NCG Mouse Details
NCG mice are triple immunodeficient and lack functional/mature T, B, and NK cells, and have reduced macrophage and dendritic cell function. These animal models have the ability to host xenograft cells, tissue, and human immune system components.
This mouse model is ideal for oncology, immunology, tumor biology, infectious disease, graft-versus-host disease (GvHD), diabetes, regenerative medicine, hematopoiesis and tissue transplant studies. The NCG mouse model can also be utilized to create humanized mice by engrafting PBMCs or CD34 stem cells to closely mimic the human immune system.
The NCG mouse is the foundation for further strains and variants including:
- Pre-humanized CD34+ and PBMC NCG mice
- The NCG portfolio of NCG variant mouse models, capable of hosting human tissues and recapitulating the human immune system
Origin and CRISPR/Cas9*-Generated Mutation
Co-developed by Nanjing Biomedical Research Institute of Nanjing University and Nanjing Galaxy Biopharma in 2014 and transferred to Charles River in 2016. The NCG mouse model was created by sequential CRISPR-Cas9 editing of the Prkdc and Il2rg loci in the NOD/Nju mouse, generating a mouse coisogenic to the NOD/Nju.
The NOD/Nju carries a mutation in the Sirpa (SIRP α) gene that allows for engrafting of foreign hematopoietic stem cells. The Prkdc knockout generates a SCID-like phenotype lacking proper T-cell and B-cell formation. The knockout of the Il2rg gene further exacerbates the SCID-like phenotype while additionally resulting in a decrease of NK cell production.
Strain Highlights
- Proven robust engraftment of human immune cells and tumors
- Clean CRISPR knockout of Prkdc and Il2rg reduces the risk of spontaneous thymic lymphomas, extending study windows
- Competitive pricing – scale your studies without stretching budgets
- Strong availability – reliable supply for longer projects
- NCG variants – optimized for CAR-T, NK cell, and immuno-oncology studies
*CRISPR/CAS9 is used under licenses to granted and pending US and international patents from The Broad Institute and ERS Genomics Limited.
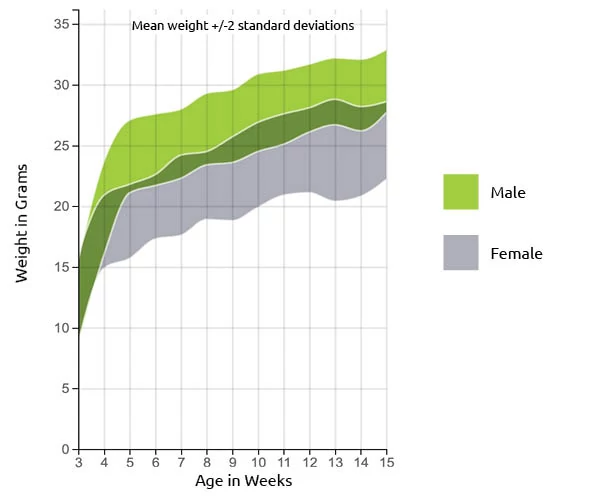.png?crop=false&position=c&color=ffffffff&u=fwtil1&w=450&h=622)
LOCATION: Hollister
UNIT: H60
Download the Report
LOCATION: Wilmington
UNIT: W08
Download the Report
➤ DOWNLOAD OUR CATALOG for instant access to Standard List Pricing
➤ TALK TO US to discuss organization or volume-based discounts
Already have an eCommerce portal account?* Login to order research models, obtain quotes, view organization-specific pricing, and see inventory. Processing time required to validate new eCommerce access requests.
*eCommerce is available in US, Canada, UK, France, Germany, Austria, Netherlands, Denmark, Finland, Norway, Sweden, Spain, Portugal, Belgium, Luxembourg, and Switzerland
Try out this model with our animal evaluation program
NCG Mouse Details
NCG mice are triple immunodeficient and lack functional/mature T, B, and NK cells, and have reduced macrophage and dendritic cell function. These animal models have the ability to host xenograft cells, tissue, and human immune system components.
This mouse model is ideal for oncology, immunology, tumor biology, infectious disease, graft-versus-host disease (GvHD), diabetes, regenerative medicine, hematopoiesis and tissue transplant studies. The NCG mouse model can also be utilized to create humanized mice by engrafting PBMCs or CD34 stem cells to closely mimic the human immune system.
The NCG mouse is the foundation for further strains and variants including:
- Pre-humanized CD34+ and PBMC NCG mice
- The NCG portfolio of NCG variant mouse models, capable of hosting human tissues and recapitulating the human immune system
Origin and CRISPR/Cas9*-Generated Mutation
Co-developed by Nanjing Biomedical Research Institute of Nanjing University and Nanjing Galaxy Biopharma in 2014 and transferred to Charles River in 2016. The NCG mouse model was created by sequential CRISPR-Cas9 editing of the Prkdc and Il2rg loci in the NOD/Nju mouse, generating a mouse coisogenic to the NOD/Nju.
The NOD/Nju carries a mutation in the Sirpa (SIRP α) gene that allows for engrafting of foreign hematopoietic stem cells. The Prkdc knockout generates a SCID-like phenotype lacking proper T-cell and B-cell formation. The knockout of the Il2rg gene further exacerbates the SCID-like phenotype while additionally resulting in a decrease of NK cell production.
Strain Highlights
- Proven robust engraftment of human immune cells and tumors
- Clean CRISPR knockout of Prkdc and Il2rg reduces the risk of spontaneous thymic lymphomas, extending study windows
- Competitive pricing – scale your studies without stretching budgets
- Strong availability – reliable supply for longer projects
- NCG variants – optimized for CAR-T, NK cell, and immuno-oncology studies
*CRISPR/CAS9 is used under licenses to granted and pending US and international patents from The Broad Institute and ERS Genomics Limited.
Not available
LOCATION: Les Oncins
UNIT: B131
For health report information, please contact us.
➤ DOWNLOAD OUR CATALOG for instant access to Standard List Pricing
➤ TALK TO US to discuss organization or volume-based discounts
Already have an eCommerce portal account?* Login to order research models, obtain quotes, view organization-specific pricing, and see inventory. Processing time required to validate new eCommerce access requests.
*eCommerce is available in US, Canada, UK, France, Germany, Austria, Netherlands, Denmark, Finland, Norway, Sweden, Spain, Portugal, Belgium, Luxembourg, and Switzerland
Try out this model with our animal evaluation program
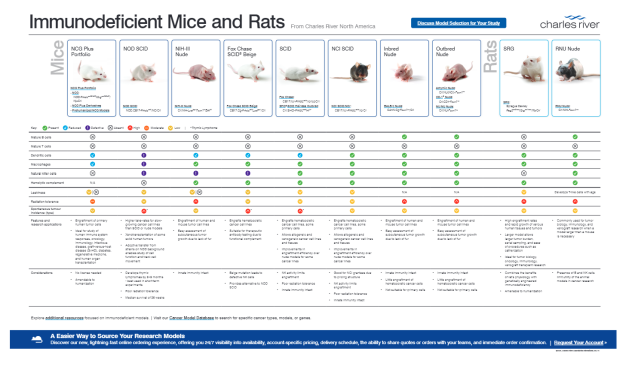
Immunodeficient Models Poster
This poster lists the major phenotypes of Charles River's immunodeficient mice and rats. It includes information on model features, degree of immunodeficiency, and gene functions.
Resources
- Technical Information
-
Posters
- The single mouse trial format predicts the sensitivity towards checkpoint
- Evaluation of in vivo anti-tumor response of solid tumors in a novel immune cell humanized NOD-Prkdcem26Cd52Il2rgem26Cd22/NjuCrl mouse model
- Combination Immune Checkpoint Inhibitors for the Treatment of Human Colon Carcinoma in hPBMC-NCG Humanized Mouse Model
- Humane end point refinement for total body irradiation and humanization of NCG mice
- NCG/CRL as a novel (Charles River) humanized mouse model for pre-clinical oncology studies: immunophenotypic characterization and performance monitoring
-
Brochure
Explore our NCG Portfolio brochure
-
Publications
- References
- Webinar
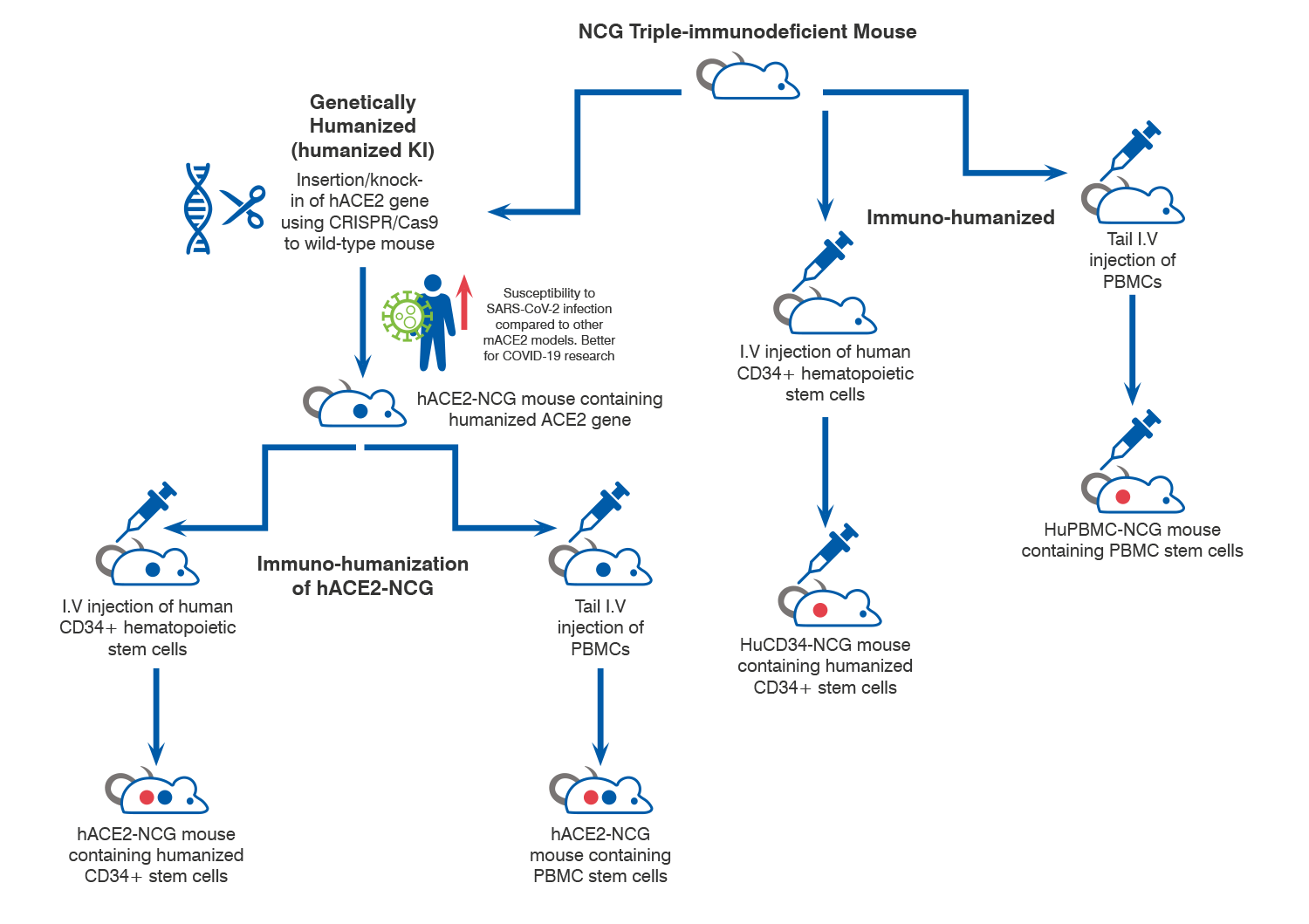
- Genetically vs. Immuno-Humanized Mice
Humanization
-
Our Humanization Portfolio
The NCG mouse is the foundation for the Charles River Hu-Mouse™ portfolio
-
Humanization Options
When your research requires humanized animal models, Charles River offers multiple options, both founded on the NCG triple-immunodeficient mouse. Note: NCG mice available in North America only.
Frequently Asked Questions (FAQs) about the NCG Mouse Model
-
How was the NCG mouse model created?
NCG mice were created using CRISPR/Cas9* technology.
*CRISPR/CAS9 is used under licenses to granted and pending US and international patents from The Broad Institute and ERS Genomics Limited.
-
What types of studies can NCG mice be used in?
These characteristics enable researchers to further study tumor biology and immuno-oncology, hematopoiesis, infectious disease, GvHD, and organ development and function because of their capability of hosting xenograft cells, tissue, and human immune system components.
-
What types of NCG humanized mice are available?
- Study-ready HuCD34-NCG mice
- Study-ready HuPBMC-NCG mice
You can also create your own humanized mice with our NCG/PBMC select humanization kit.
-
How can I evaluate a NCG mouse model?
Our Animal Model Evaluation Program helps researchers find the most appropriate model for their studies.
-
Are NCG mice good for the creation of humanized mouse models?
The NCG mouse model is the foundation of our humanized mouse model portfolio. NCG mice have a unique phenotype that allows it to host xenograft cells, tissue, and components of the human immune systems. It displays noticeable improvements of foreign tissue transplantation and engraftment, compared to previous generations of immunocompromised mice.
-
How should I house my immunocompromised animal?
We recommend isolators for housing immunocompromised animals.
WEBINAR
Advance Your Oncology and Immunology Research with Next-Generation Mouse Models
Wednesday, March 25, 2026 | 11:00 AM EDT
Learn how NCG immunodeficient and humanized mouse models advance oncology and immunology research, accelerating predictive humanized and PDX applications. Register Now