Immunogenicity Assay Development
Evaluating the immune response in preclinical models through clinical samples, immunogenicity testing is mandatory throughout the development process. Whether you’re developing a vaccine, biologic or gene therapy, immunogenicity testing measures the antibodies generated against your drug, allowing you to determine efficacy and safety with confidence. Our experienced team applies a fit-for-purpose approach to your immunogenicity testing program that accounts for your drug’s MOA, regulatory requirements, and your overall program needs.
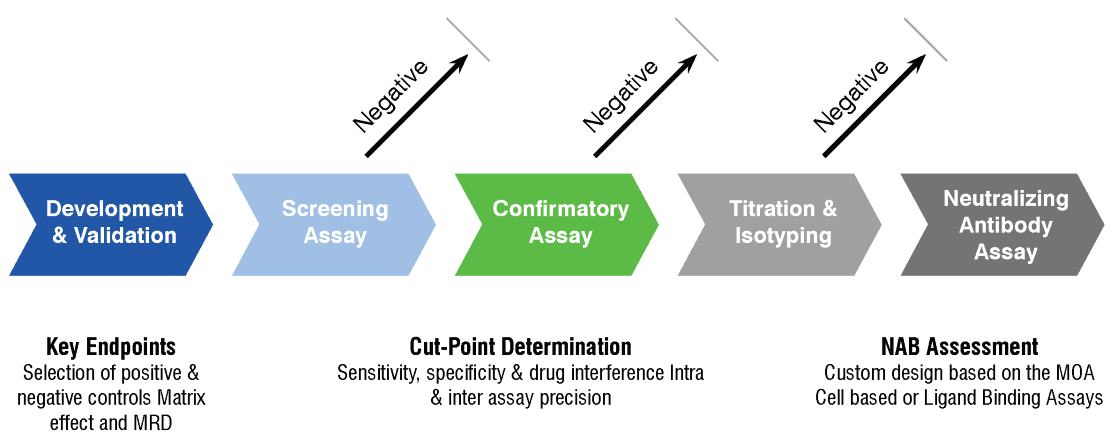
While selected assays may vary by therapeutic, step-by-step immunogenicity testing cascade is generally as follows:
Bioanalytical Strategies Unlocked: Innovative Approaches to PK, Immunogenicity, and Biomarkers
In this webinar, our scientific experts will walk you through real-world case studies that illustrate how innovative bioanalytical approaches can help you overcome common hurdles in pharmacokinetics (PK), immunogenicity, and biomarker applications.
Watch the Replay
Immunogenicity Service Capabilities
Given the need to perform immunogenicity testing throughout the development cycle of your therapeutic, you will want to ensure you have access to test facilities where and when you need them. With 12 sites across North America and Europe, Charles River can accommodate studies on your timelines using a variety of platform technologies, including:
- ELISA
- Meso Scale Discovery (MSD)/Electrochemiluminescence (ECL)
- Flow cytometry
- Gyrolab
Our team has the immunology expertise, drug development experience, and flexibility to create and execute the right testing program for your therapeutic. Consider the advantages of working with our specialists:
Ability to adapt ADA strategies
In our custom approach to immunogenicity testing, we determine your needs based on the stage of your work. In the preclinical phase, we will suggest whether your program requires anti-drug antibody (ADA) analysis is required to elucidate PK, PD, or pathology data interpretation. In the clinical phase, ADA studies may be required to monitor patient safety.
Your novel therapy has unique requirements. In a risk-based approach to preclinical immunogenicity testing, we work with you to assess your study needs and develop the proper solution, from a fit-for-purpose qualification in low-risk scenarios to fully validated assays for higher risk programs.
Using approaches ranging from single assays with a 0.1% or 1% false positive ratio, to multi-tiered analytical strategies characterizing the ADA response, we work with you to identify the best methods to support your project through the entirety of the drug development journey.
Neutralizing antibodies (nAb) assay specificities
As per the FDA guidance, the impact of ADA on PK, PD, and safety may correlate more with the nAb activity rather than ADA incidence, and therefore an assay to characterize the neutralizing activity of the detected ADAs is critical for performing immunogenicity testing in clinical studies.
We can help guide your strategies to develop, validate, and implement immunogenicity assays to detect neutralizing activity, which can range from cell-based bioassays to competitive ligand binding assay (LBA) formats to enzymatic assays, depending on the mechanism of action or known standard approach for the drug class.
Given the complexity and challenges involved in developing and validating nAb assays, especially bioassay formats, having the expertise and experience from a reputable laboratory can make all the difference in the clinical development of your drug.
eGuide: Biomarker Solutions from Discovery to Clinical Trials
Learn how strategic biomarker integration can accelerate research & de-risk pipelines to bring life-saving therapies to patients faster. This guide helps you uncover smarter ways to de-risk studies and accelerate timelines, without compromising scientific rigor.
Read the Guide
Other Immunogenicity Service Capabilities
-
Breadth of immunogenicity assay types
We’ll ensure appropriate immunogenicity assay development, utilizing a variety of methods as necessary. Our personalized approach to immunogenicity testing ensures selected assays are sensitive, specific and robust, so that the safety endpoint can be analyzed with confidence.
In addition to a standard bridging or anti-species detection format, we have extensive experience utilizing additional assay formats (see the list below) when presented with assay challenges such as high background or insufficient drug tolerance levels:
- ACE: affinity capture elution
- Acid dissociation
- SPEAD: Solid phase extraction with acid dissociation
-
Statistical support for immunogenicity testing cut point calculation
Interpreting the guidance requirements for the statistical determination of ADA assay and nAb assay cut points can be challenging. Dedicated statisticians are on hand with the expertise in immunogenicity testing and standard statistical approaches in compliance with the EMA and regulatory guidance and current literature recommendations. The team is also familiar with robust alternative approaches to address your specific requirements, complex experimental designs, and challenges like preexisting reactivity.
Learn more -
Immunogenicity testing: an efficacy measure for vaccines
Available in qualitative and quantitative assay formats, vaccine immunogenicity assays can be developed to confirm dosing and monitor efficacy.
Immunogenicity Assays & Vaccine Development
How can immunogenicity testing can accelerate vaccine development? A quantitative ELISA method combines assets of different approaches to give reliable results on vaccine efficacy with minimal time & effort.
View the PosterOur immunogenicity testing services allows you to establish the immune response to your compounds at the anti-drug antibody (ADA assay) or neutralizing antibody (nAb assay) level. At Charles River, you’ll find a dedicated multidisciplinary team of immunologists, statisticians and drug development experts who can help you devise the best strategy for your therapeutic so you can move forward to IND and monitor your drug in clinic.
For further immunogenicity support, see our monoclonal and polyclonal antibody production services to support preclinical and clinical development of immunogenicity assays.
-
When do I need to have a fully validated immunogenicity assay?
There are no guideline recommendations for nonclinical immunogenicity validation. However, for clinical assays, FDA Guidance recommends having a fully validated assay for pivotal phase trials. A fit-for-purpose suitable assay may be acceptable for early phase clinical studies if a fully validated assay cannot be used.
-
When do I need to start thinking about developing immunogenicity testing for a clinical program?
For high-risk therapeutic products, regulatory agencies (e.g., FDA) request methods to be developed at IND submission. For lower risk products, ADA assay development may be delayed, but we advise consulting regulatory agencies at IND submission to avoid adding unnecessary risk to your program.
-
How long does it take to develop an immunogenicity assay?
This depends on the robustness and availability of critical reagents. Immunogenicity testing can take anywhere from 2 weeks to a few months to develop. Typically, however, you should estimate 6-8 weeks for the development of a standard ADA assay. Consult with your assay development partner or CRO as soon as feasible to avoid delays caused by unforeseen challenges during method development (e.g. matrix interference, or insufficient drug or target tolerance).
-
For neutralizing antibody (Nab) assays, is a ligand binding or cell-based assay more suitable?
The immunogenicity testing method selected to nAb assay for ADA-positive samples should be based on the mechanism of action of the drug. There also might be known standard approaches to the assay format for the drug class of interest. It is critical to discuss this with your assay development partner or CRO to understand the advantages and disadvantages of the different formats and which is most suitable for your program based on the overall risk assessment.
- Which regulatory guidances are critical to the development and validation of immunogenicity assays?
-
What is the general starting point for method development of an ADA assay? What are the critical reagents that need to be made available?
For immunogenicity testing, selection of assay format and critical reagents is the start of the method development process. At minimum, an affinity purified positive control antibody in the same species as the study samples (or surrogate if this is not feasible) should be acquired or generated prior to starting method development work. Assays should be optimized for sensitivity and precision, and the performance of the positive control is critical to this assessment early in method development testing. The drug material will also be required and may need to be labeled during method development, depending on the assay format selected. It is advisable to consult with your assay development partner or CRO as soon as feasible to understand if other critical reagents or assay-specific reagents may be required which are not readily available.
-
Can Charles River perform immunogenicity assays for gene therapy products?
Yes. Charles River has experience with numerous viral particles such as AAVs and SARS-CoV
-
How do you know if drug tolerance is sufficient with a standard immunogenicity testing approach or if alternative approaches such as Acid Dissociation, ACE, or SPEAD are needed?
We do rely on the Sponsor to provide some insight as to the highest dose level planned and the estimated half-life of the drug. We can then look at estimated Cmax, trough levels and expected circulating concentrations of the TA at the time of ADA collections. Ideally, we would want to aim for drug tolerance to cover up to the Cmax, but if that is not feasible, we assess different formats to achieve drug tolerance that allows for proper detection at the time of ADA collection.