Microdialysis Technique
Microdialysis is a minimally invasive sampling technique that is used for continuous measurement of free, unbound analyte concentrations in extracellular fluid and was initially developed for CNS research. It can be performed in any tissue type to assess the physiological or pharmacological functions of biochemicals (e.g., neurotransmitters, hormones, glucose) or perform metabolite ID/metabolite profiling, or determine the distribution of new chemical entities within the body. Microdialysis sampling is an excellent way to model the pharmacokinetics of a given therapeutic compound.
The microdialysis technique involves the implantation of a semi-permeable membrane in the target issue. The hollow fiber membrane is connected to inlet and outlet tubing, and the probe is continuously perfused with a solution that resembles the sampled tissue of interest (e.g., artificial CSF). Molecules that are small enough to diffuse across the pores of the membrane will diffuse from the in vivo sampling site to the inside of the membrane across a diffusion gradient and will be collected at time points for analysis. While microdialysis was originally developed as a method to sample the CNS, it has been used in oncology tumor models.
Microdialysis for CNS Drug Discovery
This webinar describes how microdialysis can be used to assess pharmacokinetics and distribution of small molecules, endogenous large molecules and large molecule therapies.
Watch The Webinar
Microdialysis Services
-
Conventional Microdialysis
Microdialysis Probes
Microdialysis probes are used to measure local release of neurotransmitters, neurometabolites, and other small endogenous substances in freely moving or anesthetized rodents.Some of the molecules that are measured include:
- Monoamines
- Amino acids
- Trace amines
- Kynurenine metabolites
- Monoamine metabolites
- Cyclic nucleotides
- Polyamines
-
MetaQuant Dialysis – Pharmacokineics
MetaQuant Probes
Our MetaQuant probe provides truly quantitative dialysis, which saves time and money in the process. Typically, free-drug concentrations in tissue are calculated by measuring the total drug concentration, then subtracting the bound-drug, based on an in vitro protein binding calculation. Our proprietary MetaQuant probe allows for accurate measurement of actual free-drug concentrations, vs. inferred concentration based on standard calculations.Advantages include:
- No more anomalies between in vitro and in vivo data
- Fewer animals required for testing
- Fast turnover without extensive validation required
- Ability to track over time from the same animal and in combination with other techniques
-
Multiple Probes (PK/PD) and Combined Services
Overview
Our deep experience with microdialysis enables a wide range of options for combining various probes across a broad array of applications. We can customize assays by combining PK or PD microdialysis techniques or combining microdialysis with other methods such as behavior or EEG measurements. The primary applications of the multiple probe approach include measuring pharmacodynamic changes in neurotransmitters, modulators, and proteins in real-time, while collecting free-drug concentrations to build accurate PK/PD relationships. -
Push-Pull Microdialysis – Proteins / Peptides / Antibodies
Push-Pull CNS Probes
For recovery of larger molecules, push-pull microdialysis uses larger pore sizes in the dialysis membrane. Compared to conventional dialysis, push-pull allows the measurement of larger endogenous proteins, inflammatory mediators, peptides, and exogenously applied antibodies.Large molecules measured include:
- α-Synuclein
- β-Amyloid
- Antibodies
- Tau
- Cytokines
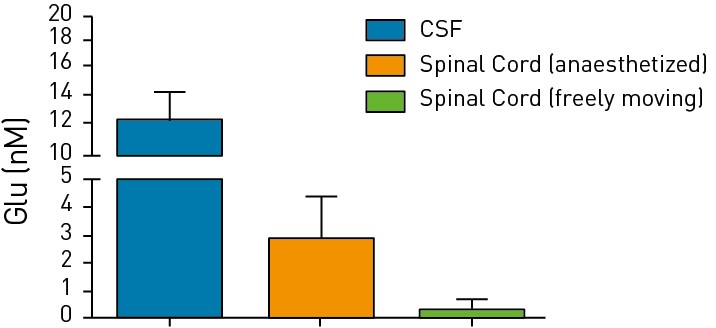
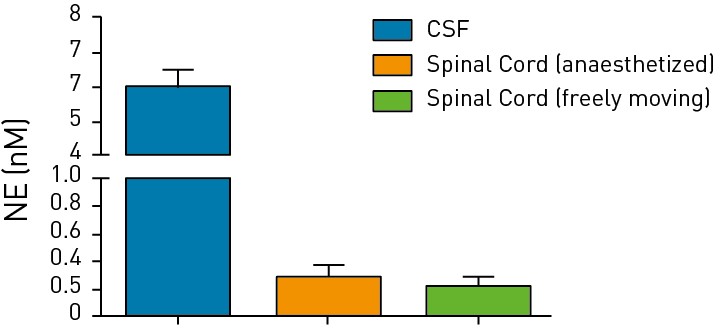
Figures 1 & 2: Measurement of Glutamine in the CSF or spinal cord of rodents via CSF sampling Norepinephrine
Measurement of neurotransmitters (glutamine and norepinephrine) in the CSF
and spinal cord of anaesthetized or freely moving rodents is shown.
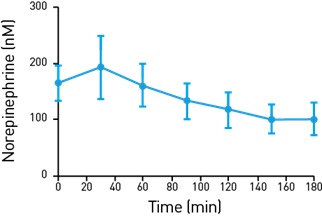
Figure 3: Measurement of Norepinephrine in Gut Tissues
Measurement of norepinephrine in gut tissues sampled using microdialysis is shown.
Apart from conventional microdialysis that sample small molecular weight analytes and support metabolite profiling, Charles River offers a push-pull method to sample high molecular weight analytes (peptides, proteins including tau and alpha-synuclein and interleukins and interleukins). The push-pull probes have a higher membrane cut-off and due to the bigger pores, a “pull” flow in the outlet is needed to ensure that the perfusion liquid is not lost along with the “push” flow in the inlet tube.
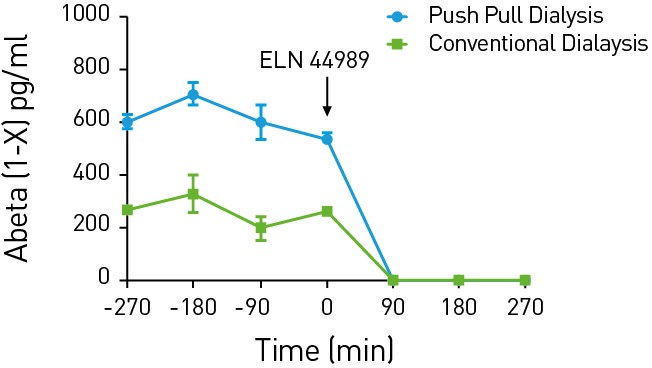
Figure 4: Measurement of Abeta in APP Transgenic Rodent Models
Conventional and push-pull microdialysis for Abeta peptides is compared. ELN 44989 is a nonselective gamma-secretase inhibitor.
The MetaQuant method is a proprietary microdialysis method that uses customizable probes to perform metabolite profiling and identification. developed by Brainlink. This method is used in various tissues including the GI tract, heart, liver, bone, kidney, lung, eye, and skin tissue, and is a modified regular microdialysis probe combining measurement of absolute levels in the tissue of interest with sampled volume collection equal to the regular microdialysis technique. Ultraslow flows are applied in this technique.
Microdialysis Resources
Check out our Microdialysis posters, publications, and other scientific resources…
Browse The Resources
Frequently Asked Questions (FAQs) About Microdialysis Services
-
Can this method be combined with other testing methods?
Yes, microdialysis is a noninvasive method where a probe is inserted to sample from the target tissue and the same animal can be used to perform behavioral studies or other assays.
-
Can this method be used to measure multiple endpoints simultaneously?
Microdialysis can be used to monitor a range of neurotransmitters, bloodborne biomarkers and most importantly, free drug concentrations in different tissues in the body as well as different compartments of the brain.
-
How can the recovered drug concentration be maximized?
One approach is to use the proprietary MetaQuant microdialysis method, that uses custom built probes for sampling combined with an ultra slow flow to maximize recovery of free drug concentrations or metabolites.