PET Imaging Studies
Preclinical PET imaging is a valuable and sensitive tool in neuroscience and oncology research studies to measure real-time in vivo brain or tumor functionality and metabolism changes and is increasingly being used as a preclinical endpoint to predict clinical success. Charles River’s expertise with small animal PET imaging studies and associated ex vivo techniques provides a comprehensive state-of-the-art toolkit for evaluating the pathophysiology and drug effects in animal models of neurological and oncological disorders.
Preclinical PET imaging studies employ radio-labelled ligands, also known as radiotracers, to image and quantify changes in functional physiological processes such as metabolism, neuroinflammation, and perfusion, as well receptor occupancy. A diverse range of radiotracers are available at Charles River to measure a wide range of physiological and cellular processes, including:
- 18F-fluorodeoxyglucose (FDG) for imaging changes in cerebral metabolism
- TPSO ligands, such as 18F-FEPPA, for imaging of neuroinflammation
- [18F]SynVesT-1 (SDM8) for imaging of synaptic density
- 18F-Flutemetamol and 11C-PiB for imaging of amyloid load in Alzheimer’s disease models
- Receptor-specific ligands for imaging of receptor expression or occupancy
- Custom labeling of therapeutic candidates to image distribution and target occupancy
-
PET Imaging Data Examples
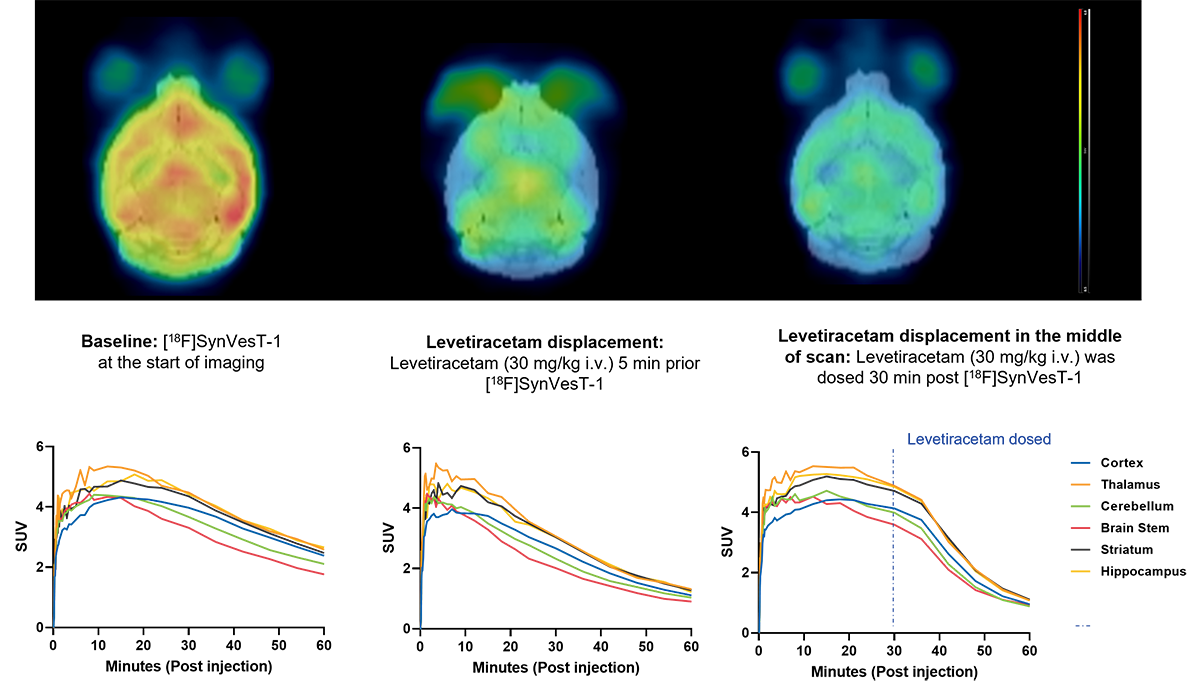
Figure 1: Imaging of synaptic density showing time course activity curves, and displacement of radioligand by levetiracetam.
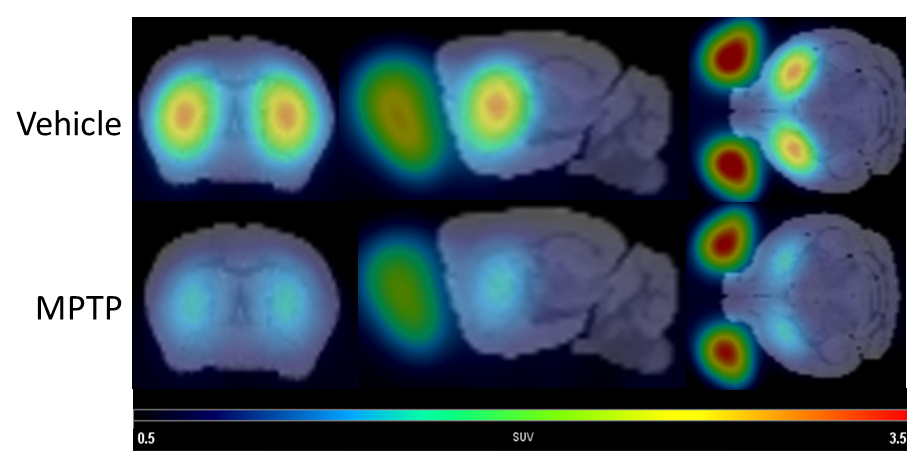
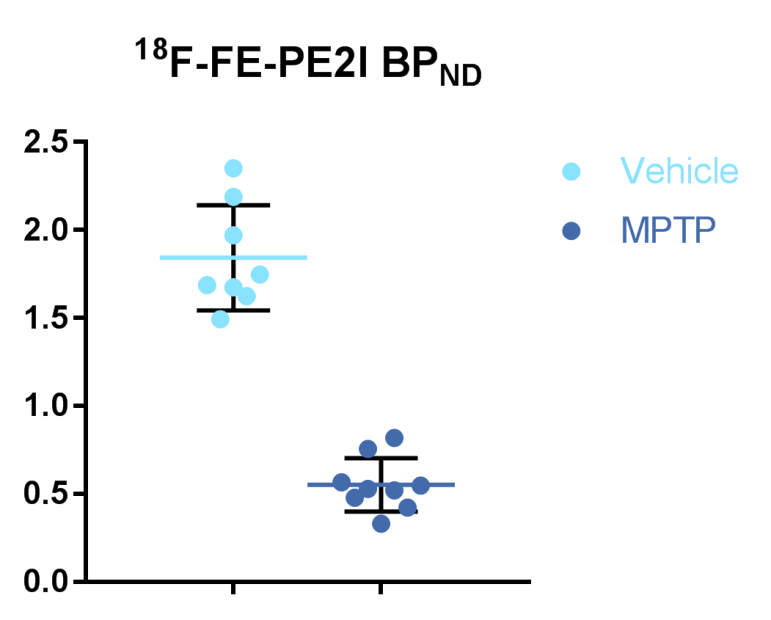
Figure 2: Chart data of reduction of dopamine transporter (DAT) levels in the MPTP model of Parkinson’s disease using the PET ligand 18F-FE-PE2I.
Preclinical PET imaging studies can be performed in combination with CT, enabling output of combined data on radiotracer accumulation and atomic localization. Additionally, arterial input function (AIF) can be generated to quantify a target area concentration that is normalized to cumulative arterial radioligand concentration from the time of injection until the end of the scan.
Noninvasive testing methods like our PET imaging services are easily combined with behavioral and biochemical readouts to provide translational preclinical data to support more optimally designed clinical trials that include PET imaging as a readout. At Charles River, we offer PET labeling and preclinical PET imaging services for therapeutic candidates including small molecules, antibodies, and cell therapies, using several radionuclides (68Ga, 89Zr and 18F).
PET Imaging in a Parkinson's Model
This poster outlines how PET imaging can be used in the MPTP Parkinson's model to examine dopamine receptor expression, neuroinflammation and brain metabolism.
View Poster
SPECT Imaging Studies
Preclinical SPECT imaging enables in vivo measurement of brain perfusion, inflammation, and biodistribution of novel compounds or cells. It is commonly used to study metabolic changes, cerebral blood flow, and oxygen levels across models of neurological diseases including Alzheimer’s disease and Parkinson’s disease. Preclinical SPECT imaging is often combined with CT scans and PET imaging to generate quantifiable data on therapeutic compound binding and brain function.
SPECT employs several gamma radioactive compounds, and various commercial ligands offer a straightforward method to follow pathological changes over time, or competitive binding of potential drugs. For example, technetium imaging (99mTc-exametazime (99mTc -HMPAO)) is a conventional method to assess cerebral blood flow in vivo and has been shown to correlate strongly with regional brain perfusion. This method is used in clinical nuclear imaging to detect stroke and other cerebrovascular diseases. Another well-studied application of SPECT/CT imaging is the application of 123Iodine beta-CIT as a diagnostic marker of Parkinson’s disease. It is used to analyze the condition of the striatum and to study the competitive binding affinity for presynaptic dopamine transporters (DAT) in the brains of mammals.
SPECT imaging can also be utilized in evaluation of therapeutic candidates. Charles River offers custom radiolabeling and SPECT imaging with several radionuclides (177Lu, 125I, and 111In).
Quantitative Real-Time Autoradiography Studies
Autoradiography is a well-established method used to assay metabolic changes, receptor activation, and G protein-coupled receptor (GPCR) signaling in response to compound stimulation in brain tissues. Non-invasive imaging methods are used extensively to study metabolic changes and alterations in ligand-receptor signaling in the brain, as a part of disease progression and in response to therapeutic intervention via small molecules or biologics. Autoradiography combined with behavioral readouts provides a comprehensive evaluation of neurological disease pathophysiology as well as mechanism of action data for therapeutic compounds.
Frequently Asked Questions (FAQs) for Preclinical PET and SPECT Imaging Services
-
Can nuclear imaging be performed along with other tests in the same animal?
Yes, one advantage of nuclear imaging is that it can be combined with other studies, such as behavioral tests, histology, and anatomical MRI. With PET and SPECT, we can also image the same animal several times during a study to follow disease progression, efficacy of test item, or biodistribution of antibody, for example.
-
What is small animal SPECT imaging used for?
SPECT imaging is a valuable translational tool to assess the impact of therapeutic compounds on inflammation, cerebral blood flow, and other neurophysiology processes in the brain. SPECT imaging can also be used to evaluate 177Lu-labelled therapeutic candidates, including small molecules, proteins, antibodies, and cell therapies.
-
What is a typical sample size for efficacy studies using SPECT and PET imaging?
Reference efficacy studies can be performed with a group of 9 animals for statistically significant data. For a novel compound study using preclinical PET imaging, the recommended sample size is at least 6 animals.
-
Does Charles River have access to a cyclotron? Can custom radiolabeling of molecules with 18F, radio-iodines, and radiometals be performed?
Charles River has access to a local cyclotron, and we have an agreement in place to synthesize custom ligands and perform radiolabeling with 18F. At our in-house radiochemistry lab, we can also develop and produce novel, protein, peptide, and cell-based ligands using 68Ga and 89Zr for PET imaging studies, and 177Lu, 125I, and 111In for SPECT and radiotherapy studies.
-
Can the distribution of supramolecular structures (e.g., liposomes, nanoparticles) be imaged?
Yes. We have done antibody- and peptide-based biodistribution and efficacy studies, and nanoparticle and cell distribution studies.
-
When biochemical analyses are performed on postmortem samples from animals injected with radioactive tracers, are these analyses conducted in a lab certified for isotope work or are the samples stored in quarantine until the isotope decays?
It depends on the half-life of the isotope. It is possible to store samples that have shorter half-life isotopes before performing additional analyses, but in cases where the isotope half-life is very long (such as 14C), the sample will need to be analyzed in a lab certified for isotope work.
-
Why is autoradiography important?
Traditionally, autoradiography has been done by exposing samples on films or phosphoscreens – a process which takes days or months, depending on the radioisotope being employed. Now we can use real-time imaging of radioisotopes, and quantitative data can be obtained within hours of samples being scanned. The combination of these assays provides a powerful tool to comprehensively measure responses in disease models to novel molecules quickly and cost-efficiently.