Purification and Structural Elucidation Capabilities
You can always contract Charles River’s expert analytical chemistry services team to perform stand-alone purification and structural elucidation studies and collect the critical data you need to progress your compound. But an integrated approach is better.
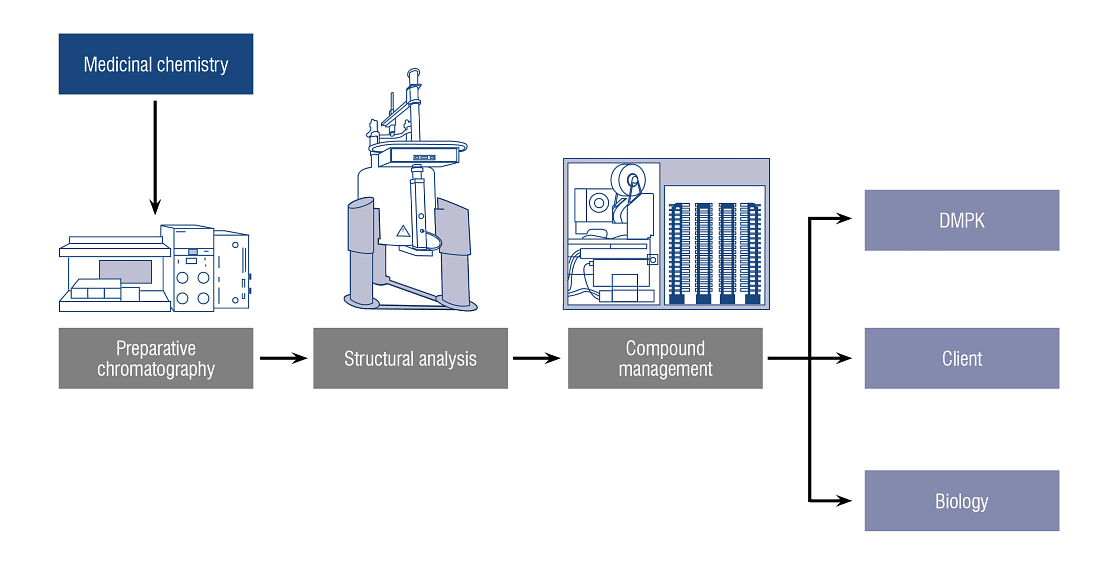
In a synchronized discovery workflow that has delivered over 100 clinical candidates and 500+ patents to our clients, our analytical chemistry team works in concert with medicinal chemistry, biology, and formulation scientists to analyze and refine your assets. Data from each cycle informs the next, moving your compound seamlessly and efficiently towards lead candidate.
Analytical Chemistry Techniques
With expertise in numerous techniques, our analytical chemistry services team can meet your most challenging requirements, from performing robust characterization to delivering routine and bespoke purification solutions, using technologies such as:
- Analytical and preparative HPLC
- Supercritical fluid chromatography
- NMR
- LC-MS
- GC-MS
Preparative HPLC Purification
Our analytical chemistry services team uses a range of reverse phase purification systems which combine standard UV-directed HPLC systems as well as mixed-mode trigger systems (UV, MS and/or ELSD). Focusing primarily on rapid turnaround of compounds from hit-to-lead and lead optimization projects, the analytical chemistry services team provides efficient separation solutions for a broad array of analytes with wide-ranging structural diversity from milligram (mg) to gram (g) scale.
Our streamlined reverse-phase screening cascades predict appropriate focused gradients, providing greater resolution of the target and impurity peaks without increasing the total run time or requiring extensive method development on most compounds. With multiple dedicated reverse phase purification systems, we can easily accommodate individual medicinal chemistry samples, small arrays, and larger libraries of compounds without compromising workflow.
For samples requiring further method development or specialist downstream techniques, we can incorporate columns and mobile phases from our extensive portfolio in parallel with our supercritical fluid chromatography and flash chromatography systems. We can also apply solid phase extraction (SPE) for buffer removal. As a standard service, we freeze dry all samples after concentrating using centrifugal evaporation.
Supercritical Fluid Chromatography
Supercritical fluid chromatography (SFC) services offer rapid enantiomeric separation of stereogenic mixtures. An essential component of validating the active conformations of most chiral compounds, the process also reduces drying time eliminating the solvent waste. When it comes to achiral separations, supercritical fluid chromatography also offers the advantages of speed and green chemistry.
Multiple SFC analytical and purification systems complement our chiral HPLC capabilities, creating a flexible approach to accommodate standard and unique requirements. Available on a milligram to multigram scale, our separation services follow processes that ensure the consistent safety and stability of your sample.
Quality Control Analytical Chemistry Services
Establishing the purity and structure of novel test compounds and pharmaceuticals is critical to the success of drug discovery programs. Once synthesized and purified by separation scientists, compounds are processed by our analytical chemistry team, undergoing rigorous characterization and structural elucidation.
Quality control (QC) studies determine if a sample is ready to progress to assay. Delivering quality compounds as efficiently as possible, QC analysts are key members of the analytical chemistry services team, helping reduce the design-make-test-analyse (DMTA) cycle of drug discovery.
Our analytical chemistry services team collaborates with many other discipline and activities, such as:
- Compound management
- Metabolite Identification
- High-throughput chemistry
- Formulation science
- Scale-up chemistry
- Pharmaceutical science
- Drug metabolism and pharmacokinetics (DMPK)
Nuclear Magnetic Resonance Spectroscopy
Whether you manufacture on site or externally, you need data that confirms the structure and purity of your potential pharmaceutical compounds and intermediates. Our team performs structural analysis of organic compounds using spectroscopic methods.
High resolution NMR spectroscopy is used extensively for structural analysis and characterization, distinguishing structural isomers and conformers, investigation of mixtures and quantitative analysis.
LC-MS Spectrometry
Our analytical chemistry services employ a range of mass spectrometers coupled to HPLC, UPLC systems and analytical SFC systems to perform structural analysis, quality control, analysis of mixtures, and metabolism identification. The systems can be operated under a variety of conditions to maximize the result and provide the highest quality data. As a supplement to the mass spectrometer, we use range of detectors (PDA, ELS, CAD) to ensure that nothing in the chromatogram is overlooked.
LC-MS is used extensively in our analytical chemistry services for analysis of chemistry and pharmaceutics screens, for physicochemical parameter measurements and any study that requires high throughput analysis. Acquiring MS data at high resolution enables us to achieve supplementary information regarding the molecular formula. Evidence is accumulated for the structure of a molecule combined with other methods such as:
- NMR
- Infrared spectroscopy
- X-ray crystallography
- Microanalysis
- Ion chromatography
From this evidence, our Analytical Sciences team can generate a professional certification of analysis (CoA) document; this is often combined with pharmaceutics details of the solid form, for regulatory submission purposes such as IND.
Library Quality Control Using High-Throughput Screening LC-MS
In the early stages of many projects, high-throughput screening (HTS) biology is a key component. Our analytics team supports HTS biology with the validation of hits through high-throughput liquid chromatography mass spectrometry (HTS LC-MS).
- Automated processing using Virscidian software delivering a robust and efficient analysis
- High capacity in support of high-throughput screening using Waters UPLC and Thermo ScientificTM Vanquish Duo for rapid, low centration analysis
- Fee-for-Service quotes available for small or large bodies of work
We also employ this process to analyze your collections, screening and validation of library components, and supporting chemistry through high-throughput chemistry analysis and metabolism screening.
Complementary Analytical and Spectroscopic Methods
The analytical sciences team at Charles River employs a range of techniques for characterization of compounds. The methods, available either internally or through our partnerships, include:
- Optical rotation via polarimeter, yielding 〖[α]〗_D for enantiomer characterization
- Infrared and UV-VIS spectroscopy
- Powdered X-ray crystallography
- Single-crystal X-ray crystallography
- ATR, DSC, GVS examination
- Measurement of pKa
- Measurement of partition coefficient (logP, logD)
- Solubility measurement in various media
- Ion chromatography
- GC-MS (Agilent 7820A GC and Agilent 5975 MSD)
- Optical and electron microscopy
- Microanalysis
- Method development package for solubility/stability assays
- High-throughput chromatographic Log D assay
Applied according to your needs, these platforms and technologies lead to detailed proof of structure credentials (PoS) and more robust patents for your APIs.
Frequently Asked Questions (FAQs) About Analytical Chemistry Services for Early Drug Discovery
-
Does Charles River have the capability for chiral purification and QC?
Our analytical chemistry services team has extensive experience purifying chiral mixtures, including the enantiomeric enrichment of compounds and multigram fee-for-service contracts. Quality control verifies the identities of all isolated compounds, confirming the exact stereogenic conformation.
-
What are your SFC capabilities?
The analytical chemistry services team can provide:
- Analytical chiral and achiral screen for method development
- Chiral purity assessment against a standard
- Chiral Separation from 5mg to over 100g of racemate
- Achiral Separation from 5mg to over 20g crude mixture
-
Can you provide a certificate of analysis for my API?
Yes, Charles River can provide a certificate of analysis for APIs. Please contact us for a quote, detailing any specific analytical chemistry techniques or requirements you wish to be included.
-
Do you have access to LC-MS libraries or compound collections?
Yes, please contact us with the details of your requirements and we will set up a consultation to discuss the libraries and timeline.
