development, ensuring compliance with global regulations at every stage to commercial manufacturing.
-
Clinical operations and logistics
Having access to shipping and distribution of patient materials and therapies as well as regulatory compliance guidance and project management expertise for clinical trials is paramount. Seamlessly progress treatments through clinical development into commercial manufacturing.
-
Assay development
We offer assay development services to support cell therapy product characterization and release testing. Develop and validate assays to facilitate the safety, purity, and potency of cell therapies with the help of our specialists.
-
Validation and qualification
Our team of experts can support developing and executing validation and qualification plans, including process and equipment, facility, and software validation.
Are you looking for support for cell therapy CDMO support for your upcoming program?
Cell Therapy Supply Chain Solutions for Minimizing Vein-to-Vein Time
We understand the intricate nature of cell therapy supply chain management within the manufacturing process. As part of our comprehensive services, we specialize in managing supply chain integration to ensure a seamless flow of materials and information throughout the development of cell therapy manufacturing, and commercial production process.
Supply Chain Management
We excel in coordinating schedules and collaborations with apheresis centers and clinics to facilitate the timely and safe collection of patient samples, including the harvest, formulation, packaging, and cryopreservation of cells and cell therapies.
Temperature-Controlled Storage & Shipment
We prioritize cell therapy integrity by meticulously managing temperature-controlled storage and transportation requirements before, during, and after shipping to preserve quality and efficacy. This includes customs clearance.
Logistics and Distribution
Our logistical solutions include the final stage of thawing and administering the therapy to maximize therapeutic benefits. Located near the FedEx Super Hub, our Memphis facility minimizes transit times and ensures timely delivery of critical materials.
Regulatory Bodies and Facilitating Clinical Trials
We take pride in our role as a trusted partner in your cell therapy manufacturing process. We not only support relationships with regulatory bodies and act as your advocate, we interface with additional Contract Manufacturing Organizations (CMOs) and regulatory agencies for global clinical trials and product roll-outs. We understand the importance of seamless coordination and liaise with patient collection facilities and your Contract Research Organizations (CROs). With our expertise and extensive network in cell therapy CDMO services, we are here to ensure that your manufacturing runs smoothly and efficiently, from Phase 1 to commercialization, allowing you to focus on advancing your therapies and bringing your products to market.
Frequently Asked Questions (FAQs) About Cell Therapy Manufacturing
-
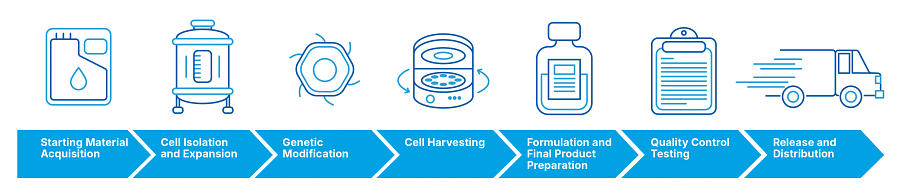
What are the steps in the cell therapy manufacturing process?
- Starting Material Acquisition: Donor cells (allogeneic therapy) or patient cells (autologous therapy) are collected and processed to obtain the initial cell population.
- Cell Isolation and Expansion: The desired cell population is isolated and expanded in culture to achieve sufficient cells for therapy.
- Genetic Modification (if applicable): Gene editing or transduction techniques may be employed to introduce therapeutic genes or modify cell characteristics.
- Cell Harvesting: The cells are harvested from the culture and processed to remove impurities and unwanted cell components.
- Formulation and Final Product Preparation: The cells are formulated into the final product, which may involve cryopreservation, formulation in a specific medium, or other processing steps.
- Quality Control Testing: The final product undergoes extensive testing to meet safety, identity, potency, and purity requirements.
- Release and Distribution: Once the final product passes quality control testing, it is released for clinical use. It is then distributed to the treatment site or patient.
Throughout the cell therapy manufacturing process, regulatory compliance, quality management, and documentation are crucial in ensuring the cell therapy product's safety, efficacy, and traceability.