Cell Line Characterization Methods
Cell line characterization is performed for Master Cell Banks (MCB), Working Cell Banks (WCB), and Cells at the Limit of In Vitro Cell Age (LIVCA) . Furthermore, Research Cell Banks (RCB) are commonly checked for identity, sterility, and freedom of mycoplasma before they are introduced into GMP facilities for MCB manufacturing. Manufacturers may also request adventitious virus tests for this purpose.
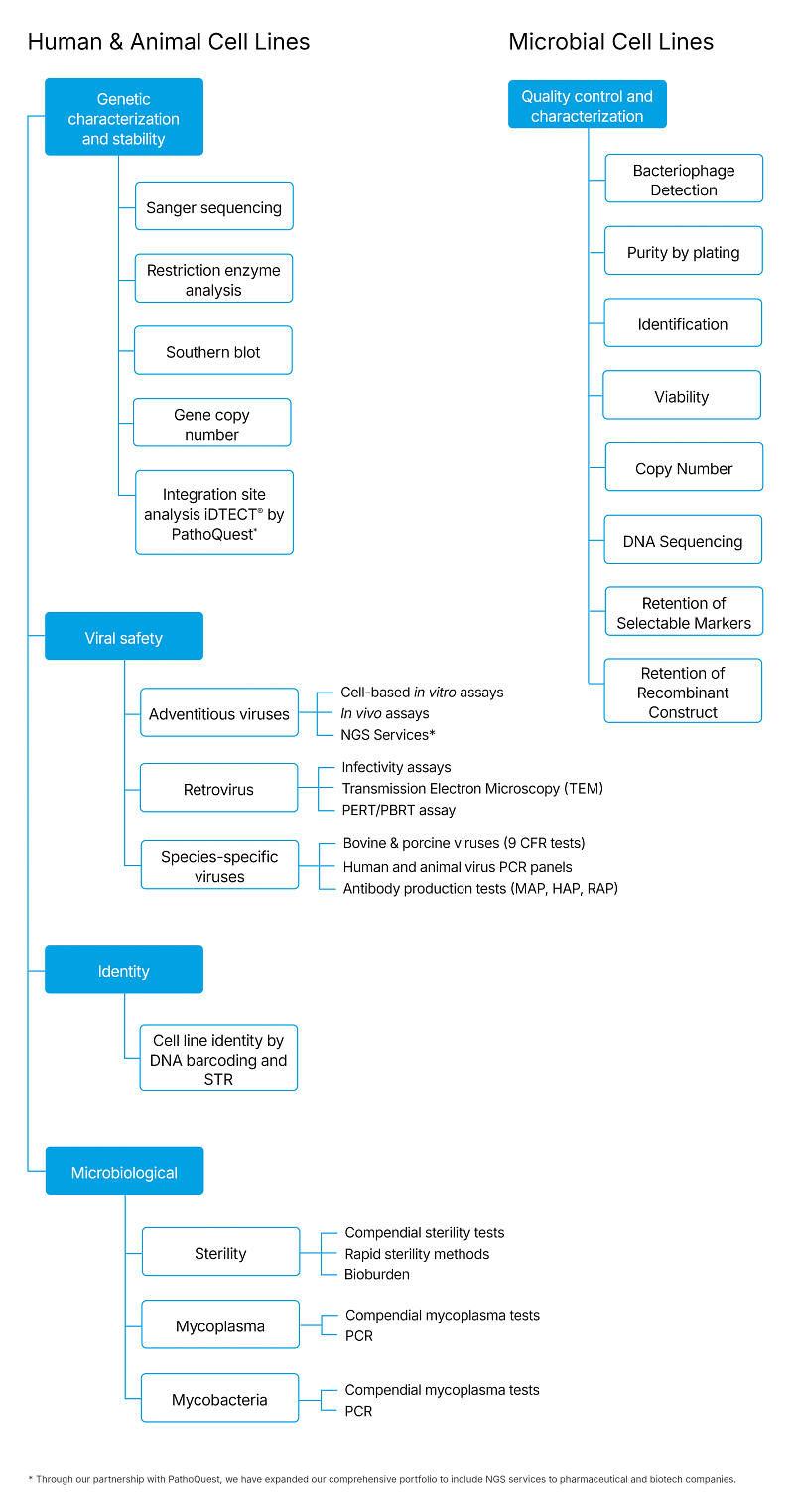
According to current regulations (e.g., ICH Q5A/Q5B/Q5D), the following tests should be considered for the characterization of animal and human cell lines:
- Identity
- Morphology and growth characteristics
- Sterility
- Mycoplasma tests
- Mycobacteria tests
- Endogenous virus (e.g., retroviruses) tests
- Adventitious virus tests
- Specific virus tests
- Genetic stability
- Tumorigenicity and oncogenicity
Using NGS to Characterize Cell Lines for Production of Biologics
View our on-demand webinar to learn about the use of the latest sequencing technologies for the characterization of your cell lines.
Watch Now
Cell line characterization uses a risk-based approach. Considerations also include species of origin of the cell line (e.g., hamster, human) and its intended use (e.g., recombinant protein production, vaccine production, cell and gene therapy). Furthermore, the source of the cell line and its development history, as well as the biological properties of the cells, determine which panel of tests is required. Our cell line characterization assays are generically validated according to ICH Q2 guideline to demonstrate their suitability for the intended purpose.
Cell Banking & Characterization eBook
Your biologic can only be as successful as the cells behind them. Learn about our full spectrum of CGMP cell banking and cell line characterization services.
Download Now
Tests to be Considered for Cell Line Characterization
The tables below list tests which should be considered for the characterization of cell substrates used to produce recombinant therapeutic proteins.
-
Characterization of Human Cells
Human Cell Line Characterization Panel MCB WCB LIVCA Cell Line Identity DNA Barcoding X X X DNA fingerprinting/STR X X Karyology (for diploid cells) X X Microbial Contamination Sterility X X X Mycoplasma X X X Mycobacteria X Retroviruses Reverse transcriptase X X Transmission electron microscopy X X In vitro adventitious viruses X X X NGS iDTECT® Transcriptome Assay or in vivo adventitious virus tests X X Adventitious Viruses Tests for specific viruses according to risk assessment. E.g.
- Bovine viruses
- Porcine viruses
- Human viruses
X X Genetic Characterization and Stability1 DNA sequencing X X Gene copy number X X Restriction endonuclease mapping analysis by Southern blot X X 1Genetic stability involves the comparison of the MCB and the LIVCA cells and includes qPCR for the determination of the gene copy number and by restriction endonuclease mapping analysis by Southern blot in addition to sequencing of the coding region.
-
CHO Cell Line Characterization
CHO Cell line Characterization Panel MCB WCB LIVCA Microbial Contamination Sterility X X X Mycoplasma X X X Cell Line Identity DNA Barcoding X X X Retroviruses Reverse transcriptase1
Transmission electron microscopy
Infectivity assaysX X In vitro adventitious viruses X X NGS iDTECT® Transcriptome Assay or in vivo adventitious virus tests3 X X Adventitious viruses Tests for specific viruses according to risk assessment. E.g.
- Bovine viruses
- Porcine viruses
- Minute virus of mice
X X NGS iDTECT® Transcriptome Assay or Antibody Production Tests in Mice and Hamsters (MAP and HAP) X Genetic characterization and stability2 DNA sequencing X X Gene copy number X X Restriction endonuclease mapping analysis by Southern blot X X 1CHO cells are known to express an endogenous retrovirus and are positive for Reverse Transcriptase activity. A test for Reverse Transcriptase activity might not be needed.
2Genetic stability involves the comparison of the MCB and the LIVCA cells and includes qPCR for the determination of the gene copy number and by restriction endonuclease mapping analysis by Southern blot in addition to sequencing of the coding region.
3According to ICH Q5A test may not be needed based on prior knowledge and other risk-based considerations.
-
Microbial Cell Line Characterization
E. coli Cell Line Characterization Panel MCB WCB EPC Identity Viability X X X Identification X X X Plasmid DNA Sequence X X X Plasmid Copy Number X X X Retention of Selectable Markers X X X Retention of Recombinant Construct X X X Restriction Endonuclease Analysis X X X Microbial contaminants and bacteriophages Microbial purity by plating X X X Bacteriophage detection X X X
With all of these things to consider, it is important to determine a regulatory compliant characterization strategy which is most effective and efficient.
How can we support your program?
Manufacturing & Virus Safety Testing of HEK293 Cells for Viral Vector Production
Whether you're in the early stages or near commercialization, it's important to understand HEK293 cells' potential for CGTs and what you need to consider when banking them. Learn what manufacturing and viral safety testing you should consider to meet regulatory guidelines.
Watch the Webinar
Frequently Asked Questions (FAQs) for Cell Line Characterization
-
What is cell line characterization?
The objective of cell line characterization is to confirm the suitability of the cell substrate for production. The identity, genetic stability, and tumorigenicity of the cells must be demonstrated. Cells must also be tested for the presence of microbial contaminants like bacteria, fungi, mycoplasmas, and viruses. The specific characterization strategy is commonly a result of a risk assessment which considers several factors including, but not limited to, the origin of the cell line, the developmental history, and the potential contaminants associated with the involved cell donors and raw materials.
-
Why is cell line characterization important?
Proper cell line characterization is crucial to ensure the quality and safety of biopharmaceutical products. Contaminations by adventitious agents like viruses, bacteria, fungi or mycoplasmas can affect the safety of a biopharmaceutical product. Each quality issue could have major consequences: failed lots and costly delays and product shortages.
-
What are the regulatory requirements for cell line characterization?
Several factors must be taken into consideration in terms of regulatory requirements, such as the product type (e.g., antibody, viral vaccine, etc.), the cell line is being used to manufacture and the geographic region where the sponsor intends to seek commercial licensure (e.g., US, EU, Japan, etc.).
Examples of guidance documents from various regulatory authorities that may be useful include:
- Points to Consider in the Characterization of Cell Lines Used to Produce Biologicals (FDA)
- Guidance for Industry for Characterization and Qualification of Cell Substrates and Other Biological Materials Used in the Production of Viral Vaccines for Infectious Disease Indications (FDA)
- ICH Q5 series (specifically Q5A, Q5B, and Q5D)
- Technical Report Series, No. 878, Annex 3, Recommendations for the evaluation of animal cell cultures as substrates for the manufacture of biological medicinal products and for the characterization of cell banks (WHO)
- 9 CFR § 113.52 Requirements for cell lines used for production of biologics
- 9 CFR § 113.46 Detection of cytopathogenic and/or hemadsorbing agents
- 9 CFR § 113.47 Detection of extraneous viruses by the fluorescent antibody technique