Why Purchase Fresh Leukopaks from Charles River?
- Research Use Only (RUO) and GMP leukopaks to support clients from discovery to manufacturing
- Low granulocyte and low red blood cell contamination
- High mononuclear cell viability
- Our donor center is registered with the FDA, certified by AABB and CLIA, and is state-licensed
- Our large, diverse, and recallable donor network can support even the most stringent inclusion and exclusion research criteria
Explore Fresh Leukopak Products from Charles River
-
Normal Fresh Leukopaks
Leukopaks Size Product Code Fresh 1 Bag PB001F-1* 1/2 Bag PB001F-2* 1/4 Bag PB001F-3* Contact us if you don’t see a specific product, size, or format, or would like to customize your request.
*Product is made to order.Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Disease State Fresh Leukopaks
Learn more about our Disease State Program and elevate your disease state research with our high quality, well-characterized Research Use Only (RUO) human disease state fresh leukopaks obtained from patients selected based on your specified inclusion / exclusion criteria (I/E). Explore Disease State Fresh Leukopaks
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
Mobilized Fresh Leukopaks
Our mobilized fresh leukopaks are derived from peripheral blood mobilized using G-CSF (Filgrastim or FDA-approved biosimilars), Plerixafor, or a combination of both. In addition to selecting the preferred mobilization agent(s), clients have the option to choose from different dosing regimens, including varying durations and collection days. Explore Fresh Mobilized Leukopaks
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
-
GMP-compliant Fresh Leukopaks
We offer two choices for our GMP-compliant fresh leukopaks: a standard protocol option (CliniPrime™ Fresh Leukopaks) to expedite collection, and a customizable option (GMPrime™ Fresh Leukopaks) tailored to your program's unique requirements. Both options ensure compliance with regulatory standards from process development to commercialization. Explore GMP Fresh Leukopaks
Your company or academic institution may be eligible for pricing discounts. Government labs located in the United States are also eligible for GSA MAS discounts. Request a quote to find out more.
GUIDEBOOK
How Much Do You Really Know About Leukopaks?
Find out what they are and why they are important for immune research and cell therapy development. Our comprehensive guide covers best practices, considerations, applications, and in-depth characterizations for leukopak collections.
Download Guidebook
Fresh Leukopak Studies Conducted by Charles River
-
Fresh Leukopak Stability Study
See insightful data comparing stability of the different PBMC populations (T cells, NK cells, B cells and monocytes) in fresh leukopaks, at different storage temperatures over time. A comparison of WBC viability and recovery between fresh and cryopreserved leukopaks is also provided. Download Whitepaper
-
Fresh Leukopak Characterization Study
To gain a better understanding of the amount of variability associated with fresh leukopak collections as a result of donor-to-donor inherent differences, we carried out an in-depth analysis of a representative sample of our fresh leukopak collections drawn from healthy donors. Read more about it in our whitepaper: A Comprehensive Analysis of Fresh Apheresis Collections: Conclusions and Best Practices
A summary of the results for total apheresis collection volume, total blood volume processed, and white blood cell yields are summarized in the data below:
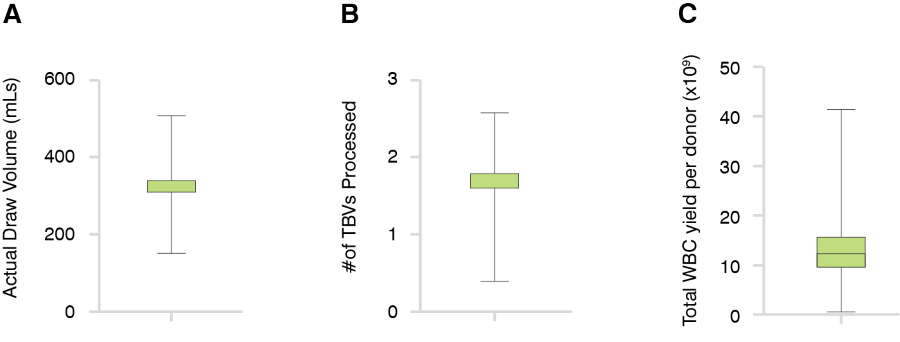
Figure 1. Analysis of Charles River’s Fresh Apheresis Collections: In a cohort of 2,490 fresh apheresis collections, our apheresis centers collected total draw volumes varying between 150 mLs-508 mLs, with a mean of 318 mLs drawn including plasma added (A). These collected draw volumes result from processing a mean of 1.64 total blood volumes (TBV) per donor (B). Total WBC yields varied between 0.5-40.1 billion white blood cells/donor, with a mean yield of 12.7 billion cells/donor (C).
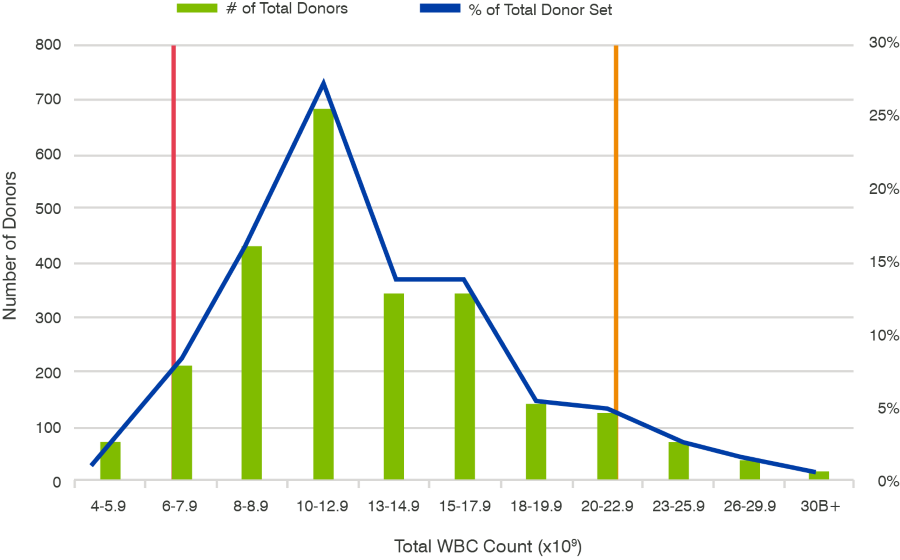
Additional analysis of white blood cell (WBC) yield frequency in these fresh leukopaks was performed as shown in Figure 2. Analysis of white blood cell yield and frequency in 2,490 fresh leukapheresis collections showed a mean yield of 12.7 billion WBCs/donor and 5th (light blue line) and 95th (purple line) percentiles of 6.4 billion and 22.9 billion WBCs/donor. Despite wide variance, the majority of data points cluster near the mean.
WBC counts are used by physicians as an indicator of potential disease presence. Abnormally high WBC counts, for example, can indicate infection, inflammation, or certain types of cancer, while an abnormally low WBC count can indicate viral infections, cancer, or autoimmune disorders. Therefore, WBC yields outside of the normal range may indicate the donor is impacted by illness. Higher WBC yields within the normal range, on the other hand, generally indicate a higher number of therapeutic target cells. Because healthy donors have a wide distribution in WBC cell counts, exercise caution when selecting for a specific range.
Figure 2. WBC Yield Analysis: Analysis of white blood cell yield and frequency in 2,490 apheresis collections with mean yield being 12.7 billion cells/donor and 5th (red line) and 95th (orange line) percentiles of 6.4 billion and 22.9 billion cells/donor.
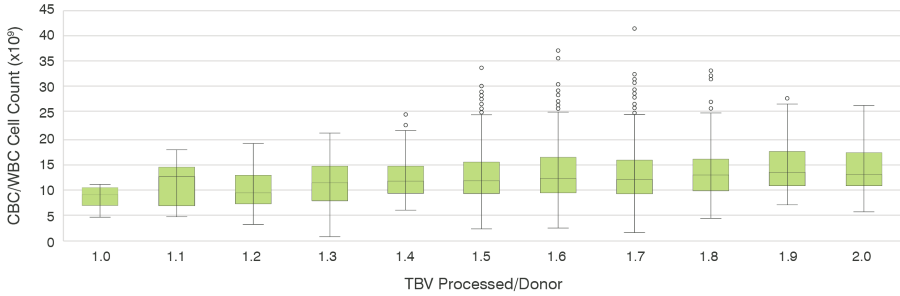
We also assessed the WBC count for varying blood volumes of apheresis collections processed (Figure. 3). Our donor centers process roughly between 1.5-1.8 total blood volumes (TBV) in the majority of donations, which is equivalent to 8-9 liters of blood in the majority of donations. As shown in Figure 3, there is no significant variance in WBC counts in apheresis collections varying between 1.0 to 2.0 processed TBVs. That WBC recovery is relatively consistent at nearly all blood volume ratios suggests that the TBV processed does not play a major role in total WBC yield per apheresis unit. Importantly, increasing TBVs processed beyond an ideal point, especially using suboptimal collection parameters, may lead to an increase in undesirable cellular subsets due to increased physical stress on the donor as well as donor discouragement for repeat donations.
Figure 3. Complete Blood Count (CBC)/WBC Count vs Total Blood Volume (TBV) Processed: Mean (X) and median (line within bar) indicate the WBC counts observed for each TBV processed. Bars indicate standard range of values with error bars and major outliers present as points outside of the error bars.
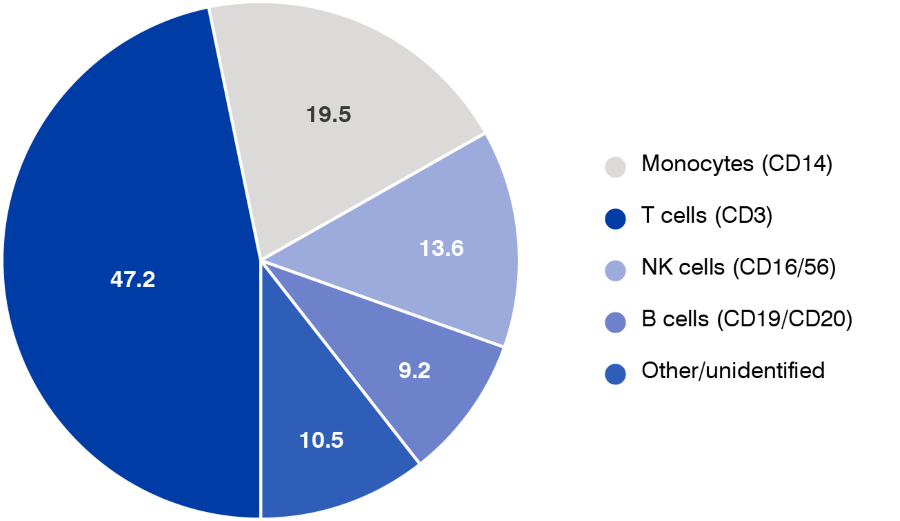
As part of the fresh leukopak characterization study, we also analyzed a smaller subset of 70 fresh leukopaks for which flow cytometry had been performed to assess the frequency/yield of key target cell populations. Results of the average percentage of major WBC subpopulations are shown in Figure 4. Within the examined 70 cohort cell collection, the largest average percentage of cells, 47.2%, were identified as T cells, while monocytes comprised and average of 19.5%, NK cells 13.6%, and B cells 9.2%, with an additional 10.5% of cells not clearly identified.
Figure 4. WBC Subpopulations (percent total): Mean percentages of individual WBC types as measured by flow cytometry.
Frequently Asked Questions (FAQs) About Fresh Leukopaks
- What is a fresh leukopak?
-
How are Charles River fresh leukopaks collected?
Fresh leukopaks are collected via Terumo's Spectria Optia® apheresis system using continuous flow centrifugal technology directly into a sterile collection bag containing ACD-A anticoagulant. All fresh leukopaks are collected using our Continuous Mononuclear Cell (CMNC) collection protocol, and collections are closely managed by an experienced apheresis expert to ensure that the balance of purity and yield is achieved. Fresh leukopaks are obtained from healthy donors who have given their consent under an IRB-approved protocol.
-
How many cells are in a fresh leukopak?
Charles River fresh leukopaks typically range between 6.4 – 22.9 billion white blood cells (WBCs) per collection. WBC counts are dependent on the donor. The average collection for a fresh leukopak yields arounds 10 – 12 billion WBCs.
Aside from white blood cells, Charles River fresh leukopaks also contain plasma, platelets, and a small amount of red blood cells. Fresh leukopaks typically have a composition of approximately 47% T cells, 20% monocytes, 14% natural killer cells, 9% B cells, and 10% other cell types such as granulocytes, platelets, and hematocrit cells.
-
What is the volume of granulocytes in a Charles River fresh leukopak?
On average, Charles River fresh leukopaks have <5% granulocytes. See the detailed findings of our comprehensive investigation into fresh leukopaks, including factors that affect yield, purity, sourcing and variability, by reading our white paper: A Comprehensive Analysis of Fresh Apheresis Collections: Conclusions and Best Practices.
-
How quickly can I expect to receive fresh leukopaks after placing my order?
Timelines are often critical in research, so we understand logistical concerns regarding product availability. We will dispatch your order according to the shipping terms specified on the Product Order Form (POF) once you submit your request and after we have reviewed and confirmed it.
Domestic Orders / Next-day Delivery
• Via specialty couriers
• Via FedEx First Overnight by 8:30 AM
• Via FedEx Priority by 10:30 AM
We also offer fresh leukopak same-day local delivery in the Los Angeles area. -
Which Charles River leukopak is most suitable for my study?
Select the best leukopak for your study from our comprehensive Charles River product portfolio, which includes fresh leukopaks, cryopreserved leukopaks, mobilized leukopaks, disease state leukopaks, and GMP leukopaks.