Antibody Characterization Services and Functionality Studies for In Vitro Antibody Validation
Our in vitro antibody validation services provide you with a complete biophysical evaluation of your antibody candidates with the goal of eliminating those that misbehave earlier in the discovery process, saving you time and money in further downstream in vivo antibody assessments. Our antibody characterization services are customizable and flexible based on your unique needs and vision for your diagnostic or therapeutic antibody and will help you answer crucial questions about your antibody candidates.
Whether you're engineering monoclonal antibodies, bispecific antibodies, or antibody-drug conjugates (ADCs), our data-rich insights are tailored for success in oncology, immunology, infectious disease, and beyond. Partner with us to de-risk your pipeline and accelerate the path to clinic-ready biologics.
Predictive In Vitro Models for Toxicity Testing
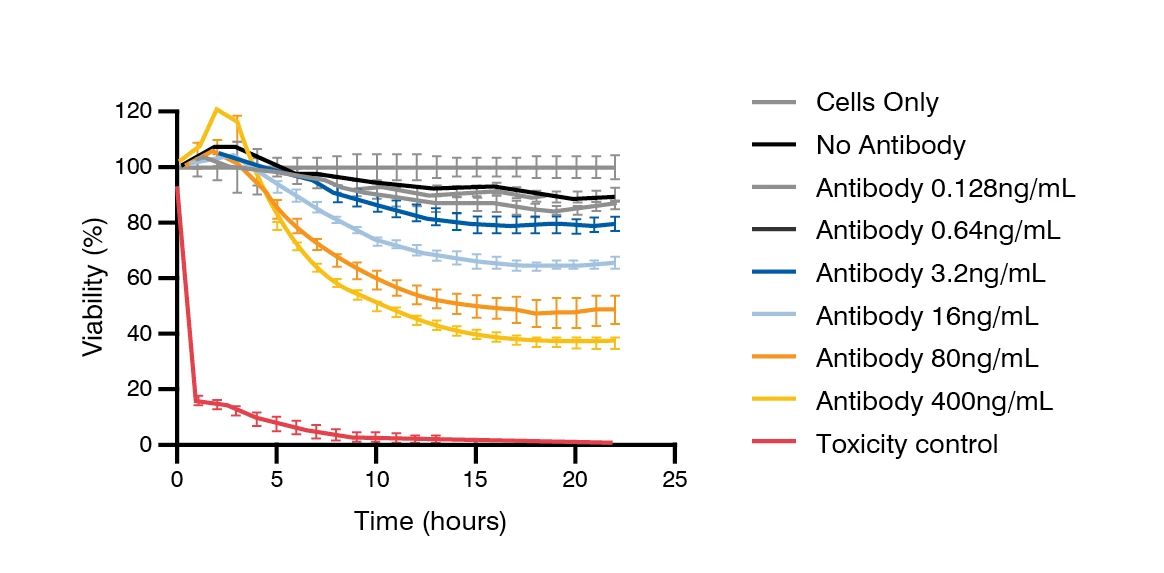
Charles River’s in vitro safety profiling and safety screening services help de-risk antibody and antibody-drug conjugate (ADC) programs by identifying potential on-target and off-target toxicities early in development. Using more than 70 optimized human primary and iPSC-derived tissue models, our assays evaluate cytotoxicity, cell viability, and cytokine release to establish a comprehensive safety profile for each candidate. Support from the scientific advisory services team with expertise in regulatory submissions is available for the selection of tissues.
Tissue panels are carefully selected based on:
- On-target Expression
- Test tissues where the target is known to be expressed
- Example: HER-2 expression in cardiomyocytes
- Potential off-target interactions (Identified via Retrogenix® screen)
- Test tissues where off-target is known to be expressed or interacts with the target
- Example: Determination of off-target interaction with protein X expressed in lung tissues via Retrogenix® screen
- Relevance to Human Toxicity
- Test major organs (~8 to 12)
- For bispecific antibodies or CAR-T therapies, often no relevant toxicology species are available
These studies are particularly valuable for bispecific antibodies and other modalities lacking predictive animal models. Data generated from these assays have supported multiple IND submissions, accelerating the advancement of safer, more effective biologics.
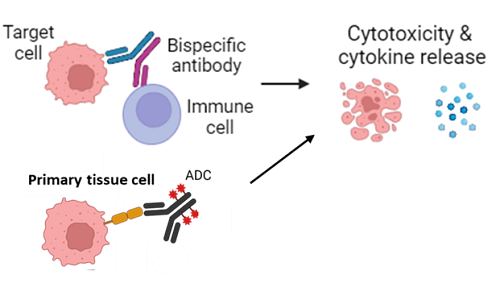
Cytotoxicity and Cytokine Release Assessment of Primary Tissues
Antibody Antigen Expression on Target Cell Systems
Antibody validation services identify relevant cell systems that express your target antigen, mimicking the clinical target expression to support downstream analysis.
- Where the target receptor is expressed – intracellular or extracellular expression
- Target expression levels – using a system with appropriate target expression can help mitigate false positives and negatives
- Prepare for downstream antibody validation success – systems suitable for functionality studies, proper antigen quantification for response predictions for antibody-based therapies
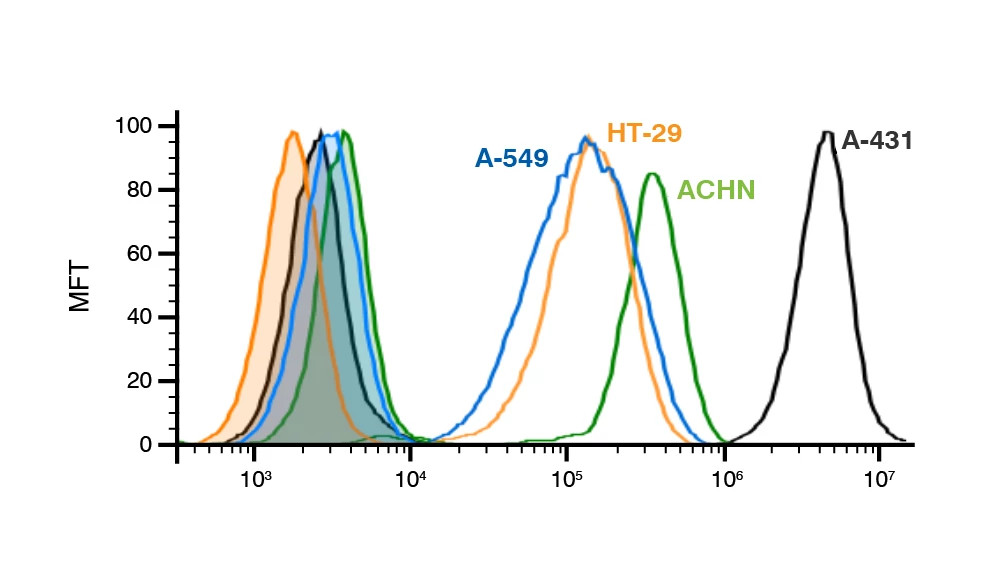
Figure 1: Blinded target expression varies across four different tumor cell lines. Quantifying expression levels aids in selection of systems for functionality studies and downstream validation of antibody candidates.
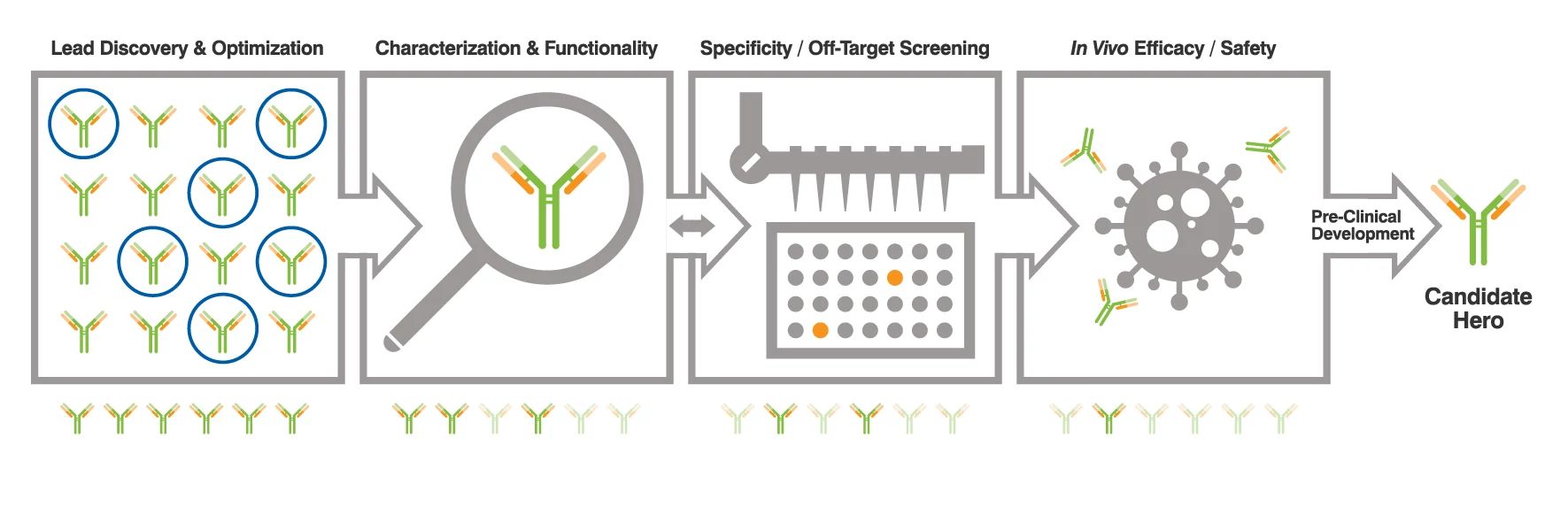
Zero to Antibody Hero: Laying the Groundwork for Preclinical Success
Are you doing everything you can to de-risk your antibody candidates? What does it take to go to preclinical development with a high degree of confidence? View this webinar to follow the journey of an antibody ‘candidate hero’ as it progresses through the early discovery gauntlet.
Watch the Webinar
Target-Specific Binding Assessment Through Antibody Characterization Services
Our antibody characterization services assess the complete antibody: receptor engagement relationship to determine the most effective candidates and best understand how your antibody will behave in complex systems.
- Evaluate binding affinity on soluble and membrane-bound targets: we offer a variety of methods for the assessment of any target binding, depending on your needs
- For soluble targets: ELISA, MSD, SPR, FRET/AlphaLISA Assays
- For membrane-bound targets: FRET-based/AlphaLISA Assays, flow cytometry
- Biochemical Characterization of the Receptor: Antibody Interaction- Assays offering nM range sensitivity and a rapid cell-free solution to aid in the development of a variety of interactions
- Supports therapeutics that competitively inhibit the receptor: ligand interaction
- Suitable for cell surface and nuclear receptors
- Amendable to high-throughput screening
- EC50 Binding Potency Assessment: supports ranking of cell lines and antibody candidates based on their binding profile
- Cell-based flow cytometry assay to determine the binding potency based on cell line expression
Target Specific Binding
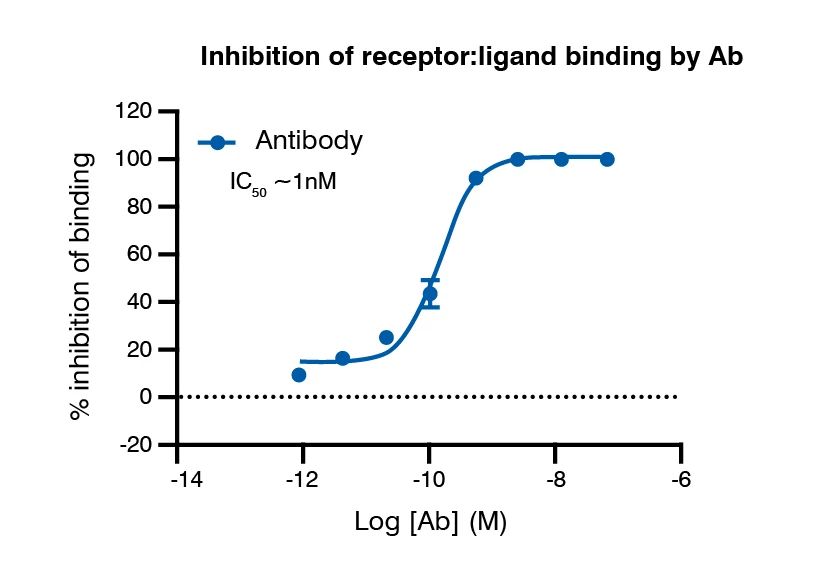
Biochemical characterization (AlphaLISA) of receptor: antibody interaction amenable to high-throughput screens.
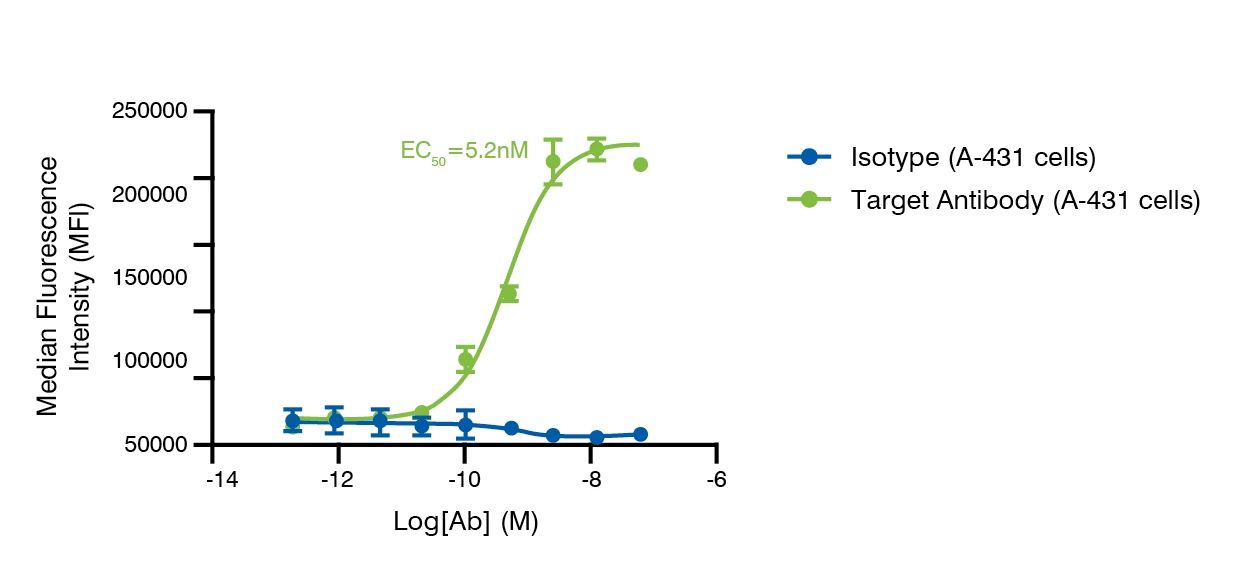
Binding potency of antibody to receptors on cells detected by flow cytometry.
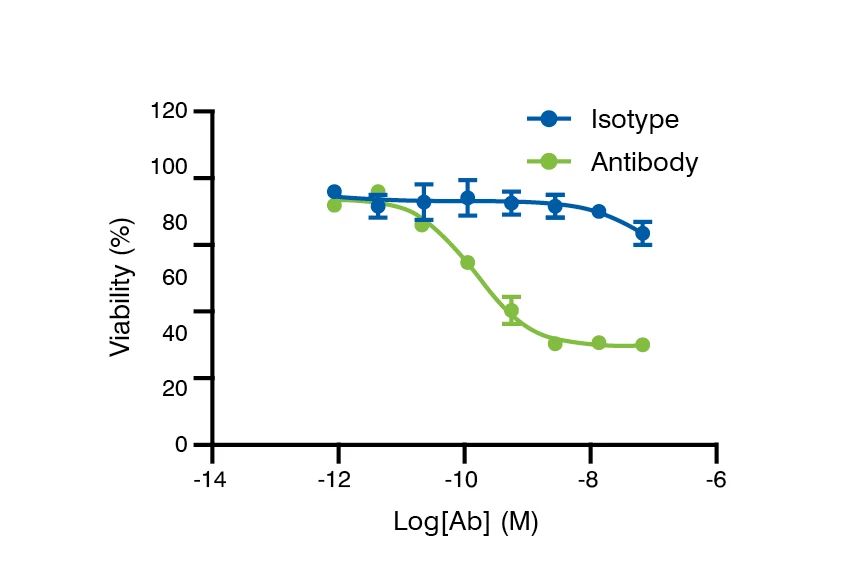
Mechanism of Action and Effector Function Studies
The functional relevance of antibody binding to its target is important to the evaluation of the potential therapeutic effects of antibodies. We provide assays to support the extensive antibody characterization of candidates.
- Activation or inhibition of enzyme activity
- Activation or inhibition of signaling pathways
- Direct target cell killing (apoptosis)
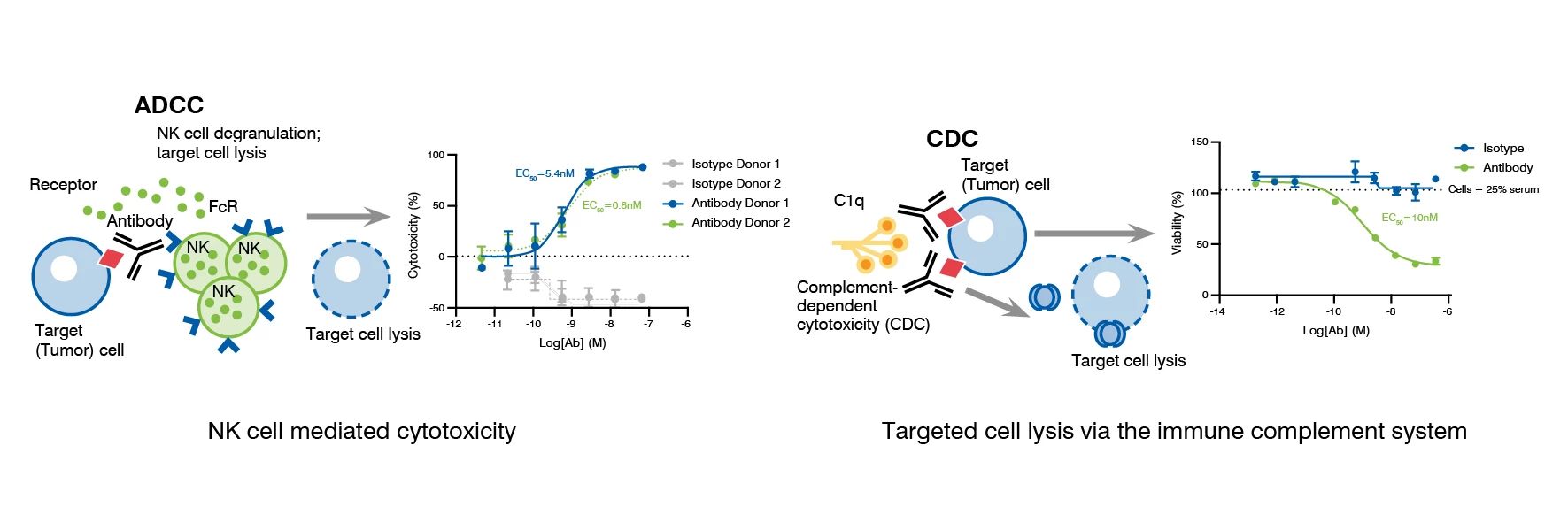
- Antibody-dependent cellular cytotoxicity (ADCC)
- Traditional mode of action using primary cell lines
- xCELLigence: Label-free approach to measure effector function using cell impedance
- Antibody-dependent cellular phagocytosis (ADCP)
- Complement-dependent cytotoxicity (CDC)
1. Mechanism of Action Studies
A. The extend of phosphorylation was assessed by the intracellular staining of the phosphorylated protein via flow cytometry. 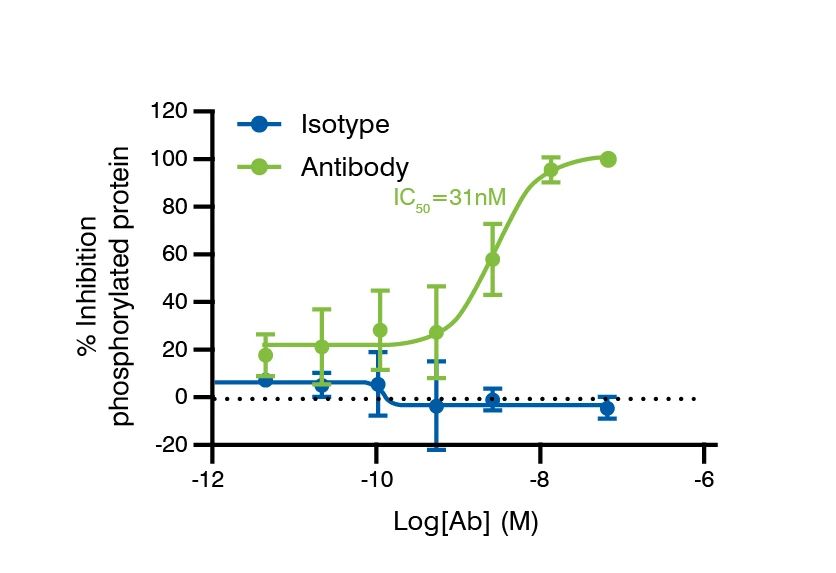
B. Effector Functions Summarized
2. Quantitative Real-Time Kinetics of Antibody Functions
A. Real-Time Effector-Mediated Cytolysis (ADCC)
and Potency Determination CDC
B. Real-Time Potency Determination (CDC)
An Integrated Antibody Discovery Experience
Our offering in antibody discovery goes beyond antibody characterization services and antibody validation. Charles River strives to be the only partner you need on your route to IND. Explore everything we have to offer in the antibody modality from discovery through development and manufacturing and learn more about the integrated candidate to clinic portfolio:
- Antibody Lead Discovery and Antibody Candidate Optimization
- Antibody In Vitro Characterization, Functionality and Safety Assessments
- Antibody Off-Target Screening and Cell-based Microarrays
- In Vivo Antibody Efficacy and Safety of Your Lead Candidates
- Safety Pharmacology
- Biologic Testing and Manufacturing Solutions
- Cell and Gene Therapies
- Microbial Solutions
WHITE PAPER- Antibodies: The Breakthrough Past, Blockbuster Present, and Cutting-Edge Future
This is an essential read for any company looking to build a biologics pipeline and seeking a deeper understanding of this advanced modality and how it is transforming the industry.
Learn More