Why Purchase Cryopreserved Leukopaks from Charles River?
Charles River has spent more than a decade perfecting its cryopreservation techniques to ensure optimal viability and functionality of its cryopreserved leukopaks.
- First to market commercially available cryopreserved leukopaks in the industry
- Cryopreserved leukopaks have >90% viability, with <5% granulocyte contamination on average
- Pre-cryopreservation white blood cell counts between 2 – 2.5 billion cells per bag
- Same-day on-site processing using GMP-compliant cell freezing media and rate-controlled cryopreservation, ensuring optimal cell viability and recovery
- Manufacturing continuity between our RUO and GMP-compliant leukopaks. Researchers can effortlessly move from the development phase to clinical trials with assurance.
- Leukopaks are obtained in accordance with stringent SOPs at our donor center, which is registered with the FDA, certified by AABB and CLIA, and licensed by the state of California
Explore Cryopreserved Leukopaks from Charles River
-
Normal, Healthy Cryopreserved Leukopaks
Leukopaks Size Product Code Normal, Healthy Cryopreserved 2-2.5 x 10e9 cells (1 bag) PB001LCLP** 4-5 x 10e9 cells (2 bags) PB001LCLP2-KIT** 8-10 x 10e9 cells (4 bags) PB001LCLP4-KIT** 12-15 x 10e9 cells (6 bags) PB001LCLP6-KIT** Full collection / Multiple bags PB001CLP-1-KIT* Contact us if you don’t see a specific product, size, or format, or would like to customize your request.
*Product is made to order.
**Granulocyte specification less than 15% per bagYour company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Disease State Cryopreserved Leukopaks
Our Disease State Program offers the opportunity to enhance your disease state research with our high-quality, well-characterized research use only (RUO) cryopreserved leukopaks. These leukopaks are obtained from patients who meet your specific inclusion and exclusion criteria (I/E), ensuring the highest quality samples for your research.
In addition to cryopreserved leukopaks, we also offer the following disease state cryopreserved peripheral blood-derived products for research use, collected in accordance with an IRB-approved protocol, and encompassing a wide range of diseases:
- Immune cells
- Serum
- Plasma
Discover more about our disease state cryopreserved leukopaks and elevate the quality of your disease state studies. Explore Disease State Cryopreserved LeukopaksYour company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
GMP Cryopreserved Leukopaks
We offer two GMP cryopreserved leukopak options: CliniPrime® and GMPrime® cryopreserved leukopaks. Both options adhere to GMP standards and meet regulatory requirements from the initial process development all the way through commercialization. Our fully developed and up-to-date quality systems, trained personnel, and detailed documentation provided during batch release ensure phase-appropriate GMP-compliance for your peace of mind. Explore GMP cryopreserved leukopaks
GMP Leukopaks Who It's For CliniPrime® Cryopreserved Leukopaks Clients who want to use a standardized protocol. Requires less resources and time GMPrime® Cryopreserved Leukopaks Clients who want a customized protocol to meet specific project needs. Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
Cryopreserved Leukopak Study Conducted by Charles River
-
Cryopreserved Leukopak Comparative Study
How does cryopreserved leukapheresis material stand up to freshly isolated leukapheresis material? Supporting data in our white paper, A Comparative Study: Are Cryopreserved Leukopaks a Viable Alternative to Fresh? reveals that cryopreserved leukopaks are a viable option to fill a growing need for reliable, on-demand starting materials to advance the development and commercialization of autologous and allogeneic cell therapies. Download White Paper
VIDEO
How to Thaw Cells in a Cryopreserved Leukopak
This video closely follows the technical protocol and shows step-by-step instructions for properly thawing cells in a cryopreserved leukopak, which is crucial to ensuring the viability and functionality of the cells.
Why Use Frozen Leukopaks as Cellular Starting Material?
The stability of starting materials and immune cells is crucial for the success of any immunotherapy product. Cryopreservation is a valuable tool in managing the variability of starting material stability, ensuring viability and potency are preserved and providing researchers with a dependable source of cellular starting material for advanced cell and gene therapies.
| Format | Viability | Recovery | Consistency | Long Haul Shipping | Flexibility | Scalability |
|---|---|---|---|---|---|---|
| Fresh | ||||||
| Cryopreserved |
Benefits of Cryopreserved Leukopaks: A Viable Alternative to Fresh Leukopaks
-
Starting Material Management
Frozen leukopaks meets the demand for readily accessible cellular starting material, addressing concerns related to sourcing donors.
-
Quality and Viability Preservation
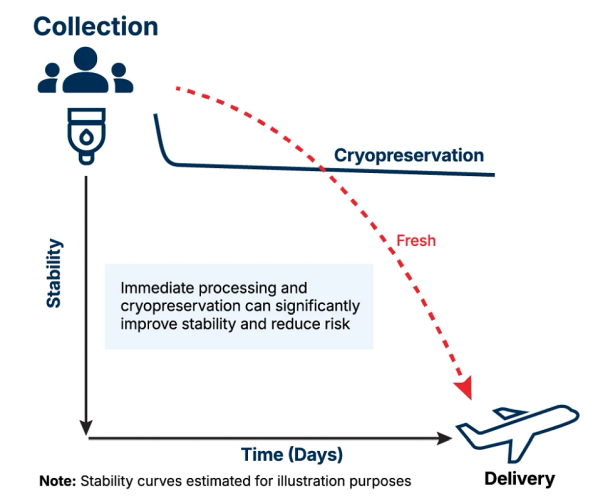
Maintaining optimal function of key raw materials used in the development of next generation cell-based products is of vital importance. Cryopreservation performed immediately after collection prolongs a product's shelf life and stability, maintaining the quality, viability, and functionality of the cells until they are required. Our frozen leukopaks are processed shortly after collection using our proprietary cryopreservation process, thus extending the shelf life and stability of the product and preserving cell viability and functionality until the cells are needed.
-
Shipping Logistics
Shipping delays may happen, especially with international deliveries. Frozen leukopaks provide a solution to the challenges of shipping and storing leukopaks by extending their durability during transit. Mitigate risk and simplify logistics with frozen leukopaks to ensure product quality and efficacy, and safeguard against unexpected delays during transport.
-
Downstream Processing Consistency
Maintain control over your project planning. Frozen leukopaks allows flexibility when planning and coordinating downstream processing activities, giving you control and access to your cellular starting materials when you’re ready to use them.
-
Staff and Project Scheduling
Scheduling can be challenging when working with fresh materials. On the other hand, frozen leukopaks offers greater flexibility when planning and coordinating subsequent processing tasks, enabling teams to confidently adhere to project timelines.
-
Supply Allocation
Effectively managing the materials needed for processing is crucial. Poor planning may result in wasting valuable resources. When working with frozen leukopaks, you minimize material waste and are able to facilitate better planning and management of processing materials ahead of time.
GUIDEBOOK
How Much Do You Really Know About Leukopaks?
Find out what they are and why they are important for immune research and cell therapy development. Our comprehensive guide covers best practices, considerations, applications, and in-depth characterizations for leukopak collections.
Download Guidebook
Frequently Asked Questions (FAQs) About Cryopreserved Leukopaks
-
What is a cryopreserved leukopak?
Cryopreserved leukopaks are made by quickly freezing freshly collected leukopaks in CryoStor® CS10 freeze media using a controlled-rate freezer. The frozen leukopaks are then cryopreserved by storing them in the gas phase of liquid nitrogen at extremely low temperature of -196 °C. This cryopreservation process ensures that the leukopak maintains its quality for extended periods.
Prior to being frozen, leukopaks are obtained through leukapheresis, where white blood cells (WBCs) are extracted from peripheral blood. The freshly collected leukopak contains a high concentration of peripheral blood mononuclear cells (PBMCs), including NK cells, T cells, B cells, and monocytes.
-
Why should I consider using cryopreserved leukopaks rather than fresh leukopaks in my research applications?
You should consider using cryopreserved leukopaks in your research applications, because not only does cryopreservation play a crucial role in the long-term preservation of biological samples, thus ensuring their viability and functionality for future use, but you’ll have access to valuable samples whenever required and according to your timeline.
Cryopreservation of cellular starting material will help simplify complex logistics associated with therapy development thus mitigating risk and ensuring successful production of therapeutics.
Planning and coordinating activities throughout the discovery and development process can be complicated, especially with cell and gene therapy manufacturing. One of the primary challenges is that starting materials must be both high quality and available on demand.
Freshly isolated starting materials are often considered the gold standard, but that standard only holds true for a short time post-collection. Room temperature and cold storage (4 °C) will keep cellular starting materials stable for 24 – 72 hours post-collection, but longer shipping and storage times will significantly decline quality.
Even during the initial 24 – 72 hour post-collection period, samples will experience a loss in quality and cell viability. This is where cryopreservation is key for the stability of starting materials and delivery of successful cell-based therapies.
-
How are Charles River leukopaks collected and cryopreserved?
Our leukopaks are collected using the Spectra Optia® apheresis system into a collection bag containing ACD-A anticoagulant. All leukopaks are collected onsite from consented healthy donors and are cryopreserved in CryoStor® CS10 freeze media same day of collection (day 0) to ensure highest quality.
Same-day processing and cryopreservation of leukopaks soon after collection guarantees minimal room temperature exposure without freeze / thaw cycles. Ultimately ensuring stability, viability, recovery, and functionality of the immune cells after thaw.
-
What impact does granulocyte contamination have on the viability and functionality of mononuclear cells in a cryopreserved leukopak?
The presence of granulocytes in a frozen leukopak can have a negative impact on the consistent recovery of cells of interest. Granulocyte contamination has been associated with the reduction of cell counts within mononuclear cells, inhibition of T cell proliferation and activation, as well as the loss of regulatory T cell integrity. They also can impact the stability and functionality of those cells. Granulocytes can release DNA and lysosomal enzymes after freezing and thawing, leading to clumping and loss of cell viability.
Reducing granulocyte contamination in cryopreserved leukopaks is critical to ensuring a consistent, stable, and functional leukopak. In our experience, indicators of high leukopak quality include granulocyte concentration of <15% with most of our leukopak collections containing an average of 5% granulocytes or less.
-
How quickly can I expect to receive cryopreserved leukopaks after placing my order?
Timelines are often critical in research, so we understand logistical concerns regarding product availability. We will dispatch your order according to the shipping terms specified on the Product Order Form (POF) once you submit your request and after we have reviewed and confirmed it.
Domestic Orders / Next-day Delivery
• Via specialty couriers
• Via FedEx First Overnight by 8:30 AM
• Via FedEx Priority by 10:30 AM


