2,000+
scientists
100
preclinical drug candidates
33%
reach clinical proof of concept
Comprehensive Biomarker Services for Confident Decision Making
From early discovery through clinical development, our biomarker services help you make more informed decisions with speed and scientific precision. We offer end-to-end biomarker support that aligns with your therapeutic goals, leveraging global platforms, advanced technologies, and regulatory insight to streamline development and reduce risk. Whether you need custom assay development, validated bioanalysis, or multi-omic integration, our experienced teams design biomarker strategies that translate across preclinical and clinical phases, so your data drives progress, not delays.
Integrated Biomarker Strategies Across the Drug Development Lifecycle
Biomarkers aren’t confined to a single study or phase – they’re decision-driving tools that impact every step of development, from discovery through clinical submission. We apply a biomarker strategy across multiple disciplines to ensure that what’s measured adds value, simplifies timelines, and improves outcomes. Whether it’s identifying early signals, confirming safety, or supporting regulatory filings, our biomarker services are embedded where they can make the greatest impact.
The applications below highlight where and how our biomarker expertise is delivered in action – from early discovery and toxicology to clinical immunogenicity and trial support. This integration ensures that biomarker data isn’t siloed, but translated into insight across programs, platforms, and therapeutic areas.
Discovery and Early Development Insights
Accelerate early-stage research with integrated biomarker discovery and validation solutions.
Safety and Nonclinical Assessment
Ensure compound safety through comprehensive biomarker-driven toxicology and pharmacology studies.
Translational Biomarker Strategy
Bridge preclinical and clinical phases with biomarkers that inform decision-making and reduce risk.
Clinical Development
Support clinical trials with validated biomarker assays for efficacy, safety, and patient stratification.
Biomarkers for Translatability from Drug Discovery to the Clinic
This webinar explores strategies to deploy early biomarker candidates in lead selection and preclinical pharmacology for early de-risking of drug development programs and setting up for success in clinical development.
Watch now
Scientific Partnership Backed by Proven Results
Our biomarker services don’t just deliver data – they deliver clarity, speed, and confidence at every stage. Our clients trust us to navigate complex development challenges and support biomarkers that translate across studies, phases, and endpoints.
We can accelerate the journey from early biomarker discovery to clinical readiness, ensuring a timely path to clinical trials through an wholistic bioanalytical support lens. You may consider expanding the discussion to ensure we cover additional aspects of your program."
Vimal Patel, MS, MBA, Senior Director, Global Lab Sciences
Advanced Biomarker Technologies for Success
We offer a comprehensive suite of biomarker technologies designed to support drug development from discovery through clinical trials. Our platforms enable precise measurement of immune responses, gene expression, and protein biomarkers, providing critical insights into therapeutic efficacy and the mechanism of action.
By integrating these advanced technologies, we facilitate the identification and validation of biomarkers that are essential for informed decision-making and regulatory submissions. Our global network ensures standardized methodologies and consistent data quality across all study phases.
ELISpot Assays
ELISpot assays are immunoassays that quantify the frequency of antigen specific T and B cells by measuring cytokine-secreting cells from blood samples.
Flow Cytometry Assays
Flow cytometry assays serve as valuable exploratory endpoints in in vitro animal studies or clinical trials by assessing how a therapy affects its intended cellular targets.
Multiplex Cytokine Assays
These assays enable simultaneous measurement of multiple cytokines, offering insights into immune response modulation.
NanoString RNA Analysis
We employ NanoString nCounter® technology to analyze changes in gene expression and help identify or validate biomarkers.
qPCR Gene Expression Profiling
Using quantitative PCR, we provide gene expression profiling to pinpoint biomarkers tailored to your research or program needs.
Genomics Services
Our genomics offerings include integrated gene expression services such as Next-Generation Sequencing (NGS), DNA microarrays, RT PCR, and multiplex assays.
OMICS
We offer a full spectrum of omics technologies, including transcriptomics, genomics, epigenetics, proteomics, metabolomics, lipidomics, and immunomics. High-Resolution LC/MS, advanced sequencing, Retrogenix, and specialty assays are supported by expert data analysis in partnership with Fios Genomics.
eGuide: Biomarker Solutions from Discovery to Clinical Trials
Learn how strategic biomarker integration can accelerate research & de-risk pipelines to bring life-saving therapies to patients faster. This guide helps you uncover smarter ways to de-risk studies and accelerate timelines, without compromising scientific rigor.
Read the Guide
Strategic Biomarker Solutions Driving Confident Decisions
Biomarkers are pivotal in transforming drug development, offering insights that drive go/no-go decisions and streamline regulatory pathways. However, challenges like fragmented data, inconsistent methodologies, and delayed analyses often hinder their effective utilization.
We address these challenges by providing integrated biomarker solutions that combine scientific expertise, advanced technologies, and global standardization. Our approach ensures that biomarkers are not only identified but also effectively translated from discovery through clinical development, accelerating timelines and enhancing data reliability.
Custom Assay Development
Tailored assays aligned to your targets, sample types, and regulatory needs.
Global Laboratory Harmonization
Standardized methods and protocols across sites for consistent, reproducible data.
Apollo™ Data Portal
Real-time access to study data, trends, and dashboards — anywhere, anytime.
Strategic Scientific Consultation
Expert advisory from design through delivery to align biomarkers with endpoints.
Regulatory-Ready Execution
Data packages and documentation built to meet global health authority expectations.
Scalable Solutions
Flexible services that adapt as your study progresses and grows in complexity.
Lead Time Advantage
Fast project startup, efficient scheduling, and optimized global site strategy to meet timelines.
Rapid Turnaround Times (TAT)
Streamlined workflows ensure faster data delivery and milestone decisions.
Operational Excellence
Experienced teams and modality-specific expertise safeguard study quality and consistency.
Multi-Omic Integration
Combine genomic, proteomic, and immunologic data to drive deeper insights.
How Modern CROs Accelerate Your Drug Development
Are you facing delays, bottlenecks, or rising costs in your drug development process? In this webinar, you will learn how leading CROs overcome these challenges with innovative strategies. Gain insights that can directly impact your timelines, reduce risks, and streamline your path to approval.
Watch the Replay
Scientific Depth and Technical Versatility Across the Biomarker Lifestyle
Our biomarker capabilities are built to deliver scientific continuity from target discovery to clinical trials. Our teams provide support across all sample types, apply validated platforms, and integrate omics, imaging, and pathway-level data interpretation, ensuring biomarker relevance across therapeutic areas and timepoints.
Sample Types and Matrix Expertise
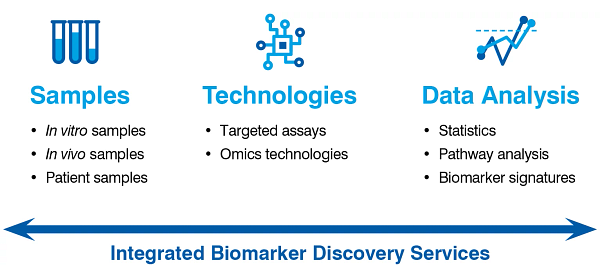
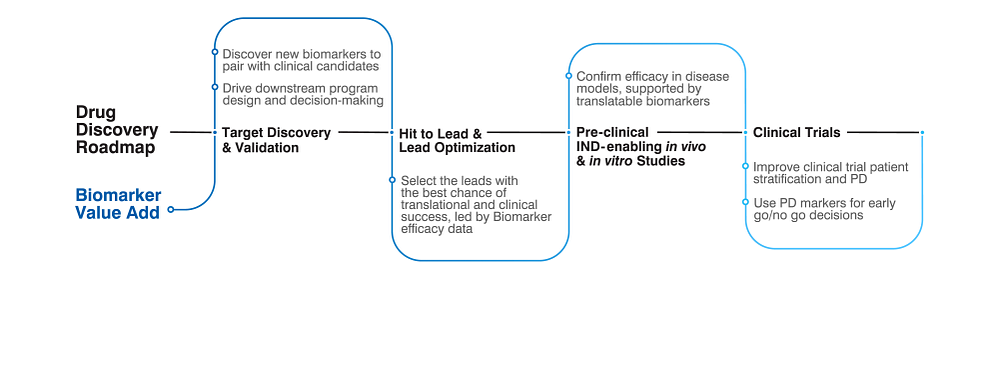
Drug Development Integration
How early do you start thinking about Biomarkers in your drug discovery pipeline?
Frequently Asked Questions (FAQs) for Biomarker Testing Services
-
How can biomarker testing services add value to my program?
Biomarker testing serves as a critical tool in drug discovery and development. The ability to develop biomarkers in animal models and translate them to the clinic can greatly accelerate candidate development as well as decrease associated risks and timelines.
-
For which therapeutic areas do you offer biomarker analysis?
We offer a comprehensive range of biomarker services in key therapeutic areas and safety assessment. From early discovery stages to clinical support, our scientists have the expertise to relate biomarkers to every stage of a drug development program. We employ multidisciplinary teams of experts in oncology, inflammation, immunology, infectious disease, ocular disease, metabolism, and diseases of the cardiovascular, renal, respiratory, and central nervous systems.
-
How does Charles River’s Apollo™ enhance the biomarker data management process?
Charles River’s Apollo™ is our proprietary data portal that offers clients real-time access to study data, facilitating efficient decision-making and streamlined project management.
-
How does Charles River ensure the quality and consistency of biomarker data across global sites?
We implement standardized protocols and rigorous quality control measures across all our laboratories to ensure consistent and reliable biomarker data, facilitating seamless integration into your development programs.







